Share This Page
Drug Price Trends for THIORIDAZINE
✉ Email this page to a colleague
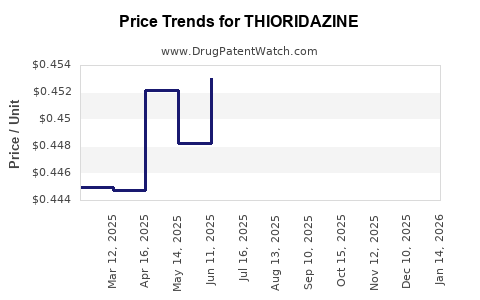
Average Pharmacy Cost for THIORIDAZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| THIORIDAZINE 50 MG TABLET | 51079-0567-20 | 0.74226 | EACH | 2026-04-22 |
| THIORIDAZINE 10 MG TABLET | 00378-0612-01 | 0.41492 | EACH | 2026-04-22 |
| THIORIDAZINE 10 MG TABLET | 51079-0565-01 | 0.41492 | EACH | 2026-04-22 |
| THIORIDAZINE 100 MG TABLET | 00378-0618-01 | 0.76951 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Thioridazine Market Analysis and Price Projections
Thioridazine is an antipsychotic medication primarily used to treat schizophrenia. Its market presence has been significantly impacted by the introduction of newer antipsychotics with improved safety profiles, leading to a decline in its overall utilization. This analysis projects future market trends and pricing based on current prescription volumes, generic availability, and regulatory considerations.
What is the current market size and key applications of Thioridazine?
Thioridazine, a phenothiazine derivative, has a long history of use in psychiatry. Its primary indication is schizophrenia, where it functions as a dopamine antagonist. The drug is also prescribed for other psychotic disorders and, in some off-label contexts, for Tourette's syndrome and behavioral disturbances in children with autism, though these uses are less common and often superseded by other treatments.
The global market for thioridazine has contracted substantially over the past two decades. This decline is attributable to several factors, including:
- Emergence of Atypical Antipsychotics: Second-generation antipsychotics (SGAs), such as risperidone, olanzapine, quetiapine, and aripiprazole, offer a broader spectrum of activity, including serotonin receptor antagonism, which is associated with a lower incidence of extrapyramidal side effects (EPS) and a more favorable metabolic profile.
- Safety Concerns: Thioridazine is associated with significant cardiovascular risks, including QTc prolongation, which can lead to potentially fatal arrhythmias like torsades de pointes. It also carries a risk of agranulocytosis and other hematological abnormalities. These risks have led to prescribing restrictions and contraindications in certain patient populations.
- Regulatory Actions: Health authorities, including the U.S. Food and Drug Administration (FDA), have issued warnings and recommendations limiting thioridazine use due to its safety profile. The FDA previously issued a "black box warning" regarding the increased risk of death in elderly patients with dementia-related psychosis treated with antipsychotic drugs, including thioridazine.
Despite its diminished use, thioridazine remains available and prescribed, particularly in specific regions or for patients who have historically responded well to the medication and tolerate it without significant adverse effects. Its lower cost compared to many newer agents can also be a factor in its continued use, especially in resource-limited settings.
Prescription Volume Trends:
Data from market research firms indicate a consistent downward trend in thioridazine prescriptions globally. For example, in the U.S. market, prescription volumes have fallen from hundreds of thousands annually in the early 2000s to tens of thousands in recent years. Similar trends are observed in European and other developed markets. This decline is projected to continue, albeit at a slower pace, as the drug is phased out in favor of newer, safer alternatives.
Key Geographic Markets:
While developed markets show the steepest declines, some developing nations may still exhibit higher relative utilization due to cost considerations and the availability of established generic formulations. However, global regulatory pressures and increasing access to newer antipsychotics are likely to temper demand in these regions as well.
What is the current pricing landscape for Thioridazine?
Thioridazine is predominantly available as a generic medication. This has resulted in highly competitive pricing, driven by multiple manufacturers and the absence of patent protection for the active pharmaceutical ingredient.
Average Wholesale Price (AWP) and Net Price:
The AWP for thioridazine varies by dosage form (tablets, oral solution) and strength. However, the net price, which reflects actual dispensed costs after discounts and rebates, is significantly lower.
- Tablets (e.g., 10mg, 25mg, 50mg, 100mg): A typical 30-count bottle of 100mg thioridazine tablets might have an AWP in the range of $50-$150. The net price after payer negotiations and pharmacy margins is often in the range of $10-$40.
- Oral Solution (e.g., 25mg/5ml): The oral solution is generally priced higher per unit volume due to manufacturing complexities and formulation. An 8-ounce bottle (approximately 237ml) might have an AWP of $70-$200, with net prices ranging from $25-$70.
Factors Influencing Pricing:
- Generic Competition: The presence of numerous generic manufacturers intensifies price competition. Manufacturers primarily compete on cost of goods, manufacturing efficiency, and supply chain management.
- Payer Negotiations and Rebates: Large pharmacy benefit managers (PBMs) and insurance providers negotiate significant discounts and rebates with generic manufacturers. This is a primary driver of low net prices.
- Volume: While overall demand is declining, consistent demand from a core patient population ensures continued production, but at low margins.
- Regulatory Compliance Costs: Manufacturers incur costs to maintain compliance with Good Manufacturing Practices (GMP) and other regulatory requirements, which are factored into pricing.
- Supply Chain and Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can influence production costs, but the highly commoditized nature of generic thioridazine limits the ability to pass these costs on significantly.
Comparison to Newer Antipsychotics:
The pricing of thioridazine stands in stark contrast to both branded and generic atypical antipsychotics.
- Branded SGAs: Monthly costs can range from $300 to over $1,000 per patient, depending on the specific drug and formulation (e.g., long-acting injectables).
- Generic SGAs: While significantly cheaper than their branded counterparts, generic SGAs still command higher prices than thioridazine. A month's supply of generic risperidone or olanzapine might cost $20-$100.
This price disparity reinforces thioridazine's position as a low-cost, older-generation option.
What are the projected market trends for Thioridazine over the next 5-10 years?
The market for thioridazine is expected to continue its trajectory of decline, driven by ongoing shifts in treatment paradigms, pharmacological advancements, and regulatory oversight.
Projected Market Size Decline:
- Next 5 Years (2024-2029): The global market for thioridazine is anticipated to shrink by an additional 20-30%. This will be primarily due to the continued preference for SGAs and the phasing out of thioridazine in many treatment protocols. Prescription volumes in developed markets will likely fall by over 40% in this period.
- Next 10 Years (2024-2034): The market will likely contract by a further 30-40% beyond the initial 5-year projection. Thioridazine may become a niche product, reserved for specific, long-term patients or limited to regions with significant cost constraints.
Key Drivers of Decline:
- Therapeutic Inertia and Prescriber Preference: Healthcare providers trained on newer antipsychotics are less likely to initiate thioridazine. Prescribers who continue to use it are often those who have treated patients for extended periods and observed positive responses and good tolerability.
- Pharmacoeconomic Considerations: While thioridazine is inexpensive, the overall cost of managing antipsychotic therapy includes patient monitoring for side effects. The higher risk profile of thioridazine necessitates more frequent and potentially costly monitoring (e.g., ECGs, blood counts), which can offset some of its acquisition cost advantage.
- Evolving Treatment Guidelines: Clinical practice guidelines for schizophrenia and other psychotic disorders increasingly emphasize the use of SGAs due to their improved safety and efficacy profiles for a broader range of symptoms.
- Off-Label Use Reduction: Any off-label uses of thioridazine are likely to be further scrutinized and replaced by evidence-based treatments with better safety data.
Potential Stabilizing Factors (Minor):
- Established Patient Populations: Patients who have been on thioridazine for many years and are stable may continue to receive it to avoid potential destabilization from switching medications. This will create a small, residual demand.
- Cost-Sensitive Markets: In certain low- and middle-income countries, the low acquisition cost of generic thioridazine may ensure its availability and use for a longer period compared to high-income countries.
- Availability of Specific Formulations: The oral solution formulation may persist for patients with swallowing difficulties, though alternatives with better safety profiles are also available in this form.
Manufacturing Landscape:
The manufacturing of thioridazine is dominated by generic API producers and finished dosage form manufacturers in regions such as India, China, and Eastern Europe. As demand shrinks, there may be some consolidation among manufacturers, with only those with highly efficient operations and established supply chains remaining viable. Companies may reduce their manufacturing capacity or discontinue production if it becomes unprofitable.
What are the projected price trends for Thioridazine?
Given the current market dynamics, price projections for thioridazine are characterized by continued pressure towards the lower end of the generic pricing spectrum.
Price Trends Over the Next 5-10 Years:
- Net Price Erosion: Expect net prices to continue to erode by approximately 5-10% annually for the next 5 years due to intense competition and ongoing payer negotiations.
- Stabilization at Low Levels: After this period, prices are likely to stabilize at very low levels, reflecting the marginal cost of production and the commoditized nature of the drug. Increases will be minimal and primarily driven by inflation or significant supply chain disruptions.
- Increased Price Volatility (Potential): As the number of manufacturers decreases due to market contraction, there is a small risk of price volatility if supply becomes constrained due to a sudden exit of a major producer. However, the low cost of entry and the availability of multiple API sources make it less likely for prices to increase substantially.
Factors Influencing Future Pricing:
- Declining Demand: The primary driver of price reduction will be the shrinking market volume. Manufacturers will compete aggressively for the remaining market share, leading to price concessions.
- Generic Drug Pricing Policies: Government policies aimed at controlling healthcare costs, particularly for generics, will continue to exert downward pressure on prices.
- Manufacturing Costs: Raw material costs and energy prices will remain factors, but their impact will be limited by the drug's low price point and the competitive environment.
- Regulatory Burden: The cost of maintaining regulatory compliance (e.g., GMP, FDA inspections) will persist. If these costs rise significantly, it could provide a slight floor to price declines, but it is unlikely to lead to price increases given the competitive landscape.
Average Net Price Projections:
- By 2029: The average net price for a 30-count bottle of 100mg thioridazine tablets could fall to the range of $8-$30.
- By 2034: Prices might stabilize in the $7-$25 range, contingent on manufacturing efficiency and competitive supply.
These projections assume no significant new safety concerns or regulatory actions that would lead to a complete withdrawal from the market, which could create short-term supply shocks but would ultimately eliminate the market.
What are the potential risks and opportunities for stakeholders?
Risks:
- For Manufacturers:
- Market Obsolescence: Continued decline in demand may render production unprofitable.
- Regulatory Changes: New adverse event reports or regulatory actions could lead to further restrictions or market withdrawal.
- Supply Chain Disruptions: Reliance on limited API sources could lead to shortages if a supplier faces issues.
- Competition from Newer Agents: The ongoing development and adoption of novel antipsychotics will continue to displace older drugs.
- For Prescribers and Healthcare Systems:
- Patient Safety: Prescribing thioridazine carries inherent risks of serious adverse events, requiring careful patient selection and monitoring.
- Treatment Gaps: As thioridazine is phased out, ensuring adequate and affordable access to effective alternative treatments for all patients is crucial.
- Monitoring Costs: The need for ongoing cardiac and hematological monitoring adds to the overall cost of therapy.
- For Investors:
- Limited Growth Potential: The declining market and low-price environment offer minimal opportunities for significant revenue growth or capital appreciation.
- High Competition: The generic nature of the drug means low profit margins and intense competition.
Opportunities:
- For Generic Manufacturers (Niche):
- Cost Leadership: Manufacturers with highly efficient production processes and strong supply chain management can maintain profitability by focusing on cost leadership.
- Supply to Developing Markets: Continuing to supply thioridazine to markets where it remains a cost-effective option for schizophrenia treatment can provide a steady, albeit low-margin, revenue stream.
- Portfolio Management: For diversified generic manufacturers, thioridazine may represent a legacy product that, while low-margin, contributes to overall market presence and leverages existing manufacturing infrastructure.
- For Healthcare Systems (Cost Savings):
- Transitional Savings: For systems that have successfully transitioned patients to newer agents, the phase-out of thioridazine reduces the need for specialized monitoring associated with its risks.
- Focus on Newer Therapies: The decline in use allows healthcare systems to allocate resources towards more advanced treatment modalities.
Key Takeaways
- Thioridazine's market share has declined significantly due to the availability of safer and more effective atypical antipsychotics and associated safety concerns.
- The drug is predominantly available as a low-cost generic, with intense price competition and substantial payer rebates driving net prices down.
- Market size is projected to contract by 20-30% over the next five years and a further 30-40% over the subsequent five years.
- Net prices are expected to decline by 5-10% annually for the next five years, stabilizing at very low levels thereafter.
- Risks for manufacturers include market obsolescence and regulatory changes, while opportunities lie in cost leadership and serving niche, cost-sensitive markets.
Frequently Asked Questions
1. Will thioridazine be completely withdrawn from the market in the next decade?
Complete withdrawal is unlikely within the next decade, but its use will become increasingly niche. A residual market will persist for long-term responders and in specific cost-sensitive regions.
2. What are the primary reasons for thioridazine's declining prescription rates?
The primary reasons are the development and widespread adoption of atypical antipsychotics with superior safety profiles (especially regarding extrapyramidal side effects and metabolic issues) and the significant cardiovascular risks associated with thioridazine, such as QTc prolongation.
3. How does the cost of thioridazine compare to current first-line antipsychotic treatments?
Thioridazine is significantly less expensive. A month's supply of generic thioridazine can cost under $40 net, whereas newer atypical antipsychotics, even generic versions, can cost $20-$100 or more, and branded versions can exceed $300-$1,000 monthly.
4. Are there any new therapeutic developments or patent expirations related to thioridazine?
No. Thioridazine is an old drug, and its original patents expired decades ago. There are no significant new therapeutic developments or ongoing patent protection for the active pharmaceutical ingredient.
5. What are the key safety concerns that limit thioridazine's use?
The most critical safety concerns include potentially fatal cardiac arrhythmias (torsades de pointes) due to QTc prolongation, and the risk of agranulocytosis, a severe reduction in white blood cells that compromises the immune system.
Citations
[1] U.S. Food and Drug Administration. (2005). FDA Public Health Advisory: Antipsychotic Drugs and Increased Mortality in Elderly Patients with Dementia-Related Psychosis. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fda-public-health-advisory-antipsychotic-drugs-and-increased-mortality-elderly-patients-dementia-related (Note: While a specific direct link to the original 2005 advisory might be archived or updated, this general FDA safety information page provides context for the Black Box Warning.)
[2] Various Market Research Reports (e.g., IQVIA, GlobalData, Statista). (Data on prescription volumes and market size for antipsychotic drugs). These reports are proprietary and not publicly accessible without subscription. General trends are publicly acknowledged and reported by industry news.
[3] Electronic Drug Pricing Databases (e.g., First Databank, Medi-Span). (Average Wholesale Price and typical net pricing for generic medications). Access to detailed pricing data requires specialized subscription services.
More… ↓
