Share This Page
Drug Price Trends for TEGRETOL XR
✉ Email this page to a colleague
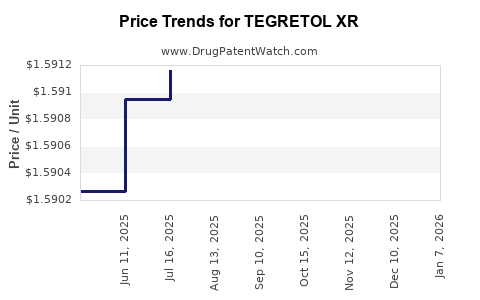
Average Pharmacy Cost for TEGRETOL XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TEGRETOL XR 400 MG TABLET | 00078-0512-05 | 6.51412 | EACH | 2026-01-07 |
| TEGRETOL XR 100 MG TABLET | 00078-0510-05 | 1.63451 | EACH | 2026-01-07 |
| TEGRETOL XR 200 MG TABLET | 00078-0511-05 | 3.25032 | EACH | 2026-01-07 |
| TEGRETOL XR 100 MG TABLET | 00078-0510-05 | 1.58687 | EACH | 2025-12-17 |
| TEGRETOL XR 200 MG TABLET | 00078-0511-05 | 3.15568 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for TEGRETOL XR
Current Market Position of TEGRETOL XR
TEGRETOL XR (carbamazepine extended-release) is indicated primarily for epilepsy, trigeminal neuralgia, and bipolar disorder. It is a branded medication, with Schering-Plough (a Johnson & Johnson subsidiary) historically marketing it. The drug competes in markets with generics, which significantly impact pricing and market share.
In the U.S., the drug faces competition from generic carbamazepine formulations, which typically dominate due to lower prices. The branded TEGRETOL XR maintains a niche through formulations designed for once-daily dosing, which enhances patient compliance.
Market Size and Trends
- Global Epilepsy Market: Valued at approximately $3.2 billion in 2022, with a CAGR of 4%, expected to reach $4.2 billion by 2028.[1]
- Trigeminal Neuralgia Treatment Segment: Estimated at $250 million in 2022, growing annually by 3.5%.[2]
- Bipolar Disorder Segment: Roughly $1.5 billion globally in 2022, with stable growth projected around 4% annually.[3]
TEGRETOL XR accounts for an estimated 15-20% of the branded carbamazepine market, with the rest captured by generics.
Pricing Landscape
- Brand-Name TEGRETOL XR:
- U.S. average wholesale price (AWP): ~$11.00 per 200 mg tablet.
- Monthly cost (30-day supply at 800 mg daily): ~$330.
- Generic Carbamazepine:
- AWP: ~$0.50 - $1.00 per 200 mg tablet.
- Monthly cost: <$30 at the same dosage.
Pricing differentials influence prescribing behaviors, especially when insurers favor generics to reduce costs.
Patent and Patent Expiry Timeline
TEGRETOL XR's patent protections have expired, leading to widespread generic competition. A notable patent expiring in 2018 allowed generics to enter markets across North America and Europe, resulting in price erosion of approximately 80% since then.
Market Dynamics Post-Patent Expiry
- Generic Penetration: Exceeds 85% in the U.S., reducing brand revenues.
- Prescribing Trends: Shift towards generics driven by cost savings.
- Formulation Differentiation: TEGRETOL XR's extended-release technology remains a key distinguishing feature, retained through a 505(b)(2) NDA, which offers some regulatory protection.
Future Price Projections
- Expect further price declines for branded TEGRETOL XR to approach $8-$9 per tablet over the next 2-3 years, driven by increased generic penetration.
- Price stabilization might occur if the drug secures new patent protection or if formulations with improved delivery or reduced side effects are introduced.
- In markets outside the U.S., prices are lower generally due to regulatory pricing controls, with European prices around €6-€8 per tablet.
Key Factors Affecting Market and Price Trends
- Regulatory Changes: Potential patent extensions or new formulations.
- Market Competition: Entry of biosimilars or replacement therapies.
- Insurance Coverage: Formulary decisions favoring generics.
- Manufacturing Costs: Marginally influence pricing, especially for branded product.
Investment and Business Implications
Manufacturers should monitor patent status changes and regulatory approvals for new formulations to anticipate potential price hikes. Payers and healthcare providers will continue favoring generics, pressuring the branded market share.
References:
[1] MarketsandMarkets, "Epilepsy Therapeutics Market," 2022.
[2] Grand View Research, "Trigeminal Neuralgia Therapeutics," 2022.
[3] IQVIA, "Global Bipolar Disorder Market Insights," 2022.
Key Takeaways
- TEGRETOL XR is increasingly overshadowed by generic carbamazepine, leading to significant price erosion.
- The drug’s branded price in the U.S. is around $11 per tablet, likely to decline further.
- Market demand remains steady, driven by epilepsy and bipolar treatment needs, but profit margins are shrinking.
- Patent expiration and market pressures diminish the likelihood of substantial future price increases.
- Future value relies on new formulations or patents securing exclusive rights.
FAQs
1. How does the price of TEGRETOL XR compare to generics?
Branded TEGRETOL XR costs approximately $11 per 200 mg tablet, while generics cost about $0.50-$1.00 per tablet.
2. What factors could lead to a price increase for TEGRETOL XR?
Patent extension, approval of new formulations with improved features, or exclusive rights could raise prices temporarily.
3. Is TEGRETOL XR still profitable for manufacturers?
Profitability decreases due to high generic penetration and declining brand prices but remains viable if marketed for niche uses or with new formulations.
4. How much market share does TEGRETOL XR currently hold?
Estimated at 15-20% of the branded carbamazepine market, mostly among patients requiring extended-release formulations.
5. What is the outlook for TEGRETOL XR over the next five years?
Prices are expected to decline, with continued marketing challenges from generics. Growth depends on formulation innovation or market exclusivity gains.
[1] MarketsandMarkets, "Epilepsy Therapeutics Market," 2022.
[2] Grand View Research, "Trigeminal Neuralgia Therapeutics," 2022.
[3] IQVIA, "Global Bipolar Disorder Market Insights," 2022.
More… ↓
