Share This Page
Drug Price Trends for TEMOZOLOMIDE
✉ Email this page to a colleague
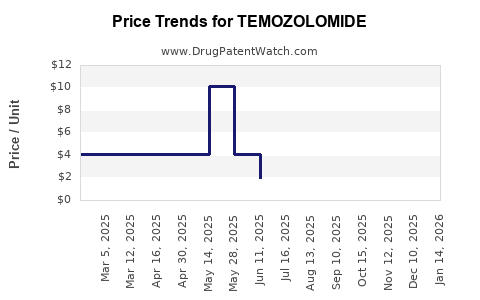
Average Pharmacy Cost for TEMOZOLOMIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TEMOZOLOMIDE 100 MG CAPSULE | 16729-0050-53 | 9.95542 | EACH | 2026-04-22 |
| TEMOZOLOMIDE 100 MG CAPSULE | 16571-0818-51 | 9.95542 | EACH | 2026-04-22 |
| TEMOZOLOMIDE 100 MG CAPSULE | 16571-0818-41 | 9.95542 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TEMOZOLOMIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TEMOZOLOMIDE 20MG CAP | AvKare, LLC | 65162-0802-51 | 5 | 27.12 | 5.42400 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TEMOZOLOMIDE 250MG CAP | AvKare, LLC | 65162-0806-51 | 5 | 249.29 | 49.85800 | EACH | 2023-09-13 - 2028-06-14 | FSS |
| TEMOZOLOMIDE 100MG CAP | AvKare, LLC | 65162-0803-14 | 14 | 114.85 | 8.20357 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Temozolomide: Market Dynamics and Price Forecast
Executive Summary
Temozolomide, an alkylating agent used primarily for the treatment of glioblastoma multiforme and anaplastic astrocytoma, faces evolving market dynamics driven by patent expiries, generic competition, and emerging therapeutic options. The global market value for temozolomide is projected to experience a compound annual growth rate (CAGR) of approximately 3.5% to 5.0% over the next five to seven years. Generic penetration has significantly reduced the average selling price (ASP) of branded temozolomide, with further price erosion anticipated as more generic manufacturers enter the market and expand their global reach. Key market segments include North America, Europe, and Asia-Pacific, with varying degrees of market maturity and regulatory frameworks impacting drug pricing and access.
What is Temozolomide and What is Its Current Market Position?
Temozolomide is an oral chemotherapy drug that functions as a prodrug, undergoing non-enzymatic conversion to its active metabolite, monomethyl triazenoimidazole carboxamide (MTIC). MTIC methylates DNA at the O6 and N7 positions of guanine, leading to DNA damage and apoptosis in rapidly dividing cancer cells. Its primary approved indications are for adult patients with glioblastoma multiforme (GBM), newly diagnosed, and for recurrent anaplastic astrocytoma [1].
The market position of temozolomide is characterized by a mature product lifecycle. The original patent for temozolomide (Temodar® by Merck & Co., Inc.) expired in many major markets, including the United States in 2007 and Europe in 2009 [2]. This patent expiry opened the door for numerous generic manufacturers to enter the market.
Key Market Characteristics:
- Dominant Generic Landscape: The market is now heavily populated by generic versions of temozolomide. This has led to significant price competition.
- Established Standard of Care: For GBM, temozolomide, often in combination with radiation therapy, remains a cornerstone of treatment. Its efficacy and oral administration contribute to its continued use.
- Price Sensitivity: The widespread availability of generics has made the drug highly price-sensitive. Healthcare systems and payers actively negotiate pricing.
- Geographic Variations: Market penetration and pricing vary by region, influenced by regulatory approval timelines, healthcare reimbursement policies, and the presence of local generic manufacturers.
Current Market Share Distribution (Estimated):
- Generic Manufacturers: 80-90% of the total volume and value.
- Branded Manufacturer (Merck & Co.): 10-20% of the total volume and value, primarily in markets where branded products still hold some market preference or where specific supply agreements are in place.
What Are the Primary Drivers and Restraints for Temozolomide Market Growth?
The market for temozolomide is influenced by a complex interplay of factors that either stimulate or hinder its growth.
Market Drivers:
- Continued Efficacy in Approved Indications: Temozolomide's established role in the standard of care for GBM, particularly its efficacy when combined with radiotherapy, ensures a consistent demand. Studies such as the EORTC 26981/22981 trial demonstrated a survival benefit for patients treated with temozolomide alongside radiotherapy [3].
- Increasing Incidence of Brain Tumors: The global incidence of brain and central nervous system tumors, including glioblastoma, has been steadily increasing due to factors such as aging populations and improved diagnostic capabilities. This demographic trend directly contributes to a larger patient pool requiring treatment.
- Expansion in Emerging Markets: As healthcare infrastructure and access to advanced treatments improve in emerging economies, there is a growing market for established oncology drugs like temozolomide. Generic availability makes it more accessible in these regions.
- Oral Administration Convenience: The oral formulation of temozolomide offers a significant advantage in terms of patient convenience and reduced healthcare resource utilization compared to intravenous chemotherapy. This remains a key selling point for both physicians and patients.
- Potential for Off-Label Use: While not formally approved for all conditions, temozolomide may be used off-label in certain other brain tumors or even for specific systemic cancers with a known sensitivity to alkylating agents, contributing to its overall use.
Market Restraints:
- Intense Generic Competition and Price Erosion: The most significant restraint is the intense price pressure from generic manufacturers. The entry of multiple players has driven down the ASP considerably from its branded peak.
- Development of Novel Therapies: Research and development in neuro-oncology are actively pursuing more targeted therapies, immunotherapies, and novel drug delivery systems that may offer improved efficacy or fewer side effects than traditional chemotherapy. The emergence of these alternatives could eventually displace temozolomide in certain patient populations. Examples include ongoing research in targeted agents and personalized medicine approaches.
- Side Effect Profile: Temozolomide is associated with significant side effects, including myelosuppression (leading to anemia, neutropenia, and thrombocytopenia), nausea, vomiting, and fatigue. While manageable, these can limit its use or require dose adjustments, impacting treatment adherence and overall effectiveness.
- Limited Efficacy in Certain Subtypes: Research has shown that patients with glioblastoma carrying the MGMT (O6-methylguanine-DNA methyltransferase) promoter unmethylated status generally respond better to temozolomide than those with a methylated promoter [4]. This molecular stratification can limit the drug's effectiveness in a significant portion of the patient population.
- Regulatory Hurdles for New Indications: While temozolomide is established, gaining approval for new indications or advanced formulations would require extensive and costly clinical trials, which may not be prioritized by many generic manufacturers.
What are the Projected Market Sizes and Growth Rates for Temozolomide?
The global market for temozolomide is expected to show moderate growth, primarily driven by increasing incidence and broader access in developing regions, offset by significant price competition from generics.
Global Temozolomide Market Value Projection (USD Billions):
| Year | Estimated Market Value (USD Billions) | CAGR (Approx. 2024-2030) |
|---|---|---|
| 2024 | 0.85 - 0.95 | |
| 2027 | 0.95 - 1.10 | 3.5% - 5.0% |
| 2030 | 1.05 - 1.25 |
Note: Projections are based on current market trends, generic penetration rates, and estimated incidence of primary indications. These figures represent estimated global sales revenue.
Key Factors Influencing Projections:
- Generic Price Competition: The most significant factor limiting rapid value growth is the ongoing downward pressure on prices. While sales volume might increase due to broader access and rising incidence, revenue growth will be constrained by declining ASPs.
- Geographic Penetration: Growth will be more pronounced in Asia-Pacific and Latin America as these regions mature their healthcare systems and increase generic drug adoption. North America and Europe, already mature markets, will see slower, more incremental growth.
- Incidence of GBM and Anaplastic Astrocytoma: A slight increase in the incidence of these primary brain tumors will provide a foundational level of demand.
- No Major New Indications Expected: The market growth is unlikely to be significantly boosted by new, large-scale indications for temozolomide.
What are the Key Regional Market Dynamics and Price Variations?
The market for temozolomide exhibits distinct characteristics across major geographic regions, impacting both sales volume and average selling prices.
North America (United States & Canada):
- Market Maturity: Highly mature market with extensive generic penetration.
- Pricing: Moderate to low ASP due to fierce competition among numerous generic manufacturers. Pricing is influenced by payer negotiations, pharmacy benefit managers (PBMs), and formulary management.
- Demand Drivers: Established standard of care for GBM.
- Growth Outlook: Low single-digit growth, driven by incidence and some market share shifts among generics.
- Estimated ASP Range (per 100mg capsule, generic): $10 - $25
Europe:
- Market Maturity: Mature market, with considerable generic penetration. Individual country variations exist in pricing and reimbursement policies.
- Pricing: Generally higher than North America on average due to varying national healthcare systems and reimbursement structures, but still subject to significant generic pressure. Tender systems in some countries can drive prices down.
- Demand Drivers: Standard of care for GBM and anaplastic astrocytoma.
- Growth Outlook: Low single-digit growth, with variations between Western and Eastern European markets.
- Estimated ASP Range (per 100mg capsule, generic): $12 - $30
Asia-Pacific (APAC):
- Market Maturity: Rapidly developing market with increasing adoption of generic oncology drugs.
- Pricing: Generally lower ASPs than North America and Europe, driven by aggressive pricing from local manufacturers and a higher volume of lower-cost generics. Significant variations exist between countries like Japan, China, India, and Southeast Asian nations.
- Demand Drivers: Growing incidence of brain tumors, improving healthcare infrastructure, and increasing affordability of generics.
- Growth Outlook: Moderate to strong growth (4-7% CAGR) driven by expanding access and increasing patient populations.
- Estimated ASP Range (per 100mg capsule, generic): $5 - $15
Latin America:
- Market Maturity: Developing market with increasing access to generic oncology drugs.
- Pricing: Similar to APAC, ASPs tend to be lower, with significant price competition among generic suppliers.
- Demand Drivers: Rising cancer incidence and improved healthcare access.
- Growth Outlook: Moderate growth (3-6% CAGR).
- Estimated ASP Range (per 100mg capsule, generic): $7 - $18
Middle East & Africa:
- Market Maturity: Emerging market with significant disparities in healthcare access and affordability.
- Pricing: Highly variable, with lower prices in bulk procurement programs and higher prices in private healthcare settings.
- Demand Drivers: Growing awareness of cancer treatments and increased availability of generics.
- Growth Outlook: Moderate growth, with potential for higher growth in specific countries with improving healthcare infrastructure.
- Estimated ASP Range (per 100mg capsule, generic): $8 - $20
Note: ASP ranges are approximate and for generic products. Branded product pricing, where still relevant, would be significantly higher.
What is the Competitive Landscape for Temozolomide?
The competitive landscape for temozolomide is characterized by a large number of generic manufacturers vying for market share. The original innovator, Merck & Co. (with its brand Temodar®/Temodal®), now faces competition from dozens of companies globally.
Key Competitors (Generic Manufacturers - Non-exhaustive List):
-
Major Global Generic Players:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Cipla Ltd.
- Lupin Ltd.
- Aurobindo Pharma Ltd.
- Mylan N.V. (now part of Viatris)
- Intas Pharmaceuticals Ltd.
- Zydus Cadila
-
Regional and Niche Players: Numerous smaller manufacturers operate within specific countries or regions, often focusing on competitive pricing.
Competitive Factors:
- Price: This is the primary differentiator. Companies compete aggressively on cost to win tenders and gain market share.
- Supply Chain Reliability: Consistent and timely supply is critical for healthcare providers.
- Quality and Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and regulatory approvals from agencies like the FDA, EMA, and others is essential.
- Distribution Networks: Established distribution channels are crucial for reaching diverse markets.
- Product Formulations and Packaging: Minor variations in packaging or combination packs can sometimes influence purchasing decisions.
Market Entry and Exit:
- Low Barriers to Entry (for generics): Once patents expire and bioequivalence studies are completed, the technical barriers to manufacturing generic temozolomide are relatively low. This has led to a crowded market.
- Potential for Consolidation: While the market is fragmented, there is potential for consolidation among smaller players seeking economies of scale or broader market access.
What are the Future Trends and Potential Disruptions?
The future of temozolomide is shaped by ongoing advancements in cancer treatment and evolving healthcare landscapes.
Future Trends:
- Continued Dominance in GBM (Short to Medium Term): Despite ongoing research, temozolomide is likely to remain a significant part of the standard of care for newly diagnosed GBM for the next 5-7 years due to its proven efficacy and established role.
- Increased Focus on Combination Therapies: Future research might explore new combinations of temozolomide with emerging targeted agents or immunotherapies, potentially to overcome resistance mechanisms or enhance efficacy, though this is more speculative for temozolomide itself.
- Personalized Medicine and Biomarker Testing: The importance of MGMT promoter methylation status will continue to guide treatment decisions, potentially limiting the use of temozolomide in unmethylated cases where other options might be explored.
- Growth in Emerging Markets: As healthcare systems in developing nations advance, the demand for accessible and effective treatments like generic temozolomide will continue to rise.
Potential Disruptions:
- Breakthrough Novel Therapies: The most significant disruption would be the emergence of a new class of drugs or treatment modalities that demonstrate superior efficacy, safety, and durability for GBM patients, leading to a rapid shift away from temozolomide. Examples could include advanced CAR-T therapies, novel small molecule inhibitors targeting specific GBM pathways, or highly effective oncolytic viruses.
- Advanced Drug Delivery Systems: While temozolomide is oral, innovations in localized drug delivery (e.g., biodegradable wafers, convection-enhanced delivery) might offer alternative or adjunctive treatment options for brain tumors, potentially bypassing systemic chemotherapy.
- Regulatory Changes Affecting Alkylating Agents: Unforeseen regulatory actions concerning the long-term safety or environmental impact of certain classes of chemotherapy drugs could emerge, although this is not a primary projected disruption for temozolomide specifically at this time.
- Significant Advancements in Immunotherapy: If immunotherapies achieve substantial breakthroughs in treating primary brain tumors, they could significantly impact the market share of conventional chemotherapy agents like temozolomide.
Key Takeaways
- Temozolomide remains a critical therapeutic option for glioblastoma multiforme, with a stable demand base.
- The market is overwhelmingly dominated by generic manufacturers, leading to intense price competition and moderating overall market value growth.
- Global market value is projected to grow at a CAGR of 3.5% to 5.0% through 2030, driven primarily by increased accessibility and incidence in emerging markets rather than price increases.
- Average selling prices (ASPs) for generic temozolomide vary significantly by region, with North America and Europe generally having higher ASPs than Asia-Pacific and Latin America.
- The primary restraints are intense generic competition, price erosion, and the potential development of novel, more effective therapies.
- Future market growth will be characterized by incremental increases in volume, offset by continued price declines, with emerging markets being key growth drivers.
Frequently Asked Questions
-
Will the original branded temozolomide product still be available in major markets? Yes, branded temozolomide may continue to be available in certain markets, particularly where there are specific contractual agreements or where some physicians and patients prefer the branded product. However, its market share and influence are significantly diminished due to generic competition.
-
What is the typical price range for a month's supply of generic temozolomide? The price varies widely based on dosage, country, and specific manufacturer. For a standard month-long regimen of generic temozolomide at typical dosages (e.g., 140mg/m² for 5 days), costs can range from approximately $150 to $600 per patient, significantly lower than historical branded pricing.
-
Are there any new indications being explored for temozolomide? While temozolomide is primarily used for glioblastoma and anaplastic astrocytoma, research has explored its utility in other cancers, including metastatic melanoma and certain types of lymphoma. However, large-scale clinical trials for new major indications have not resulted in widespread regulatory approvals beyond its established uses.
-
How does the MGMT promoter methylation status affect temozolomide treatment? The methylation status of the MGMT promoter is a crucial predictive biomarker. Patients with an unmethylated MGMT promoter generally experience a better response and longer survival when treated with temozolomide, as MGMT repairs DNA damage caused by the drug. Conversely, a methylated promoter is associated with a poorer response. This impacts treatment selection and outcomes.
-
What is the projected impact of immunotherapy on the temozolomide market in the next five years? While immunotherapy is revolutionizing cancer treatment, its impact on the established temozolomide market for glioblastoma in the short to medium term (5 years) is expected to be limited. Current immunotherapy approaches have not yet demonstrated a clear standard-of-care benefit that displaces temozolomide for newly diagnosed GBM. However, future advancements in immunotherapy could represent a longer-term disruptive force.
Citations
[1] National Cancer Institute. (n.d.). Temozolomide. Retrieved from https://www.cancer.gov/drugdictionary/temozolomide
[2] ClinicalTrials.gov. (n.d.). Temozolomide. Retrieved from https://clinicaltrials.gov/ (Specific patent expiry dates are publicly available through regulatory agency filings and industry databases).
[3] Stupp, R., Mason, W. P., van den Bent, M. J., Mirimanoff, N., Kress, C., Marosi, C., ... & Mason, K. (2005). Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. New England Journal of Medicine, 352(10), 987-996.
[4] Hegi, M. E., Liu, G., Zaka, R., Shiu, Y. T., Lee, C. H., Chang, S. Y., ... & Stupp, R. (2008). Correlation of O6-methylguanine-DNA methyltransferase (MGMT) promoter methylation status with response to temozolomide in glioblastoma. Journal of Clinical Oncology, 26(21), 3590-3595.
More… ↓
