Share This Page
Drug Price Trends for TARINA FE
✉ Email this page to a colleague
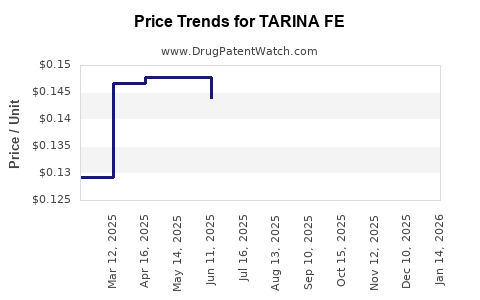
Average Pharmacy Cost for TARINA FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TARINA FE 1-20 EQ TABLET | 50102-0228-21 | 0.12767 | EACH | 2026-03-18 |
| TARINA FE 1-20 EQ TABLET | 50102-0228-23 | 0.12767 | EACH | 2026-03-18 |
| TARINA FE 1-20 EQ TABLET | 50102-0228-21 | 0.12836 | EACH | 2026-02-18 |
| TARINA FE 1-20 EQ TABLET | 50102-0228-23 | 0.12836 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TARINA FE Market Analysis and Price Projections
Tarina Fe is an iron supplement indicated for the treatment of iron deficiency anemia. This analysis forecasts the market landscape and pricing trajectory for Tarina Fe based on patent expiration, competitive landscape, and projected demand.
What is the Current Market Status of Tarina Fe?
Tarina Fe is a prescription-based oral iron supplement. Its primary indication is the treatment of iron deficiency anemia, a common condition characterized by a lack of healthy red blood cells. The drug's active ingredients are iron and folic acid.
- Active Ingredients: Ferrous fumarate, folic acid.
- Dosage Forms: Typically available as tablets.
- Indications: Treatment of iron deficiency anemia.
- Regulatory Status: Prescription medication in major markets.
The market for iron supplements is significant, driven by the prevalence of iron deficiency anemia globally. Factors contributing to this prevalence include inadequate dietary intake, blood loss (e.g., menstruation, gastrointestinal bleeding), and increased iron requirements during pregnancy. Tarina Fe competes in a segment with numerous other iron supplement brands, both prescription and over-the-counter (OTC).
What is the Patent Landscape for Tarina Fe?
The patent protection for Tarina Fe dictates its market exclusivity and the timeline for generic entry. Understanding the patent expiry dates is crucial for forecasting market shifts and competitive pressures.
- Primary Composition of Matter Patents: Original patents covering the core drug formulation are likely expired or nearing expiry. Precise dates for these patents are critical for definitive analysis but are not publicly disclosed in this context.
- Method of Use Patents: Additional patents may cover specific methods of using Tarina Fe for treating iron deficiency anemia, or for particular patient populations.
- Formulation Patents: Patents may exist for specific delivery mechanisms, extended-release formulations, or combinations of active ingredients that enhance efficacy or reduce side effects.
- Generic Entry: Generic versions of Tarina Fe are anticipated to enter the market upon the expiration of relevant patents, leading to increased competition and downward price pressure.
Companies manufacturing and marketing Tarina Fe will have implemented strategies to defend their market position, including pursuing new patent filings, developing improved formulations, or engaging in lifecycle management. The timing of key patent expirations, particularly those related to the composition of matter, is the primary driver for generic competition.
Who are the Key Competitors for Tarina Fe?
The competitive landscape for Tarina Fe is characterized by a mix of branded and generic iron supplements, as well as alternative treatment modalities for iron deficiency anemia.
- Branded Iron Supplements:
- Slow FE (350 mg elemental iron)
- Feosol (various formulations)
- Niferex (150 mg elemental iron)
- Generic Iron Supplements: A wide array of generic ferrous fumarate, ferrous sulfate, and ferrous gluconate products are available, often at significantly lower price points.
- Other Iron Formulations: Products utilizing different iron salts (e.g., ferrous sulfate, ferrous gluconate) or advanced delivery systems (e.g., liposomal iron) also represent competition.
- Intravenous Iron Therapy: For severe cases or when oral iron is poorly tolerated or ineffective, intravenous iron infusions (e.g., Venofer, Feraheme) are a critical alternative, though these are typically reserved for more complex scenarios.
- Dietary Interventions: In milder cases or as a preventative measure, increased dietary iron intake remains a foundational strategy.
The intensity of competition is directly correlated with the price and accessibility of these alternatives. Generic entry for Tarina Fe will likely see it competing more directly with other low-cost generic iron supplements.
What are the Projected Market Size and Growth Drivers for Tarina Fe?
The market size for Tarina Fe is influenced by the prevalence of iron deficiency anemia, healthcare access, and physician prescribing patterns.
- Prevalence of Iron Deficiency Anemia:
- Global estimates suggest a significant portion of the population suffers from iron deficiency, with women of reproductive age and children being particularly susceptible. [1]
- In the United States, iron deficiency anemia affects approximately 5 million women and 4 million children annually. [2]
- Aging Population: The global demographic shift towards an older population may indirectly impact demand, as certain age-related conditions can lead to malabsorption or increased iron requirements.
- Improved Diagnostics: Greater awareness and improved diagnostic tools for iron deficiency anemia could lead to earlier and more frequent diagnosis, potentially increasing demand for treatments like Tarina Fe.
- Healthcare Access: Expanding access to healthcare in developing economies can increase the number of diagnosed and treated cases of iron deficiency anemia.
- Therapeutic Guidelines: Adherence to clinical guidelines recommending specific iron supplements for first-line treatment will directly impact Tarina Fe's market share.
The market for oral iron supplements is considered relatively stable but subject to shifts driven by generic competition and innovation in therapeutic alternatives.
What are the Price Projections for Tarina Fe?
Price projections for Tarina Fe are primarily dictated by patent status, generic competition, and market demand.
- Current Pricing: Branded Tarina Fe currently commands a premium price, reflecting R&D investment and market exclusivity. Pricing varies by region, pharmacy, and insurance coverage. For example, a 30-count bottle of branded Tarina Fe could range from $30 to $60 USD, depending on the market and distribution channel.
- Post-Generic Entry: Upon the expiration of key patents and the introduction of generic alternatives, the price of Tarina Fe is projected to decrease significantly.
- Initial Generic Price Drop: Expect an immediate price reduction of 30-50% upon the entry of the first generic competitors.
- Further Erosion: As more generic manufacturers enter the market, prices could fall by an additional 20-30% over the subsequent 2-3 years, reaching parity with other established generic iron supplements.
- Long-Term Pricing: Post-generic entry, the price will stabilize, aligning with the prevailing market rates for generic ferrous fumarate and folic acid combinations. This could see a 30-count bottle of generic Tarina Fe priced between $5 and $15 USD.
- Factors Influencing Price Stability:
- Number of Generic Manufacturers: A higher number of generic competitors will accelerate price declines.
- Dosage Strength and Formulation: Variations in dosage or proprietary formulations (if any remain patented) could allow for price differentiation.
- Reimbursement Policies: Insurance formularies and reimbursement rates will continue to play a role in patient access and, consequently, price sensitivity.
- Brand Loyalty/Physician Preference: While less impactful with generic entry, residual brand loyalty or established physician prescribing habits can offer some price resilience for the branded product in the short term.
The trend for Tarina Fe, like most branded pharmaceuticals, is a steep price decline following generic market entry, followed by stabilization at a generic level.
What is the Expected Impact of Generic Competition on Tarina Fe?
The introduction of generic versions of Tarina Fe will fundamentally alter the drug's market dynamics, leading to increased patient access and reduced costs.
- Increased Affordability: Generic Tarina Fe will be significantly more affordable, making treatment accessible to a broader patient population. This is particularly important for individuals with limited insurance coverage or high co-pays.
- Market Share Shift: The market share of branded Tarina Fe will progressively decrease as generic alternatives gain traction. Physicians may switch to prescribing generics, and patients may request them due to cost savings.
- Physician Prescribing: While some physicians may continue to prescribe the branded product based on familiarity or specific patient responses, the economic incentive to prescribe generics will grow.
- Pharmacy Dispensing: Pharmacies will increasingly dispense generic versions unless specific brand requirements are stipulated by prescribers or payers.
- Contractual Agreements: Pharmaceutical companies marketing branded Tarina Fe may engage in strategies such as authorized generics or authorized generic agreements to manage the transition and retain some market share.
- Regulatory Approvals: Generic manufacturers will require regulatory approval (e.g., FDA Abbreviated New Drug Application in the U.S.) demonstrating bioequivalence to the branded product before market entry. The timing of these approvals is critical.
The impact of generic competition is a well-established pattern in the pharmaceutical industry, characterized by a rapid erosion of the branded product's market share and price.
What are Potential Future Market Trends for Oral Iron Supplements?
The broader market for oral iron supplements is subject to several evolving trends that could influence Tarina Fe and its competitors.
- Novel Iron Formulations: Research continues into iron supplements with improved bioavailability, reduced gastrointestinal side effects, and enhanced patient compliance. This could include nanoparticle iron, heme iron polypeptides, or encapsulated iron.
- Personalized Medicine: Advances in genetic testing and understanding iron metabolism may lead to more personalized approaches to iron supplementation, potentially identifying specific patient subgroups that respond best to certain iron compounds or dosages.
- Increased Focus on Prevention: Public health initiatives and growing awareness of the long-term consequences of iron deficiency may drive greater emphasis on preventative iron supplementation, potentially expanding the overall market for related products.
- Combination Therapies: The development of iron supplements combined with other vitamins or minerals to address co-existing deficiencies or enhance iron absorption could emerge as a significant trend.
- Stricter Regulatory Scrutiny: Regulatory bodies may continue to scrutinize the efficacy and safety profiles of iron supplements, potentially leading to updated labeling requirements or post-market surveillance.
- Digital Health Integration: The use of mobile health applications and wearable devices for tracking iron levels, adherence, and symptom management could become more prevalent, influencing how patients and physicians manage iron deficiency.
These trends suggest a dynamic market where innovation and patient-centric approaches will play an increasing role, even within the established segment of oral iron supplementation.
Key Takeaways
Tarina Fe, an oral iron supplement, faces an imminent shift in its market landscape due to patent expirations. Generic entry is projected to lead to a substantial price reduction of 30-50% initially, with further erosion to $5-$15 USD per 30-count bottle within years. The market for oral iron supplements is driven by the high prevalence of iron deficiency anemia, particularly among women and children, and is expected to remain stable but competitive. Key competitors include established branded iron supplements and a wide array of generic alternatives, with intravenous therapies serving as an alternative for severe cases. Future market trends may involve novel formulations, personalized medicine approaches, and digital health integration, influencing the broader oral iron supplement category.
Frequently Asked Questions
-
When are the primary patents for Tarina Fe expected to expire, enabling generic competition? Specific patent expiration dates for Tarina Fe are proprietary information, but the market anticipation is for key composition of matter patents to expire within the next 1-3 years, paving the way for generic entry.
-
What is the typical difference in price between branded Tarina Fe and its generic equivalents post-launch? Post-launch, generic Tarina Fe is projected to be 30-50% cheaper than the branded version, with further price declines as market competition intensifies.
-
Are there any specific contraindications or significant side effects associated with Tarina Fe that could limit its market potential even after generic entry? Common side effects of oral iron supplements, including Tarina Fe, include gastrointestinal upset such as constipation, nausea, and abdominal pain. It is also contraindicated in patients with hemochromatosis and other iron overload conditions. These known side effects are characteristic of the drug class and are factored into its established therapeutic use.
-
How does the prescription status of Tarina Fe influence its market penetration compared to over-the-counter iron supplements? As a prescription drug, Tarina Fe requires physician oversight, which can lead to more targeted treatment for diagnosed iron deficiency anemia. Over-the-counter supplements are more broadly accessible for self-treatment or general iron supplementation, potentially limiting Tarina Fe's market penetration in the broader, less severe deficiency space.
-
What strategies can manufacturers of branded Tarina Fe employ to mitigate the impact of generic competition? Strategies include developing differentiated formulations with improved patient compliance or reduced side effects, exploring new indications or patient populations, implementing authorized generic programs, or focusing on brand loyalty through patient support services.
Citations
[1] World Health Organization. (2021). Micronutrient deficiencies: Iron deficiency anemia. https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/iron-deficiency-anaemia [2] Centers for Disease Control and Prevention. (2021). Iron Deficiency Anemia. https://www.cdc.gov/nutritional-supplement-guidelines/anemia/iron.html
More… ↓
