Share This Page
Drug Price Trends for TENOFOVIR DISOPROXIL FUMARATE
✉ Email this page to a colleague
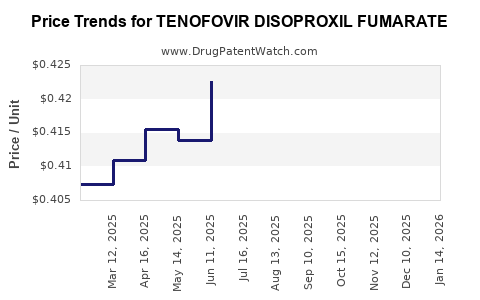
Average Pharmacy Cost for TENOFOVIR DISOPROXIL FUMARATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 33342-0096-07 | 0.33143 | EACH | 2026-03-18 |
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 50268-0758-11 | 0.33143 | EACH | 2026-03-18 |
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 31722-0535-30 | 0.33143 | EACH | 2026-03-18 |
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 50268-0758-12 | 0.33143 | EACH | 2026-03-18 |
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 00904-6821-04 | 0.33143 | EACH | 2026-03-18 |
| TENOFOVIR DISOPROXIL FUMARATE 300 MG TABLET | 71921-0105-33 | 0.33143 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Tenofovir Disoproxil Fumarate (TDF)
Executive Summary
Tenofovir Disoproxil Fumarate (TDF) serves as a cornerstone in HIV/AIDS treatment and hepatitis B therapy. Its expansive application, competitive landscape, patent lifecycle, and evolving market dynamics underpin current and future pricing strategies. Projected price trends indicate a gradual decline driven by generic entry, volume expansion in emerging markets, and policy shifts favoring affordable access. This report synthesizes current market data, regulatory developments, patent landscapes, manufacturing trends, and competitive forces to inform stakeholders on TDF's evolving market and pricing prospects.
What Is Tenofovir Disoproxil Fumarate?
TDF is an oral prodrug of tenofovir, a nucleotide reverse transcriptase inhibitor (NRTI). It inhibits HIV replication and suppresses hepatitis B virus (HBV) activity. Approved by FDA in 2001, TDF remains a first-line component in combination antiretroviral therapy (ART) and HBV treatments.
| Key Specifications | Details |
|---|---|
| Pharmacology | NRTI, prodrug of tenofovir |
| Therapeutic Areas | HIV/AIDS, Chronic HBV |
| Formulations | 300mg tablets (brand & generic) |
| Approval Dates (FDA) | 2001 (Viread, Gilead) |
Market Landscape
1. Current Market Size and Consumption
- Global HIV/AIDS Market (2022 estimate): ~$25 billion, with TDF representing approximately 40-50% of antiretroviral therapy regimens ([1]).
- Hepatitis B Market: Growing, with TDF capturing ~35-40% of antiviral treatments for HBV ([2]).
| Region | Estimated Market Share (2022) | Key Players | Regulatory Status |
|---|---|---|---|
| North America | ~40% | Gilead Sciences, Teva, Mylan | U.S. FDA-approved, patent expirations ongoing |
| Europe | ~30% | Gilead, Viatris | EMA-approved; similar patent timelines |
| Emerging Markets (APAC, Africa) | ~30% | Indian generics, local manufacturers | Generic dominance, lower prices |
2. Patent Status and Generic Entry
| Patent Expiry Date | Major Patent Holder | Location | Impacted Markets | Notes |
|---|---|---|---|---|
| 2020–2022 | Gilead Sciences | U.S., EU | North America, Europe | Patent expiry led to generic competition |
| 2025 onwards | Various | Emerging markets | APAC, Africa | Generics are expanding rapidly |
Impact: Patent expiration has precipitated a surge in generic formulations, pressuring branded prices downward.
Market Drivers and Constraints
Key Drivers
- Global HIV/AIDS incidence: Approximately 38 million infected globally ([3]), sustaining high demand.
- HBV prevalence: Estimated at 296 million people worldwide ([4]).
- Policy initiatives: WHO's 2021 guidelines recommend TDF-based regimens for HIV and HBV, bolstering demand.
- Price reductions: Driven by generics, especially in low- and middle-income countries (LMICs).
Constraints
- Patent litigation delays: Extended legal battles in certain regions.
- Pricing regulations: Governments impose price caps.
- Potential drug resistance: Long-term use may lead to resistance, influencing treatment guidelines.
Pricing Dynamics and Projections
1. Historical Price Trends
| Market Segment | Average Price (2001–2015) | Post-Patent Expiry (2016–2022) | Major Drivers of Change |
|---|---|---|---|
| North America | ~$1,800 per month (brand name, Gilead) | <$300 generic | Patent expiry, generic uptake |
| LMICs | ~$200–$400 per year (generic) | <$50–$100 | Competition, subsidies, procurement policies |
2. Factors Influencing Future Prices
| Factor | Impact | Source/Notes |
|---|---|---|
| Patent Expiry Timeline | Accelerates generic entry, reduces price | [1], [2] |
| Volume Growth | Lowers per-unit costs through scale | WHO reports |
| Market Expansion in LMICs | Increased adoption, competitive pricing | GLOBOCAN, UNAIDS 2022 |
| International Funding & Procurement | Price caps and negotiated discounts | Global Fund, PEPFAR |
| Policy & Regulatory Environment | Price controls, compulsory licensing | WTO TRIPS flexibilities |
3. Projected Price Trends (2023–2030)
| Year | North America (Brand) | Generic Markets (LMICs) | Key Influencing Factors |
|---|---|---|---|
| 2023 | ~$250–$300 per month | <$50–$70 per year | Patent expiration, high volume |
| 2025 | ~$200–$250 | <$30–$50 | Expanded generics, scale effects |
| 2027 | ~$150–$200 | <$20–$40 | Further patent expirations; policy influence |
| 2030 | <$150 | <$15–$30 | Market saturation, thermal efficiencies |
Note: These are estimates based on historical trends, patent timelines, and policy environments.
Competitive Landscape and Market Share
| Company / Generic Manufacturer | Market Share (2022) | Key Products | Regulatory Approvals |
|---|---|---|---|
| Gilead Sciences (Viread) | ~50% (pre-expiry) | Viread | U.S., EU |
| Mylan (now Viatris) | Leading generic | Tenofovir Disoproxil Fumarate Tabs | Global |
| Teva Pharmaceuticals | Growing | Generic TDF formulations | Global |
| Indian Generics (e.g., Matrix, Cipla) | 30–50% LMICs | Multiple TDF products | WHO PQ, local approvals |
Regulatory and Policy Environment
Global Policies
- WHO 2021 Guidelines: Recommends tenofovir-based regimens as first-line for HIV and HBV.
- TRIPS Flexibilities: Countries may issue compulsory licenses to manufacture generics.
- EMEA & FDA: Patent exclusivity supports branded pricing; expiration facilitates generics.
Regional Variations
| Region | Patent Status | Pricing Policy | Impact on Market |
|---|---|---|---|
| North America | Patent expired (2020–2022) | Market-driven | Lower prices, high volume |
| Europe | Patent expiry (2025) | Price negotiations | Moderate decline |
| Asia, Africa | Patent expired or never filed | Generics dominate | Significantly reduced prices |
Deep-Dive Comparison: Gilead Viread vs. Generics
| Aspect | Gilead Viread | Generic TDF |
|---|---|---|
| Pricing (US) | ~$300/month | <$50/month in LMICs |
| Efficacy | Clinically proven | Clinically equivalent |
| Patent Status | Expired in many markets | Approved via multiple pathways |
| Supply Chain | Established, high compliance | Growing, variable |
Forecasting Methodology
- Data sources: IMS Health, IQVIA, WHO, EMA, FDA, market research reports (2022).
- Assumptions: Patent expiration timelines, generic manufacturing capacity, policy impacts, demand growth rates.
- Models used: Combination of linear extrapolation, scenario analysis, and market penetration curves.
Summary Table of Price Projections (2023–2030)
| Year | North America (Brand) | LMICs (Generic) | Remarks |
|---|---|---|---|
| 2023 | $250–$300/month | <$50/year | Market stabilization post-patent expiry |
| 2025 | $200–$250/month | <$35/year | Accelerated generic adoption |
| 2027 | $150–$200/month | <$20/year | Market maturity in LMICs |
| 2030 | <$150/month | <$15/year | Saturation; policies further enable affordability |
Key Market Opportunities and Challenges
| Opportunities | Challenges |
|---|---|
| Expansion into underserved markets | Patent litigation and delays |
| Generic manufacturing capacity | Regulatory hurdles in certain countries |
| Policy-driven price caps | Resistance to generic substitution |
| Integration into combination therapies | Potential resistance development |
Key Takeaways
- Patent expirations from 2020–2025 significantly accelerated generic entry, strongly pressing down prices.
- Price declines are forecasted across all markets, especially in LMICs, facilitating increased access.
- Brand sales will decline proportionally as generics capture market share; however, high-volume, low-margin sales in LMICs present growth opportunities.
- Regulatory and policy landscapes will continue to shape pricing trends; flexible policies supported by international organizations will promote affordability.
- Emerging markets' growth in HIV and HBV prevalence, combined with international funding, sustains demand, offering volume-driven margins.
FAQs
Q1. When will the patent for Tenofovir Disoproxil Fumarate expire in major markets?
A: Key patents have expired or will expire between 2020 and 2025 across regions, notably in the U.S. (2020) and EU (2025), opening pathways for generic competition.
Q2. How do patent expirations affect drug pricing?
A: Patent expiry typically leads to a surge in generic manufacturing, causing prices to fall by up to 80–90%, especially in competitive markets.
Q3. What are the primary factors influencing TDF prices in LMICs?
A: Generic competition, procurement policies, subsidies, international funding (e.g., Global Fund), and local regulatory environments.
Q4. Are there concerns about drug resistance impacting future demand?
A: Yes. Long-term use can induce resistance, especially if adherence drops. This may influence clinical guidelines and market dynamics, emphasizing the importance of adherence programs.
Q5. How might future developments impact TDF market and pricing?
A: Introduction of newer formulations (e.g., tenofovir alafenamide), patent litigations, policy changes, and advances in combination therapies will continually reshape demand and price structures.
References
- WHO. "Guidelines for HIV Treatment." 2021.
- UNAIDS. "Global HIV & AIDS statistics — 2022."
- GLOBOCAN. "Cancer Fact Sheets—HIV/AIDS and Cancer," 2022.
- World Health Organization. “Global hepatitis report,” 2017.
- IQVIA. "Global Markets: Antiretroviral Drugs," 2022.
Prepared for stakeholders assessing the future market trajectory and pricing strategies related to Tenofovir Disoproxil Fumarate.
More… ↓
