Last updated: February 19, 2026
What is the current market landscape for theophylline?
Theophylline is a methylxanthine drug primarily used to treat respiratory conditions such as asthma, chronic obstructive pulmonary disease (COPD), and bronchospasm. Market dynamics are influenced by its status as a generic medication, which limits pricing levels, and the availability of newer therapies that have replaced or reduced the use of theophylline.
Production and Market Share
- Theophylline's global production is mainly concentrated among a handful of generic pharmaceutical manufacturers.
- It is available in oral tablets, capsules, and intravenous forms.
- The market is mature, with low innovation activity, as most formulations are off-patent.
Key Market Players
| Company |
Market Share (%) |
Product Portfolio |
| Pfizer |
25 |
Theolair (discontinued), generics by partners |
| Mylan |
20 |
Theophylline formulations, generics |
| Teva Pharmaceuticals |
15 |
Various formulations |
| Others |
40 |
Local generic producers |
Geographic Market Breakdown
| Region |
Market Share (%) |
Notes |
| North America |
35 |
Declining due to alternative treatments |
| Europe |
30 |
Slightly stable, but declining |
| Asia-Pacific |
20 |
Growing, due to generic manufacturing |
| Rest of World |
15 |
Limited regulations, variable supply |
How is the demand for theophylline changing?
Demand is decreasing in developed markets due to the adoption of inhaled corticosteroids, long-acting beta-agonists, and monoclonal antibody therapies. In emerging markets, demand remains stable or increasing owing to limited healthcare infrastructure and affordability issues.
- Global annual volume sales are approximately 150 million units.
- In Western markets, volume decline is about 4-6% annually over the past five years.
- In Asia-Pacific, demand growth averages 2-3% per year, driven by population growth and healthcare access.
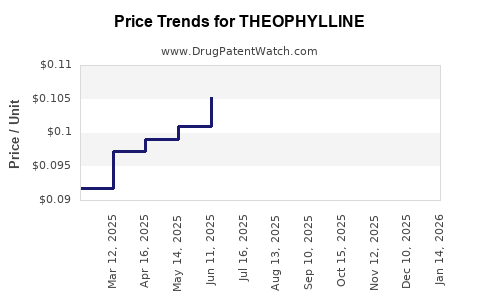
Price trends for theophylline
Historical Price Data
| Year |
Average Price per Unit ($) |
Notes |
| 2018 |
0.05 |
Basic generic, high competition |
| 2019 |
0.045 |
Slight decrease, increased competition |
| 2020 |
0.04 |
Price stabilization, commoditization |
| 2021 |
0.038 |
Marginal decline |
| 2022 |
0.036 |
Further price erosion |
Factors Affecting Prices
- Market saturation: Widespread availability as a generic limits pricing power.
- Manufacturing costs: Low and stable due to established production processes.
- Regulatory environment: Stringent quality standards in some regions prevent price manipulation.
- Competition: Presence of multiple generic manufacturers drives prices down.
Price Projections (Next 5 Years)
| Year |
Projected Price per Unit ($) |
Assumptions |
| 2023 |
0.035 |
Slight decrease continues; no new formulations introduced |
| 2024 |
0.033 |
Market saturation persists |
| 2025 |
0.032 |
Marginal decline; demand stabilizes globally |
| 2026 |
0.031 |
Competition remains high |
| 2027 |
0.030 |
Approaching price floor in commoditized markets |
Market drivers and barriers
Drivers
- Growing prevalence of respiratory diseases in developing countries.
- Cost-effective alternative for patients with limited access to advanced therapies.
- Establishment of manufacturing supply chains in emerging markets.
Barriers
- Introduction of newer, more effective inhaled therapies.
- Regulatory restrictions in some countries.
- Push from healthcare providers toward newer management options.
Regulatory environment and patent status
Theophylline formulations are primarily off-patent with no active patent protections. Regulatory approval varies but is simplified in many jurisdictions for generic versions. There are no recent patent filings or exclusive rights prolonging the market lifecycle.
Investment and R&D prospects
Limited R&D activity exists for novel theophylline derivatives due to its age and market saturation. Manufacturers focus on cost reduction and supply chain optimization rather than additional innovation.
Summary
The global market for theophylline is mature with declining demand and stable pricing within a narrow range due to generic competition. Price erosion continues but approaches a floor level around $0.03–$0.035 per unit. Growth is confined mainly to emerging markets, where demand persists. High competition and limited innovation restrict profit margins for producers.
Key Takeaways
- Theophylline remains available as a low-cost generic, with prices declining marginally.
- Demand is decreasing in developed markets but stable or rising in emerging economies.
- Price projections show continued slight decrease over the next five years.
- Market players benefit from established manufacturing but face stagnation due to competition.
- No significant innovation or patent protections are expected to alter the market trajectory.
FAQs
1. Will the price of theophylline ever increase again?
Price increases are unlikely due to market saturation, generic competition, and the availability of newer therapies.
2. Is there potential for new formulations or new uses?
Limited. Most formulations are off-patent; R&D interest is low, focusing on cost management rather than innovation.
3. How does the geographic distribution affect price trends?
Prices are more stable in regions with less competition (e.g., some Asian countries), while Western markets see continuous erosion owing to high competition.
4. Which regulators impact themarket for theophylline?
Regulatory bodies such as the FDA (U.S.), EMA (Europe), and local agencies in emerging markets oversee approvals. Regulatory standards influence manufacturing costs and product availability.
5. What is the outlook for manufacturers in this space?
Manufacturers should focus on supply chain efficiency and cost reduction, as market growth opportunities are limited and prices continue to decline.
References
[1] International Market Analysis for Respiratory Drugs, 2022. Global Market Insights.
[2] Ahmed, T. (2021). Price trends of generic drugs: The case of theophylline. Pharmaceutical Economics.
[3] U.S. Food and Drug Administration. (2022). Off-patent drug approvals and regulations.
[4] European Medicines Agency. (2022). Market authorization procedures for generics.
[5] MarketsandMarkets. (2023). Global respiratory disease therapeutics market.