Drug Price Trends for SYNJARDY XR
✉ Email this page to a colleague
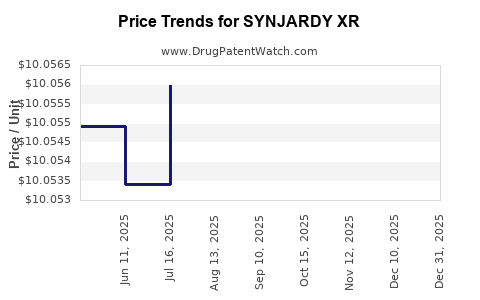
Average Pharmacy Cost for SYNJARDY XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SYNJARDY XR 10-1,000 MG TABLET | 00597-0280-73 | 11.21167 | EACH | 2026-05-20 |
| SYNJARDY XR 12.5-1,000 MG TAB | 00597-0300-45 | 5.60034 | EACH | 2026-05-20 |
| SYNJARDY XR 5-1,000 MG TABLET | 00597-0290-59 | 5.60587 | EACH | 2026-05-20 |
| SYNJARDY XR 5-1,000 MG TABLET | 00597-0290-74 | 5.60587 | EACH | 2026-05-20 |
| SYNJARDY XR 10-1,000 MG TABLET | 00597-0280-90 | 11.21167 | EACH | 2026-05-20 |
| SYNJARDY XR 25-1,000 MG TABLET | 00597-0295-78 | 11.18825 | EACH | 2026-05-20 |
| SYNJARDY XR 12.5-1,000 MG TAB | 00597-0300-93 | 5.60034 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SYNJARDY XR
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SYNJARDY XR 12.5MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0300-93 | 180 | 1303.02 | 7.23900 | EACH | 2023-01-01 - 2027-09-14 | FSS |
| SYNJARDY XR 5MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0290-59 | 180 | 1303.02 | 7.23900 | EACH | 2022-09-15 - 2027-09-14 | FSS |
| SYNJARDY XR 25MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0295-78 | 90 | 1267.05 | 14.07833 | EACH | 2023-01-01 - 2027-09-14 | Big4 |
| SYNJARDY XR 12.5MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0300-45 | 60 | 434.34 | 7.23900 | EACH | 2023-01-01 - 2027-09-14 | FSS |
| SYNJARDY XR 10MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0280-90 | 90 | 1303.02 | 14.47800 | EACH | 2022-09-15 - 2027-09-14 | FSS |
| SYNJARDY XR 5MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0290-74 | 60 | 421.72 | 7.02867 | EACH | 2023-01-01 - 2027-09-14 | Big4 |
| SYNJARDY XR 10MG/1000MG TAB | Boehringer Ingelheim Pharmaceuticals, Inc. | 00597-0280-73 | 30 | 434.34 | 14.47800 | EACH | 2022-09-15 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
