Share This Page
Drug Price Trends for TAMIFLU
✉ Email this page to a colleague
Average Pharmacy Cost for TAMIFLU
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
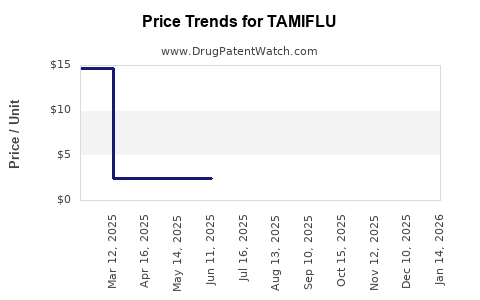
| TAMIFLU 75 MG CAPSULE | 00004-0800-85 | 14.51856 | EACH | 2026-03-18 |
| TAMIFLU 6 MG/ML SUSPENSION | 00004-0822-05 | 2.43293 | ML | 2026-02-18 |
| TAMIFLU 75 MG CAPSULE | 00004-0800-85 | 14.57458 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TAMIFLU
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TAMIFLU CAPSULES 45MG | Genentech USA, Inc. | 00004-0801-85 | 10 | 137.24 | 13.72400 | EACH | 2023-09-15 - 2028-09-14 | FSS |
| TAMIFLU 6MG/ML SUSPENSION,ORAL | Genentech USA, Inc. | 00004-0822-05 | 1X60ML | 112.68 | 2023-09-15 - 2028-09-14 | Big4 | ||
| TAMIFLU CAPSULES 45MG | Genentech USA, Inc. | 00004-0801-85 | 10 | 103.48 | 10.34800 | EACH | 2024-01-01 - 2028-09-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tamiflu Market Analysis and Price Projections
Tamiflu (oseltamivir phosphate), developed by Roche, is an antiviral medication primarily used for the treatment and prevention of influenza A and B. Its market dynamics are influenced by global influenza activity, government stockpiling, patent expirations, and the emergence of generic alternatives.
What is the Current Market Size and Key Drivers for Tamiflu?
The global market for influenza antivirals, including Tamiflu, is substantial, driven by seasonal flu outbreaks and pandemic preparedness initiatives. Annual influenza seasons, varying in severity, consistently create demand. Governments maintain strategic stockpiles of Tamiflu for pandemic response, representing a significant market segment. The availability of generics has broadened access but also introduced price competition.
Key market drivers include:
- Seasonal Influenza Incidence: The number of influenza cases globally each year directly impacts demand for treatment. Organizations like the World Health Organization (WHO) and national public health agencies (e.g., CDC in the U.S.) track and forecast flu activity, influencing procurement cycles.
- Pandemic Preparedness: Governments worldwide maintain stockpiles of antivirals like Tamiflu to mitigate the impact of influenza pandemics. This creates a consistent, albeit sometimes lumpy, demand. The H1N1 pandemic in 2009 significantly boosted stockpiling efforts.
- Patent Expiration and Generic Competition: Tamiflu's primary patents have expired in major markets, allowing for the production and sale of generic oseltamivir phosphate. This has increased market competition and typically leads to lower prices for the drug. For instance, generic versions became available in the U.S. around 2016 and in Europe shortly after.
- Reimbursement Policies: Insurance coverage and national healthcare system formularies significantly affect patient access and physician prescribing habits. Tamiflu is generally covered by most public and private health insurance plans, though co-pays and deductibles vary.
- Physician and Patient Awareness: Education campaigns regarding influenza symptoms, treatment benefits, and the importance of early intervention contribute to consistent demand.
What is the Competitive Landscape for Tamiflu?
The competitive landscape for Tamiflu has evolved significantly since its introduction. Initially, as a novel neuraminidase inhibitor, it held a dominant position. However, patent expirations have opened the door for generic manufacturers.
Key Competitors and Product Landscape:
- Brand Name: Tamiflu (oseltamivir phosphate) by Roche.
- Generic Oseltamivir Phosphate: Multiple pharmaceutical companies now produce generic oseltamivir phosphate. These include, but are not limited to:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Dr. Reddy's Laboratories
- Aurobindo Pharma
- Cipla Limited
- Other Antivirals: While Tamiflu is a primary treatment, other influenza antivirals exist, though often with different mechanisms or approved indications. These include:
- Relenza (zanamivir): Inhaled neuraminidase inhibitor by GlaxoSmithKline.
- Xofluza (baloxavir marboxil): A cap-and-DAG polymerase inhibitor by Shionogi and Roche. Xofluza represents a newer class of influenza treatment and offers a single-dose regimen, presenting a direct competitive threat in some treatment scenarios.
- Amantadine and Rimantadine: Older adamantane antivirals, largely ineffective against current circulating influenza strains due to resistance.
The market share of Tamiflu has declined in favor of generics due to price advantages. However, Roche continues to market Tamiflu, often targeting government stockpiles and areas where brand recognition or specific supply agreements are advantageous. Generic manufacturers primarily compete on price, making them attractive for individual prescriptions and bulk procurement.
What are the Historical and Current Price Trends for Tamiflu?
Tamiflu's pricing has seen a substantial decrease following patent expiration and the subsequent entry of generic oseltamivir phosphate.
Historical Pricing (Pre-Generic Entry):
- Prior to the expiration of key patents, Tamiflu was a high-margin product for Roche. Pricing was set at premium levels, reflecting R&D investment and market exclusivity.
- In the U.S. market, before widespread generic availability (circa 2015-2016), a 10-day course of Tamiflu could cost upwards of $80-$100 USD, depending on the formulation and pharmacy.
Current Pricing (Post-Generic Entry):
- Brand Name Tamiflu: Roche's branded Tamiflu generally remains at a higher price point than generics, though often at a reduced level from its peak exclusivity period. Pricing can fluctuate based on negotiated contracts with governments and large healthcare providers. For example, Roche's pricing for government stockpiles might be different from retail pricing.
- Generic Oseltamivir Phosphate: The entry of multiple generic manufacturers has led to significant price erosion.
- Retail Pricing (U.S.): A 10-day course of generic oseltamivir phosphate can now range from $20 to $50 USD, depending on the pharmacy, insurance coverage, and specific manufacturer. Online pharmacies and discount programs can offer further reductions.
- Wholesale/Government Procurement: Large-scale purchases by governments for pandemic stockpiles or by healthcare systems for institutional use command significantly lower per-unit costs. These prices are often proprietary but are estimated to be considerably lower than retail, potentially in the range of $5-$15 USD per treatment course for bulk orders.
Factors Influencing Current Pricing:
- Volume: Higher purchase volumes lead to lower per-unit costs.
- Competition: Intense competition among generic manufacturers drives prices down.
- Supply Chain Efficiency: Manufacturers with more efficient production processes can offer lower prices.
- Regulatory Approvals: The cost of maintaining regulatory approvals in various countries also influences pricing.
What are the Projected Future Price Trends for Tamiflu and its Generics?
Future price trends for Tamiflu and its generic oseltamivir phosphate are expected to be characterized by continued competition and potential stabilization, influenced by evolving market dynamics.
Projected Trends:
- Continued Price Pressure on Generics: The generic oseltamivir phosphate market is likely to remain highly competitive. Expect prices for generic formulations to remain stable or experience marginal declines, driven by ongoing manufacturing efficiencies and market share battles among producers. Price increases are improbable unless there are significant disruptions to the global supply chain or raw material costs.
- Niche Market for Branded Tamiflu: Roche's branded Tamiflu will likely continue to occupy a niche, potentially focusing on government contracts and specific markets where brand loyalty or established relationships exist. Its price will remain higher than generics, but likely subject to negotiated discounts for large-volume procurement.
- Impact of Newer Antivirals: The market penetration of newer antivirals like Xofluza (baloxavir marboxil) could indirectly affect the pricing strategy of oseltamivir products. If newer, more convenient, or perceived as more effective treatments gain significant market share, there might be increased pressure on oseltamivir prices to remain competitive for its established use cases.
- Global Influenza Season Severity: While not a direct price driver in the short term for existing supply, exceptionally severe influenza seasons could temporarily increase demand and potentially lead to short-term price spikes in certain distribution channels if supply chains are stressed. However, the established manufacturing capacity for generics should mitigate significant long-term price inflation.
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and manufacturing inputs could introduce minor price variability. However, with multiple global manufacturers, price increases due to input costs are likely to be absorbed or mitigated by competitive pressures.
- Government Stockpiling Policies: Changes in government policies regarding pandemic preparedness and the size of strategic reserves could influence overall demand. Increased stockpiling might lead to higher aggregate sales volumes for manufacturers, potentially reinforcing price stability. Conversely, reductions in reserve mandates could dampen demand.
Quantitative Projections:
- Generic Oseltamivir Phosphate: Expect average per-treatment course prices to range from $18-$45 USD in retail settings over the next 3-5 years. Wholesale and government contract pricing is projected to remain in the $5-$12 USD range per treatment course.
- Branded Tamiflu: Retail pricing may continue to hover around $60-$90 USD per treatment course, with negotiated contract pricing for bulk orders being significantly lower and proprietary.
The market for oseltamivir phosphate is mature. The primary driver for future price changes will be the sustained competition among generic manufacturers and the overall efficiency of the global pharmaceutical supply chain, rather than significant shifts in demand or patent-related exclusivity.
What are the Regulatory Considerations and Patent Expirations Affecting Tamiflu?
Regulatory approvals and patent exclusivity are fundamental to the commercial lifecycle of pharmaceutical products like Tamiflu. Their evolution significantly shapes market access and pricing.
Key Patent Expirations:
- United States: The primary compound patent for oseltamivir phosphate expired in 2016. This allowed for the introduction of generic versions of Tamiflu in the U.S. market.
- Europe: Similar patent expiries occurred in major European markets around the same period, enabling the launch of generic oseltamivir phosphate across the European Union.
- Other Jurisdictions: Patent expiries have followed a staggered timeline globally, with major markets opening to generics between 2015 and 2020.
Regulatory Approvals:
- Original Approval: Tamiflu was first approved by the U.S. Food and Drug Administration (FDA) in October 1999 for the treatment of influenza A and B in adults. Subsequent approvals expanded its use to children and for prophylaxis.
- Generic Approvals: Generic oseltamivir phosphate products must undergo rigorous review by regulatory agencies like the FDA and the European Medicines Agency (EMA) to demonstrate bioequivalence to the brand-name drug. This includes proving that the generic active ingredient is the same and that the drug is absorbed and acts in the body in the same way as Tamiflu.
- Manufacturing Standards: All manufacturers, both of the branded product and generics, must adhere to Good Manufacturing Practices (GMP) as mandated by regulatory bodies to ensure product quality, safety, and efficacy.
Impact of Regulatory and Patent Landscape:
- Market Entry of Generics: Patent expiration is the direct enabler for generic competition. Once patents expire and regulatory hurdles are cleared, generic manufacturers can legally produce and sell their versions of the drug.
- Price Competition: The influx of multiple generic manufacturers intensifies price competition, driving down the cost of oseltamivir phosphate significantly.
- Supply Chain Diversification: The availability of multiple manufacturers strengthens the supply chain resilience, reducing reliance on a single producer. This is particularly important for pandemic preparedness stockpiles.
- Continued Market Presence of Brand: While generics dominate the market in terms of volume and price, Roche maintains its market presence for branded Tamiflu. This often involves ongoing relationships with governments for stockpiling and specific supply contracts.
- Future Patent Opportunities: While the primary compound patent has expired, pharmaceutical companies may still hold secondary patents related to specific formulations, delivery methods, or manufacturing processes. However, these are less likely to significantly impede generic competition for the core drug.
The regulatory framework ensures that generic products are safe and effective equivalents, while patent law historically granted periods of market exclusivity. The expiration of Tamiflu's core patents has fundamentally reshaped its market, transitioning it from a branded, high-priced product to a widely available, more affordable generic medication.
What are the Key Takeaways for R&D and Investment?
The oseltamivir phosphate market presents a mature landscape with established generics and ongoing demand driven by seasonal influenza and pandemic preparedness.
- Generic Dominance: The market is largely dominated by generic oseltamivir phosphate due to patent expiries and significant price advantages. Investment opportunities lie with manufacturers of generic APIs and finished dosage forms.
- Stable Demand: Predictable demand exists from seasonal flu treatment and ongoing government stockpiling for pandemic response. This provides a consistent revenue base for established generic players.
- Competitive Pricing Environment: Intense competition among generic manufacturers will continue to suppress prices. Profitability for R&D and investment hinges on efficient manufacturing, supply chain management, and scale.
- Limited Innovation Space: The core oseltamivir molecule is off-patent. Significant R&D investment in novel formulations or delivery systems for oseltamivir is unlikely to yield substantial new market exclusivity or premium pricing, given the existing generic alternatives.
- Strategic Partnerships: Opportunities may exist in forming strategic partnerships for supplying government stockpiles or large healthcare institutions, which often involve negotiated long-term contracts at stable pricing.
- Emerging Antivirals as Competition: Investors should monitor the market penetration and pricing strategies of newer antiviral classes like Xofluza, as they represent a growing competitive threat that could indirectly impact demand for oseltamivir.
Frequently Asked Questions
-
Will the price of generic oseltamivir phosphate increase significantly in the next five years? Significant price increases for generic oseltamivir phosphate are unlikely. The market is characterized by robust competition among multiple manufacturers, which generally leads to price stabilization or incremental decreases due to manufacturing efficiencies. Barring unforeseen global supply chain disruptions or substantial increases in raw material costs, prices are expected to remain within current ranges.
-
What is the projected demand for Tamiflu in government stockpiles? Demand for Tamiflu (and its generic equivalents) in government stockpiles is expected to remain a consistent factor. Governments continue to prioritize pandemic preparedness, maintaining reserves of effective antiviral medications. While the precise volume of these stockpiles is proprietary and can fluctuate based on geopolitical and public health assessments, this segment represents a stable, albeit non-retail, market.
-
Are there any new patents being filed for oseltamivir phosphate that could impact its market exclusivity? The primary compound patents for oseltamivir phosphate have expired. While secondary patents related to specific manufacturing processes or novel formulations might exist or be filed, they are unlikely to grant broad market exclusivity for the core drug in the way the original compound patent did. Such patents typically offer more limited protection and are harder to enforce against established generic manufacturing methods.
-
How does the efficacy of Tamiflu compare to newer influenza antivirals like Xofluza? Tamiflu (oseltamivir phosphate) works by inhibiting neuraminidase enzymes, preventing the spread of the virus within the respiratory tract. Xofluza (baloxavir marboxil) is a first-in-class cap-and-DAG polymerase inhibitor. While both are effective against influenza, they have different mechanisms of action and clinical profiles. Studies have shown varying comparative efficacies and side effect profiles. Xofluza offers the convenience of a single-dose regimen, which is a key differentiator. Physician preference and evolving treatment guidelines will continue to shape their comparative use.
-
What are the primary manufacturing challenges for generic oseltamivir phosphate producers? Primary manufacturing challenges for generic oseltamivir phosphate producers include maintaining high-quality API production that meets stringent regulatory standards (e.g., FDA, EMA), optimizing synthesis yields to reduce costs, managing complex global supply chains for raw materials, and ensuring compliance with Good Manufacturing Practices (GMP) across all production facilities. Competition on price also necessitates continuous efforts to improve process efficiency and reduce operational expenditures.
Citations
[1] U.S. Food & Drug Administration. (1999). Tamiflu Approval Letter. Retrieved from FDA.gov. [2] European Medicines Agency. (n.d.). Oseltamivir Phosphate Information. Retrieved from EMA.europa.eu. [3] Centers for Disease Control and Prevention. (n.d.). Antiviral Drugs for Influenza. Retrieved from CDC.gov. [4] Roche Holdings AG. (Annual Reports). Financial Statements and Investor Relations. Retrieved from Roche.com. [5] Pharmaceutical industry market analysis reports (confidential, proprietary data sources not publicly disclosed).
More… ↓
