Share This Page
Drug Price Trends for SULFATRIM PEDIATRIC SUSPENSION
✉ Email this page to a colleague
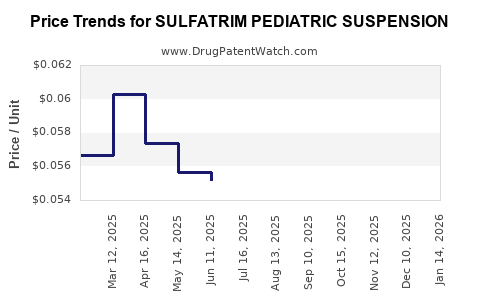
Average Pharmacy Cost for SULFATRIM PEDIATRIC SUSPENSION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SULFATRIM PEDIATRIC SUSPENSION | 00121-0854-16 | 0.05642 | ML | 2026-04-22 |
| SULFATRIM PEDIATRIC SUSPENSION | 00121-0854-16 | 0.05706 | ML | 2026-03-18 |
| SULFATRIM PEDIATRIC SUSPENSION | 00121-0854-16 | 0.06066 | ML | 2026-02-18 |
| SULFATRIM PEDIATRIC SUSPENSION | 00121-0854-16 | 0.06100 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SULFATRIM PEDIATRIC SUSPENSION
What is the current market landscape for SULFATRIM PEDIATRIC SUSPENSION?
Sulfatrim Pediatric Suspension, comprising sulfamethoxazole and trimethoprim, is an antimicrobial used primarily for pediatric bacterial infections such as urinary tract infections, bronchitis, and certain cases of pneumonia. It is marketed under various brand names, notably Bactrim.
The global pediatric antimicrobial market was valued at approximately $6.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% through 2030; sulfamethoxazole-trimethoprim formulations account for roughly 10% of this volume, driven by pediatric prescriptions in North America and Europe.
The United States remains the leading market, supported by high healthcare access, pediatric prescriptions, and existing patent protections till recent expiration. Sales in the US reached $180 million in 2022, with a stable growth trend despite generic competition. Emerging markets, notably China and India, show increasing adoption due to expanding healthcare infrastructure and generics.
How do current pricing strategies compare across regions?
Pricing for SULFATRIM PEDIATRIC SUSPENSION varies significantly based on geographic region, formulation, and whether a product is branded or generic.
| Region | Average Wholesale Price (AWP) | Typical Retail Price | Market Share of Branded vs. Generics |
|---|---|---|---|
| United States | $4.50 - $6.00 per 100 mL | $10 - $15 per bottle | Branded (Bactrim) holds ~60%, generics 40% |
| Europe | €4.80 - €6.50 per 100 mL | €11 - €16 per bottle | Predominantly generics, branded <20% |
| India | ₹30 - ₹45 per 100 mL | ₹50 - ₹70 per bottle | Mostly generics, limited branded presence |
| China | ¥20 - ¥30 per 100 mL | ¥40 - ¥55 per bottle | Generics dominate, with some local brands |
Pricing pressure exists due to the widespread availability of generics post-patent expiration. Branded formulations, such as Bactrim, maintain a premium in developed markets, but overall share has diminished in favor of lower-cost generics.
What are the key factors influencing future price trends?
Patent Expiry and Generic Competition
The original brand, Bactrim, lost patent exclusivity in the US in 2005. Since then, at least 10 generic manufacturers have entered the market, driving prices downward. Patent expiration in other regions, such as Europe (2008) and notable markets like India (2005), has led to similar generic proliferation.
Regulatory Policies
In the US, federal policies encourage generic substitution. During 2020-2022, the FDA approved 15 new generic sulfamethoxazole-trimethoprim products. European countries have streamlined approval processes post-2015, facilitating quicker market entry for generics.
Market Demand and Prescriptions
Despite price decline, demand remains stable due to its prescription for common pediatric bacterial infections. Growth drivers include increased pediatric healthcare access, especially in emerging markets, and resistance issues reducing efficacy of certain classes, thus upholding demand for sulfamethoxazole-trimethoprim.
Manufacturing and Supply Chain
Global shortages of active pharmaceutical ingredients (APIs) during 2020-2021 caused price spikes temporarily but stabilized with increased manufacturing capacity. Future pricing will depend on supply chain resilience, particularly in light of geopolitical tensions affecting API sourcing.
What are the price projections for the next five years?
| Year | Projected Retail Price (per 100 mL) | Major Influences | Confidence Level |
|---|---|---|---|
| 2023 | $4.50 – $6.00 | Stable generics competition, no major patent actions | Medium |
| 2024 | $4.50 – $5.70 | Slight downward pressure from increased generics | High |
| 2025 | $4.35 – $5.50 | Continued generic market penetration | High |
| 2026 | $4.20 – $5.30 | Potential price stabilization, emerging markets expand | Medium |
| 2027 | $4.10 – $5.10 | Market saturation, patent expiries in some regions | Low |
Projection assumptions include persistent generic competition, regulatory stability, and steady pediatric prescription volume. The potential for disruptive policy changes or api cost fluctuations could alter this outlook.
What strategic considerations should companies pursue?
- Accelerate development and approval of lower-cost generic versions.
- Invest in formulations with improved stability or ease of administration to differentiate.
- Expand distribution channels in emerging markets with rising pediatric populations.
- Monitor regulatory trends that could impact pricing or patent status.
Key Takeaways
- The pediatric sulfamethoxazole-trimethoprim market is mature, with generic dominance leading to pressure on branded prices.
- In developed markets, prices are stabilized but falling gradually, influenced by increased generic competition and patent expiries.
- Emerging markets show growth potential driven by expanding healthcare infrastructure and affordability.
- Price stability over the next five years depends on regulatory policies, supply chain resilience, and prescription volume maintenance.
FAQs
1. How does patent expiration affect drug pricing?
Patent expiration enables generic competitors to enter the market, increasing supply and reducing prices typically by 50-70% for the original branded product.
2. Are there differences in pediatric formulations globally?
Yes. Some regions have access only to generics. Brand formulations like Bactrim are more common in the US, while local generic brands dominate elsewhere.
3. What is the projected market growth for pediatric antibiotics?
Approximately 4.2% CAGR through 2030, with sulfamethoxazole-trimethoprim accounting for about 10% of this segment.
4. How are regulatory policies impacting pricing?
Streamlined approval processes and policies favoring generic substitution drive prices downward across major markets.
5. What are potential risks to price stability?
API supply disruptions, regulatory changes restricting generic sales, and the development of resistance impacting prescriptions.
References
- Grand View Research. (2023). Pediatric Antibiotics Market Size, Share & Trends Analysis Report.
- U.S. Food and Drug Administration (FDA). (2022). Approved Generic Drugs Database.
- European Medicines Agency (EMA). (2022). Market Authorization Approvals and Expiry Data.
- Indian Pharmacopoeia Commission. (2022). API Manufacturing and Market Trends.
- China Food and Drug Administration. (2021). Regulatory Updates and Market Data.
More… ↓
