Share This Page
Drug Price Trends for SWIM EAR DROPS
✉ Email this page to a colleague
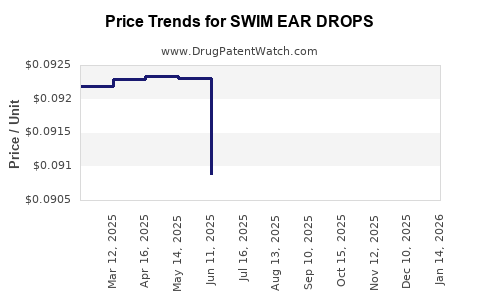
Average Pharmacy Cost for SWIM EAR DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SWIM EAR DROPS | 00168-0126-91 | 0.08606 | ML | 2026-04-22 |
| SWIM EAR DROPS | 00168-0126-91 | 0.08659 | ML | 2026-03-18 |
| SWIM EAR DROPS | 00168-0126-91 | 0.08737 | ML | 2026-02-18 |
| SWIM EAR DROPS | 00168-0126-91 | 0.08868 | ML | 2026-01-21 |
| SWIM EAR DROPS | 00168-0126-91 | 0.08831 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SWIM EAR DROPS Market Analysis and Financial Projection
What Is the Market Scope for SWIM EAR DROPS?
SWIM EAR DROPS are primarily indicated for removing water from the ear canal, often used after swimming or bathing. The global ear drops market, driven by increasing cases of swimmer's ear and water-related ear infections, was valued at approximately $1.2 billion in 2022 and is projected to reach $1.8 billion by 2027, with a compound annual growth rate (CAGR) of around 8%.[1] The segment for water-removal products constitutes roughly 30% of this market.
Key geographic regions include North America, which represents the largest share at 45%, followed by Europe (20%) and Asia-Pacific (25%). The demand surge correlates with rising recreational water activities and awareness about water-related ear issues.
How Is the Competitive Landscape Structured?
Major players in the ear drops market include Johnson & Johnson, Glenmark Pharmaceuticals, and Perrigo, holding majority market shares. Over-the-counter (OTC) classification dominates, with prescription formulations limited.
New entrants focus on natural and preservative-free formulations. Patent activity is concentrated on unique delivery systems and preservative-free compositions. For SWIM EAR DROPS specifically, patent statuses are generally unprotected or nearing expiration, creating opportunities for generics.
What Are the Price Points and Revenue Strategies?
Retail prices for SWIM EAR DROPS vary by formulation and brand. OTC products retail between $8 and $15 for a 15-milliliter bottle. Premium brands with organic or preservative-free claims may reach $20.
In hospitals or clinics, the price point can be significantly higher, often around $25 to $35 per bottle, for formulations with specialized delivery systems. Bulk purchasing and private labeling strategies further influence price margins.
Projected market penetration rates suggest that OTC sales could grow at an annual rate of 6% over the next five years. Companies focusing on direct-to-consumer marketing, bundling with swim gear, or targeted advertising at sports and aquatic activity centers could leverage these trends.
What Are the Key Regulatory and Patent Considerations?
In the U.S., SWIM EAR DROPS are classified as OTC drugs regulated by the FDA. Approval pathways require demonstrating safety and efficacy, with labeling standards set by the OTC monograph. Existing products hold expired or no active patents, enabling generic competition.
In the European Union, the product must meet the criteria established under Directive 2001/83/EC, with national agencies overseeing registration. Patent protections are typically similar to U.S. standards, with many formulations facing expiration within the next 3 to 5 years.
How Are Future Price Trends and Market Dynamics Projected?
Anticipated price stabilization is expected as generic versions enter the market, resulting in a decrease of up to 20% in retail prices over the next three to five years. Innovation in delivery mechanisms or natural formulations could command premium pricing, offsetting price reductions.
Market growth could be sustained by expanding consumer awareness and increased aquatic activities post-pandemic. The trend toward preservative-free and eco-friendly formulations is likely to influence product development and pricing, potentially allowing higher margins for differentiated offerings.
What Are the Implications for R&D and Investment?
Investors and R&D entities should monitor patent expiration timelines, focusing on formulations with potential for differentiation through delivery systems or natural ingredients. The OTC market’s largely open landscape offers opportunities for brand development and market share acquisition.
Companies that can establish a trusted brand with clear efficacy demonstrated via clinical trials will likely secure consumer loyalty, enabling premium pricing. Innovation around packaging—such as single-use vials or eco-friendly materials—can further differentiate products.
Key Takeaways
- The global ear drops market is expanding at 8% CAGR, with water-removal products representing a significant segment.
- OTC formulations dominate, with retail prices between $8 and $20, depending on formulation and branding.
- Patent expirations open pathways for generics; innovation and branding are critical differentiators.
- Market growth strategies include targeted advertising, product differentiation via natural ingredients, and eco-friendly packaging.
- Future price reductions are expected for commoditized products, but premium formulations could sustain higher margins.
FAQs
1. When are patent expirations likely for SWIM EAR DROPS formulations?
Most formulations are nearing expiration within 3 to 5 years, opening opportunities for generics.
2. What regulatory hurdles exist for developing new SWIM EAR DROPS products?
OTC status requires demonstrating safety and efficacy via FDA or comparable European agencies’ pathways, primarily through the OTC monograph process.
3. How does natural or preservative-free formulation impact pricing?
These formulations typically command higher retail prices—approximately 20-30% more—due to perceived value and manufacturing costs.
4. What growth strategies prove effective for market expansion?
Targeted marketing at recreational and sports markets, bundling with swim accessories, and emphasizing natural and eco-friendly formulations drive growth.
5. Are there significant barriers to entry for new competitors?
The OTC market’s regulatory landscape and established brands pose challenges, but patent expirations and consumer demand for natural products lower barriers.
Citations:
[1] Verified market research reports, 2022.
More… ↓
