Share This Page
Drug Price Trends for TAZAROTENE
✉ Email this page to a colleague
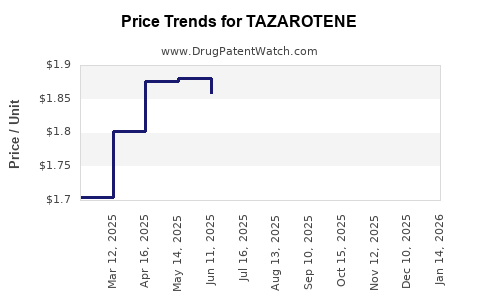
Average Pharmacy Cost for TAZAROTENE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TAZAROTENE 0.05% CREAM | 00713-0765-31 | 7.21737 | GM | 2026-03-18 |
| TAZAROTENE 0.05% CREAM | 45802-0706-94 | 7.21737 | GM | 2026-03-18 |
| TAZAROTENE 0.05% CREAM | 00713-0765-60 | 10.60116 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TAZAROTENE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TAZAROTENE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-1373-03 | 60GM | 76.25 | 1.27083 | GM | 2023-06-15 - 2028-06-14 | FSS |
| TAZAROTENE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-1373-03 | 60GM | 86.06 | 1.43433 | GM | 2023-06-23 - 2028-06-14 | FSS |
| TAZAROTENE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-1373-02 | 30GM | 42.96 | 1.43200 | GM | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tazarotene: Patent Landscape and Market Outlook
Tazarotene, a third-generation topical retinoid, faces a dynamic market shaped by patent expirations and the emergence of generic competition. Its established efficacy in treating psoriasis and acne continues to drive demand, but pricing strategies and market penetration will be critical for stakeholders in the post-patent era.
What is Tazarotene's Current Patent Status?
The core patents covering tazarotene have largely expired in major markets, opening the door for generic manufacturers.
- Original U.S. Patent: The foundational patent for tazarotene, U.S. Patent No. 4,906,634, expired in 2009. This patent covered the compound itself.
- Formulation Patents: Subsequent patents addressed specific formulations, such as gels and creams, and their applications. For example, patents related to specific excipients or delivery systems may have had later expiration dates.
- Authorized Generics: Pharmaceutical companies often release authorized generics after their primary patents expire. This strategy allows them to compete directly with independent generic manufacturers and maintain a market presence.
- Exclusivity Periods: While compound patents have expired, specific regulatory exclusivities, such as those granted for new uses or pediatric studies, may have provided extended market protection for the branded product. These have largely lapsed for the primary indications.
How Has Generic Entry Impacted Tazarotene Pricing?
The introduction of generic tazarotene has led to significant price reductions.
- Price Erosion: Following patent expiry and the subsequent influx of generic alternatives, the average selling price (ASP) of tazarotene has declined substantially. This is a standard market dynamic for drugs transitioning from branded to generic status.
- Competitive Pricing: Generic manufacturers compete on price to gain market share. This competition directly benefits payers and patients through lower out-of-pocket costs.
- Manufacturer Price Adjustments: Branded manufacturers may adjust their pricing strategies for the branded product to remain competitive, often by introducing authorized generics or focusing on value-added services or formulations.
- Market Share Shift: Generics have captured a dominant share of the tazarotene market, with pricing being a primary driver for prescription volume.
What are the Key Indications for Tazarotene?
Tazarotene is prescribed for specific dermatological conditions.
- Psoriasis: It is approved for the topical treatment of plaque psoriasis of the skin, including on the face. Tazarotene works by normalizing keratinocyte differentiation and reducing inflammation.
- Acne Vulgaris: Tazarotene is also indicated for the topical treatment of facial acne vulgaris in patients 12 years of age and older. It helps to reduce inflammatory and non-inflammatory lesions.
- Photoaged Skin: While not its primary approved indication in all regions, tazarotene has been studied and used off-label for improving the appearance of fine wrinkles, mottled hyperpigmentation, and other signs of photoaging.
What are the Projected Market Dynamics for Tazarotene?
The tazarotene market will continue to be characterized by generic competition and evolving treatment landscapes.
- Dominance of Generics: The market will remain dominated by generic tazarotene products. The cost-effectiveness of generics will drive prescription volume.
- Stable Demand for Core Indications: Demand for tazarotene in psoriasis and acne is expected to remain stable due to its established efficacy and the chronic nature of these conditions.
- Competition from Newer Therapies: Newer topical and systemic therapies for psoriasis and acne may offer alternative treatment options, potentially impacting tazarotene's market share growth, especially in severe or recalcitrant cases. However, tazarotene's affordability will likely preserve its position for less severe presentations and as a maintenance therapy.
- Formulation Innovation: Manufacturers may explore novel formulations or combination products to differentiate their offerings, potentially commanding a premium price. However, broad patent protection for such innovations would be challenging given the mature nature of the compound.
- Geographic Variations: Market dynamics will vary by region, influenced by local regulatory environments, healthcare reimbursement policies, and the availability of generic alternatives.
What is the Estimated Market Size and Price Projection for Tazarotene?
Estimating precise market size for a drug with widespread generic availability is challenging as data is fragmented across multiple manufacturers. However, trends indicate continued volume but price pressure.
- Market Size: The global market for topical retinoids, including tazarotene, is substantial. While specific tazarotene market figures are not readily available due to generic fragmentation, the broader topical retinoid market is valued in the billions of dollars. Tazarotene's share within this is significant, particularly in the treatment of moderate to severe psoriasis and acne.
- Price Projections:
- Branded Product: The branded tazarotene product (e.g., Tazorac) will likely continue to command a premium, but its market share will be limited. Pricing will be influenced by competitive pressures from authorized generics and independent generics. Prices may range from $100 to $250+ per tube, depending on formulation and quantity, though this will be subject to significant payer negotiations and patient assistance programs.
- Generic Tazarotene: Generic tazarotene prices are expected to remain low and continue to face downward pressure. The average wholesale price (AWP) for a generic 30g tube of 0.1% cream or gel can range from $20 to $60. This price is highly variable and depends on the specific generic manufacturer, pharmacy, and insurance coverage.
- Price Stability (Generics): While overall price levels are low, incremental price fluctuations for generics are common, driven by manufacturing costs, competitive intensity, and supply chain dynamics. Significant price increases for generics are unlikely in the absence of new patent protection or a major supply disruption.
- Future Trends: Pricing will be further influenced by health technology assessments and formulary tier placements by major insurance providers. The focus will remain on cost-effectiveness for payers.
What are the Key Competitors and Alternatives to Tazarotene?
Tazarotene competes within a crowded dermatological market.
- Other Topical Retinoids:
- Tretinoin (Retin-A, Atralin, etc.): The most established topical retinoid, available in various formulations and strengths. It faces similar generic competition.
- Adapalene (Differin): Available over-the-counter (OTC) in lower strengths (0.1%) and by prescription in higher strengths (0.3%). It has a generally better tolerability profile than tazarotene.
- Trifarotene (Aklief): A newer topical retinoid with selective RAR-γ activity, approved for acne. It represents a newer generation of retinoids and has a different patent and market exclusivity profile.
- Topical Corticosteroids: Commonly used in combination or as adjunctive therapy for psoriasis, offering anti-inflammatory benefits but without the retinoid's keratolytics properties. Examples include clobetasol, betamethasone, and triamcinolone.
- Vitamin D Analogues: Calcipotriene and calcitriol are frequently used in combination with corticosteroids for psoriasis.
- Systemic Therapies: For moderate to severe psoriasis, biologic agents (e.g., adalimumab, ustekinumab, secukinumab) and oral small molecules (e.g., apremilast, deucravacitinib) represent significant competition, offering higher efficacy but at considerably higher costs and with different risk profiles.
- Antibiotics and Benzoyl Peroxide: Common first-line treatments for acne, often used in combination with retinoids.
What are the Regulatory Considerations for Tazarotene Products?
Regulatory bodies govern the approval, manufacturing, and marketing of tazarotene.
- FDA (U.S. Food and Drug Administration): Approves new drug applications (NDAs) and abbreviated new drug applications (ANDAs) for tazarotene. Generic tazarotene products require FDA approval demonstrating bioequivalence to the reference listed drug.
- EMA (European Medicines Agency): Oversees drug approvals in the European Union, following a similar process for generic applications.
- Manufacturing Standards: All tazarotene products, branded and generic, must adhere to Current Good Manufacturing Practices (cGMP) to ensure quality, safety, and efficacy.
- Labeling Requirements: Prescribing information must accurately reflect approved indications, contraindications, warnings, precautions, and adverse reactions. This includes specific warnings regarding photosensitivity and potential teratogenicity.
- Post-Marketing Surveillance: Regulatory agencies monitor the safety of marketed tazarotene products and can require label changes or further studies based on adverse event reports.
What are the Intellectual Property Trends Affecting Tazarotene?
While core patents have expired, innovation and patenting continue in related areas.
- New Formulations: Companies continue to file patents on novel formulations designed to improve tolerability, efficacy, or patient compliance. This might include sustained-release gels, microemulsions, or combination products.
- Combination Therapies: Patents may cover specific fixed-dose combinations of tazarotene with other active pharmaceutical ingredients (APIs) for synergistic effects in treating dermatological conditions.
- Manufacturing Processes: Patents can also protect novel or improved methods of synthesizing tazarotene or manufacturing its formulations, aiming for cost reduction or higher purity.
- Method of Use Patents: While less common for established indications, patents for new uses of tazarotene discovered through research could emerge, though these would require significant clinical validation and regulatory approval.
- Litigation: Disputes over existing patents or alleged patent infringement remain a possibility, particularly concerning new formulations or manufacturing processes.
Key Takeaways
Tazarotene's market is now firmly in the generic era, characterized by intense price competition and broad accessibility for its primary indications of psoriasis and acne. While the branded product faces limited market share, the compound itself remains a cornerstone therapy. Future market dynamics will be shaped by the cost-effectiveness of generics, the emergence of newer dermatological agents, and potential, albeit limited, innovation in formulation or combination products.
Frequently Asked Questions
-
Are there any remaining patents that protect the original branded tazarotene product from generic competition? No, the primary compound patents for tazarotene have expired in major markets like the U.S. and Europe. While some formulation or method-of-use patents may have had later expiration dates, they do not prevent generic manufacturers from producing and selling the active ingredient and its established formulations.
-
How does tazarotene's efficacy compare to other topical retinoids like tretinoin or adapalene? Tazarotene is generally considered more potent than tretinoin and adapalene, leading to potentially faster or more significant results for some patients, particularly in psoriasis. However, this increased potency is often associated with a higher incidence of skin irritation, redness, and peeling, requiring careful patient counseling and dose titration.
-
What is the typical cost difference between branded and generic tazarotene? Generic tazarotene is significantly less expensive than the branded version. While the branded product could cost upwards of $150-$250 for a tube, generic versions are typically available for $20-$60, depending on the pharmacy, insurance, and specific product. This price difference is a primary driver for generic adoption.
-
Can tazarotene be used for conditions other than psoriasis and acne? Tazarotene is primarily approved and prescribed for plaque psoriasis and facial acne vulgaris. It has been studied and used off-label for photoaging, but this is not an approved indication in all regions and carries specific risks. Physicians consider off-label use based on individual patient needs and clinical judgment.
-
What are the main side effects associated with tazarotene use? The most common side effects of topical tazarotene include local skin reactions such as redness, peeling, dryness, itching, and burning. Increased sensitivity to sunlight (photosensitivity) is also a significant concern, necessitating strict sun protection. More severe, though less common, side effects can occur.
More… ↓
