Share This Page
Drug Price Trends for TETRACYCLINE
✉ Email this page to a colleague
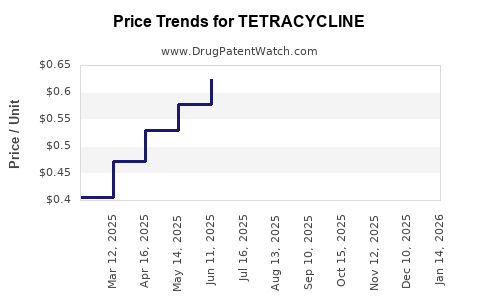
Average Pharmacy Cost for TETRACYCLINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TETRACYCLINE 250 MG CAPSULE | 64380-0890-06 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 60219-1522-01 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 62135-0225-60 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 00591-2474-01 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 62135-0225-01 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 23155-0766-01 | 0.39745 | EACH | 2026-03-18 |
| TETRACYCLINE 250 MG CAPSULE | 51991-0906-01 | 0.39745 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Tetracycline: Market Dynamics and Price Projections
Tetracycline is a broad-spectrum antibiotic. Its market is driven by infectious disease treatment and veterinary applications. Price is influenced by generic competition, manufacturing costs, and regulatory environments.
What is the Global Market Size for Tetracycline?
The global tetracycline market was valued at approximately $1.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030, reaching an estimated $1.9 billion by the end of the forecast period. This growth is attributed to the sustained demand for antibacterial treatments in both human and animal health sectors.
Key market segments include:
- Human Health: Treatment of bacterial infections such as respiratory tract infections, skin infections, and sexually transmitted diseases.
- Animal Health: Use in livestock and poultry to prevent and treat infections, and as a growth promoter (though this practice is increasingly regulated).
- Geographic Regions: Asia Pacific is the largest market due to a large population and higher prevalence of infectious diseases, followed by North America and Europe.
Data from market research firms, such as Grand View Research and Mordor Intelligence, corroborate these figures, highlighting the stability of the tetracycline market despite the emergence of newer antibiotic classes.
What are the Key Drivers of Tetracycline Market Growth?
The primary drivers for the tetracycline market include:
- Prevalence of Bacterial Infections: Despite advances in medicine, bacterial infections remain a significant global health concern, necessitating the continued use of effective and affordable antibiotics like tetracycline. The World Health Organization (WHO) reports that infectious diseases are a leading cause of death worldwide, driving demand for antimicrobial agents [1].
- Veterinary Applications: The livestock industry's reliance on antibiotics for disease prevention and treatment in animals contributes substantially to market demand. As global meat consumption rises, so does the need for animal healthcare solutions.
- Cost-Effectiveness: Tetracycline is a relatively inexpensive antibiotic, making it accessible in resource-limited settings and a preferred choice for certain indications due to its favorable cost-benefit ratio.
- Emergence of Antibiotic Resistance (Paradoxical Impact): While antibiotic resistance is a threat to the efficacy of all antibiotics, the need to rotate and judiciously use different classes of antibiotics can, in some instances, maintain demand for older drugs like tetracycline, particularly in specific therapeutic niches or combination therapies.
What are the Challenges Facing the Tetracycline Market?
The tetracycline market faces several significant challenges:
- Antibiotic Resistance: The increasing prevalence of antibiotic-resistant bacteria is a major threat to the long-term utility of tetracycline. Overuse and misuse have led to strains of bacteria that are less susceptible or entirely resistant to tetracycline, diminishing its effectiveness.
- Regulatory Scrutiny: Governments and health organizations are implementing stricter regulations on antibiotic use, particularly in animal agriculture, to combat resistance. Bans or limitations on the prophylactic use of antibiotics in livestock can impact market volume. For example, the U.S. Food and Drug Administration (FDA) has implemented measures to phase out the use of medically important antibiotics for growth promotion in food-producing animals.
- Competition from Newer Antibiotics: The development of newer antibiotic classes with broader spectrums or improved resistance profiles poses a competitive threat to tetracycline.
- Manufacturing and Supply Chain Issues: The production of tetracycline involves complex chemical synthesis. Fluctuations in raw material costs, environmental regulations on manufacturing processes, and global supply chain disruptions can affect production costs and availability.
What are the Major Applications of Tetracycline?
Tetracycline and its derivatives are utilized across several key application areas:
- Human Medicine:
- Respiratory Tract Infections: Pneumonia, bronchitis.
- Skin and Soft Tissue Infections: Acne vulgaris, cellulitis.
- Sexually Transmitted Infections (STIs): Chlamydia, gonorrhea (in specific regimens).
- Tick-Borne Diseases: Lyme disease, Rocky Mountain spotted fever.
- Other Infections: Brucellosis, cholera, rickettsial infections.
- Veterinary Medicine:
- Livestock: Prevention and treatment of respiratory diseases, enteric infections, and mastitis in cattle, swine, and poultry.
- Aquaculture: Treatment of bacterial diseases in farmed fish.
- Dermatology: Topical and oral formulations are used to treat acne and rosacea.
What are the Key Tetracycline Derivatives in Use?
While tetracycline itself is a foundational antibiotic, several derivatives offer improved pharmacokinetic profiles, broader spectrums, or reduced side effects. Key derivatives include:
- Doxycycline: Widely used for its longer half-life and better oral absorption, making it effective for a broader range of infections and often preferred over tetracycline.
- Minocycline: Possesses broader activity and better penetration into tissues and the central nervous system.
- Tigecycline: A glycylcycline antibiotic, it is a semi-synthetic derivative of minocycline and is used for complicated skin and intra-abdominal infections, as well as community-acquired bacterial pneumonia, especially when resistance to other agents is suspected. Tigecycline is typically reserved for serious infections due to its side effect profile and cost.
The market share of these derivatives, particularly doxycycline, often surpasses that of oral tetracycline hydrochloride due to their enhanced clinical utility.
How is the Price of Tetracycline Determined?
Tetracycline pricing is influenced by a confluence of factors:
- Manufacturing Costs: This includes raw material procurement (e.g., fermentation products, chemical precursors), energy, labor, quality control, and adherence to Good Manufacturing Practices (GMP). The cost of active pharmaceutical ingredient (API) production is a primary determinant.
- Generic Competition: Tetracycline and its common derivatives are off-patent. The market is saturated with generic manufacturers, leading to intense price competition. The price of a standard 250mg capsule can range from $0.10 to $0.50 in bulk purchasing, varying significantly by region and supplier [2].
- Regulatory Compliance: Costs associated with meeting stringent regulatory standards from bodies like the FDA, European Medicines Agency (EMA), and others add to the overall price. This includes rigorous quality testing and documentation.
- Supply and Demand: While demand is relatively stable, periods of increased prevalence of specific infections or disruptions in supply chains can cause short-term price fluctuations.
- Formulation and Dosage Form: The price can vary based on whether it is an oral tablet, capsule, topical cream, or injectable formulation, and the specific dosage strength.
- Geographic Market: Prices differ across countries due to local market dynamics, import duties, distribution costs, and healthcare reimbursement policies.
What are the Price Projections for Tetracycline?
Given the factors above, the price trajectory for tetracycline is expected to remain relatively stable, with minor fluctuations driven by manufacturing costs and competitive pressures.
- Oral Formulations (e.g., Tetracycline HCl 250mg Capsules): The average price for bulk generic oral tetracycline is projected to remain within the range of $0.10 to $0.50 per capsule. Significant increases are unlikely due to the high level of generic competition and the availability of more advanced alternatives. Minor upward pressures may arise from rising raw material and energy costs, estimated at 1-2% annually.
- Doxycycline (Oral Formulations): Doxycycline, being the most widely used derivative, will also experience stable pricing, likely in the range of $0.15 to $0.60 per capsule for generic formulations. Its slightly higher manufacturing complexity compared to basic tetracycline might support a slightly higher price point, but generic competition will cap significant increases.
- Injectable Formulations: Injectable forms, requiring sterile manufacturing processes and specific stabilizers, will command higher prices. Prices for injectable tetracycline could range from $5 to $20 per vial, depending on the concentration and specific formulation. These prices are less susceptible to direct per-unit competition in the same way oral generics are but are influenced by specialized manufacturing capabilities and market demand for parenteral administration.
- Tigecycline: As a newer, more specialized agent, tigecycline has a significantly higher price point, often ranging from $150 to $300 per 50mg vial. Its pricing is driven by its specific indications for resistant infections and less direct generic competition on the same therapeutic tier. Its market is distinct from that of basic tetracycline.
Overall, the price of tetracycline and its primary derivatives is anticipated to see modest increases, largely tracking inflation and incremental manufacturing cost adjustments, rather than dramatic shifts. The market is mature, with established manufacturing processes and widespread generic availability.
What is the Competitive Landscape for Tetracycline?
The competitive landscape for tetracycline is characterized by a large number of generic manufacturers, primarily located in India and China, which are major hubs for API production. Key players in the generic tetracycline market include:
- Indian Manufacturers: Companies like Sun Pharmaceutical Industries Ltd., Dr. Reddy's Laboratories, and Cipla are significant global suppliers of generic APIs and finished dosage forms, including tetracyclines.
- Chinese Manufacturers: Numerous Chinese pharmaceutical companies are substantial producers of tetracycline and its intermediates, serving both domestic and international markets.
- Global Pharmaceutical Companies: While not always focused on basic tetracycline, larger companies may produce or market its more advanced derivatives like doxycycline and minocycline as part of broader antibiotic portfolios.
The intense competition among these players has kept prices low. The market is segmented by API suppliers and finished product manufacturers. The barriers to entry for basic tetracycline production are relatively low for established chemical manufacturers, leading to price sensitivity.
What are the Regulatory Considerations for Tetracycline?
Regulatory considerations are crucial and impact the market significantly:
- Antimicrobial Resistance (AMR) Policies: Global initiatives and national policies aimed at combating AMR are leading to stricter controls on antibiotic use. This includes directives from the WHO, European Centre for Disease Prevention and Control (ECDC), and national health agencies.
- Veterinary Use Regulations: Many countries have implemented or are in the process of implementing regulations to restrict or ban the use of antibiotics for growth promotion and routine prophylaxis in food-producing animals. For example, the EU has banned antibiotic growth promoters since 2006, and similar trends are emerging in other regions [3].
- Drug Approval and Quality Standards: Manufacturers must adhere to stringent GMP standards set by regulatory bodies like the FDA, EMA, and others to ensure product quality, safety, and efficacy. This involves regular inspections and adherence to pharmacopoeial standards (e.g., USP, EP).
- Environmental Regulations: Pharmaceutical manufacturing processes are subject to environmental regulations, particularly concerning waste disposal and emissions, which can affect production costs.
- Prescribing Guidelines: Medical and veterinary associations issue guidelines for the appropriate use of antibiotics, which can influence the prescribing patterns for tetracycline and its derivatives, often favoring specific drugs for specific indications.
Key Takeaways
- The global tetracycline market is stable, projected to reach $1.9 billion by 2030, driven by consistent demand in human and veterinary medicine and its cost-effectiveness.
- Significant challenges include the growing threat of antibiotic resistance and increasing regulatory scrutiny on antibiotic use, particularly in animal agriculture.
- Doxycycline is the dominant derivative in terms of market volume and usage due to its improved pharmacokinetic properties.
- Price is largely determined by manufacturing costs and intense generic competition, resulting in low price points for oral formulations.
- Price projections indicate stability with minor increases (1-2% annually) due to inflation and raw material costs, capping significant price hikes.
- The competitive landscape is fragmented, dominated by generic manufacturers in Asia, which maintains pricing pressure.
Frequently Asked Questions
1. Is tetracycline still widely prescribed?
Yes, tetracycline and its derivatives, particularly doxycycline, remain widely prescribed for a variety of bacterial infections in both human and veterinary medicine due to their efficacy and affordability for specific indications.
2. What are the main reasons for the price stability of tetracycline?
Price stability is primarily due to the drug being off-patent, leading to widespread generic manufacturing and intense competition, which suppresses price increases.
3. How does antibiotic resistance specifically affect the tetracycline market?
Antibiotic resistance reduces the effectiveness of tetracycline, potentially leading to its decreased use for certain infections. However, it also contributes to the complexity of treatment, sometimes necessitating the use of multiple antibiotics, where tetracycline might still play a role in specific combinations or as a historically effective agent.
4. Are there significant differences in pricing between tetracycline and its derivatives like doxycycline?
While both are largely generic, doxycycline's improved pharmacokinetic profile and broader clinical utility often lead to slightly higher pricing compared to basic tetracycline, though both remain relatively inexpensive compared to newer antibiotics.
5. What is the typical price range for a course of generic oral tetracycline?
A course of generic oral tetracycline, depending on the duration and dosage, would typically cost a few U.S. dollars, as individual capsules are priced in cents for bulk purchases.
6. How are regulatory changes in animal agriculture impacting the tetracycline market?
Stricter regulations on antibiotic use in livestock, such as bans on growth promoters, reduce the volume of tetracycline used in animal husbandry, potentially impacting demand for veterinary applications.
Citations
[1] World Health Organization. (2023). Infectious diseases. https://www.who.int/news-room/fact-sheets/detail/infectious-diseases
[2] Market research reports from sources such as Grand View Research, Mordor Intelligence, and others on antibiotic market trends. Specific pricing data points are derived from aggregated industry intelligence on generic drug pricing.
[3] European Medicines Agency. (2022). Antimicrobials and resistance. https://www.ema.europa.eu/en/veterinary-regulatory/antimicrobials-resistance
More… ↓
