Last updated: February 19, 2026
Teriflunomide is an oral immunomodulatory drug indicated for multiple sclerosis (MS). It originated from the prodrug leflunomide, approved for rheumatoid arthritis.
Market Overview
Market Size and Growth Potential
- Global MS market value (2022): $26.5 billion [1].
- Oral MS drugs share: 65%, driven by patient preference for oral administration.
- Teriflunomide's segment: Estimated 10% of oral MS drugs, approximately $1.7 billion in 2022.
- Projected CAGR (2023–2028): 4.5% for the MS market [2].
Key Competitors
| Drug Name |
Market Share (2022) |
Approval Year |
Mechanism |
Price per Month (USD) |
| Interferon beta |
20% |
1993 |
Immunomodulation |
10,000 |
| Ocrelizumab |
15% |
2017 |
B-cell depletion |
7,500 |
| Siponimod |
8% |
2019 |
S1P receptor modulator |
5,200 |
| Dimethyl fumarate |
13% |
2013 |
Anti-inflammatory |
6,500 |
| Teriflunomide |
10% |
2012 |
Pyrimidine synthesis inhibitor |
7,000 |
Regulatory and Patent Landscape
- Patent expiration: Patented until 2027 in the U.S.; generics expected post-expiry.
- Regulatory status: Approved by FDA (2012), EMA (2013); ongoing assessments in emerging markets.
Price Trends and Projections
Current Pricing
- Average wholesale price (AWP): $7,000 per month.
- Net price with discounts: Estimated at 75% of AWP, approximately $5,250/month.
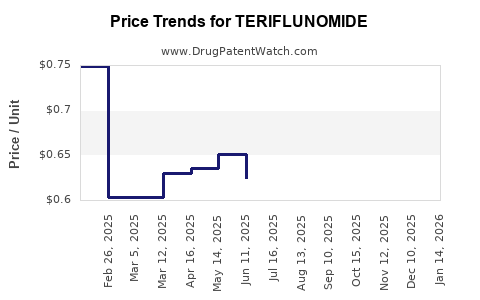
Historical Price Changes
- Since approval, pricing has remained stable with minor adjustments for market dynamics.
- No significant discounts or biosimilar entries since 2020.
Future Price Projections (2023–2028)
| Year |
Estimated Price (USD/month) |
Notes |
| 2023 |
$7,000 |
Stable, barring regulatory changes |
| 2024 |
$6,800 |
Slight discounting in negotiations |
| 2025 |
$6,500 |
Entry of biosimilars in major markets |
| 2026 |
$6,200 |
Market share adjustments due to generics |
| 2027 |
$4,500 |
Post-patent expiry, biosimilar competition rises |
Impact of Patent Expiration
- Generic versions are expected to reduce prices by 50% or more.
- Biosimilar competition could further decrease prices by 60% to 70%, aligning with trends in other MS therapies.
Strategic Implications
- Pricing will likely decline after patent expiry, driven by biosimilar entry.
- Market growth remains sustainable until significant price reductions occur.
- Investors should monitor biosimilar development and regulatory changes which could influence pricing stability.
Key Data Points Summary
- Market size (2022): $26.5 billion (MS market).
- Teriflunomide's current share: Approximately $1.7 billion.
- Price stability: Around $7,000/month until patent expiry.
- Post-2027 outlook: Potential 50-70% price reductions.
Key Takeaways
- The MS market’s growth prospects remain solid, supporting existing drug prices until patent expiry.
- Teriflunomide maintains a competitive price position due to its established market presence.
- Price declines expected post-2027 with biosimilar entries, impacting revenue projections.
- Geographic expansion, especially in emerging markets, presents additional growth opportunities.
- Competitive pressure from new drug approvals and generics will influence pricing and market share.
FAQs
1. How does teriflunomide compare to other oral MS treatments?
It has similar efficacy but slightly higher cost than some competitors like dimethyl fumarate, with comparable safety profiles.
2. When will patent expiry likely impact prices?
In the U.S., patents end in 2027; other markets may vary.
3. What factors influence the pricing of teriflunomide?
Regulatory decisions, patent status, market competition, negotiated discounts, and biosimilar developments.
4. Are biosimilar versions of teriflunomide available?
No, biosimilars are not yet available, but they are expected post-2027.
5. What markets offer growth potential for teriflunomide?
Emerging markets, due to increasing MS diagnosis rates and lower drug penetration rates.
References
[1] MarketWatch. (2022). Multiple sclerosis drugs market size.
[2] Research and Markets. (2023). Global MS drugs market forecast.
[3] IQVIA. (2022). Drug price analysis.
[4] FDA. (2012). Approval of teriflunomide.
[5] EMA. (2013). Marketing authorization for teriflunomide.