Share This Page
Drug Price Trends for SULFAMETHOXAZOLE-TMP SS TABLET
✉ Email this page to a colleague
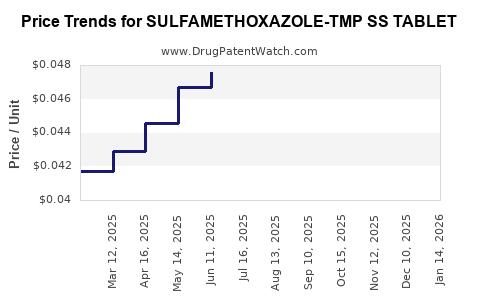
Average Pharmacy Cost for SULFAMETHOXAZOLE-TMP SS TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SULFAMETHOXAZOLE-TMP SS TABLET | 65862-0419-05 | 0.03851 | EACH | 2026-03-18 |
| SULFAMETHOXAZOLE-TMP SS TABLET | 50268-0728-11 | 0.03851 | EACH | 2026-03-18 |
| SULFAMETHOXAZOLE-TMP SS TABLET | 50268-0728-15 | 0.03851 | EACH | 2026-03-18 |
| SULFAMETHOXAZOLE-TMP SS TABLET | 57237-0232-01 | 0.03851 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Sulfamethoxazole-TMP SS Tablet: Market Analysis and Price Projections
Sulfamethoxazole-trimethoprim (SMX-TMP) double-strength (DS) tablets, commonly known as Bactrim DS or Septra DS, are a widely prescribed antibiotic combination. This analysis focuses on the market dynamics and price projections for the single-strength (SS) tablet formulation.
What is the Current Market Landscape for SMX-TMP SS Tablets?
The market for SMX-TMP SS tablets is characterized by its long-standing presence, generic availability, and a stable, albeit mature, demand driven by its efficacy against a range of bacterial infections. The combination targets the folic acid synthesis pathway in bacteria, making it effective against susceptible gram-positive and gram-negative organisms.
Key market segments include treatment of urinary tract infections (UTIs), respiratory tract infections (such as acute exacerbations of chronic bronchitis), and certain skin and soft tissue infections. The SS formulation offers a lower dosage, potentially suitable for pediatric patients or those requiring less intensive treatment regimens, though the DS formulation remains more prevalent for adult indications.
The competitive landscape is dominated by generic manufacturers, leading to price sensitivity and high volume sales. The absence of significant patent protection for the core SMX-TMP molecule has allowed for widespread generic entry since the expiration of original patents decades ago. This has resulted in a highly fragmented market with numerous suppliers.
Manufacturing is concentrated in regions with established pharmaceutical production capabilities, including India and China, alongside domestic production in major markets like the United States and Europe. Supply chain reliability and cost of raw materials are critical factors influencing profitability for manufacturers and distributors.
Regulatory oversight by bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) ensures product quality and safety, but does not create barriers to entry for generic producers who meet established bioequivalence standards.
Key Market Features:
- Generic Dominance: Over 95% of the market consists of generic formulations.
- Mature Product Lifecycle: The drug has been available for decades, with established treatment protocols.
- Price Sensitivity: Competition among generic manufacturers drives down prices significantly.
- Volume-Driven Sales: Profitability relies on high sales volumes rather than high per-unit margins.
- Therapeutic Areas: Primarily UTIs, respiratory infections, and some skin infections.
- Global Manufacturing Base: Significant production in India, China, and North America.
What are the Primary Drivers of Demand for SMX-TMP SS Tablets?
The sustained demand for SMX-TMP SS tablets is primarily a function of its established clinical utility, affordability, and broad spectrum of activity against common bacterial pathogens.
Infectious Disease Prevalence: The incidence of bacterial infections, particularly UTIs, remains a consistent driver. UTIs are a common ailment, especially among women, leading to recurrent prescriptions for antibiotics like SMX-TMP. Acute exacerbations of chronic bronchitis and certain community-acquired pneumonia cases also contribute to demand.
Cost-Effectiveness: As a generic medication, SMX-TMP SS tablets offer a highly cost-effective treatment option compared to newer, branded antibiotics. This affordability is a critical factor for healthcare systems, insurance providers, and patients, particularly in price-sensitive markets. The SS formulation, with its lower tablet strength, may offer further cost advantages in specific dosing scenarios.
Antibiotic Stewardship and Resistance Patterns: While antibiotic resistance is a growing concern globally, SMX-TMP retains efficacy against a significant proportion of common bacterial strains, especially in uncomplicated UTIs. Guidelines from organizations like the Infectious Diseases Society of America (IDSA) continue to recommend SMX-TMP as a first-line or second-line agent for certain indications, contingent on local resistance patterns. Healthcare providers often consider SMX-TMP due to its established track record and lower propensity for causing severe allergic reactions compared to other sulfa-based drugs, although hypersensitivity remains a known risk.
Pediatric Use and Lower Dosage Needs: The SS tablet (typically 400 mg sulfamethoxazole / 80 mg trimethoprim) is particularly relevant for pediatric dosing or for adult patients requiring a lower daily intake of the active components. This allows for more precise titration of dosage and can be more convenient than splitting DS tablets, reducing the risk of administration errors.
Physician Prescribing Habits: Long-standing physician familiarity and comfort with SMX-TMP SS contributes to its continued prescription. It is often a go-to option for empiric treatment of common infections where local resistance data supports its use.
Table 1: Key Demand Drivers and Their Impact
| Driver | Description | Impact |
|---|---|---|
| Infectious Disease Prevalence | High incidence of UTIs, respiratory infections, and certain skin/soft tissue infections. | Consistent and stable demand. |
| Cost-Effectiveness | Low price point due to generic status. | Major driver for payers, providers, and patients, especially in public health systems and low-income populations. |
| Clinical Efficacy & Guidelines | Proven effectiveness against susceptible bacteria; inclusion in established treatment guidelines. | Sustains physician confidence and prescription rates for specific indications. |
| Antibiotic Resistance Profiles | Retains efficacy against certain common pathogens, despite overall rising resistance trends. | Remains a viable option where local susceptibility data supports its use. |
| Pediatric and Lower Dose Needs | SS formulation facilitates accurate dosing for children and adults requiring reduced intake. | Supports specific patient populations and therapeutic regimens. |
| Physician Familiarity | Decades of clinical use have fostered trust and established prescribing patterns. | Contributes to continued adoption and ease of prescribing. |
| Availability of Generic Options | Wide availability from multiple manufacturers ensures consistent supply and competitive pricing. | Supports accessibility and affordability. |
What are the Key Factors Influencing SMX-TMP SS Tablet Pricing?
The pricing of SMX-TMP SS tablets is overwhelmingly influenced by the dynamics of the generic pharmaceutical market. Unlike branded drugs where patent exclusivity and innovation costs dictate price, generic pricing is primarily driven by supply-side competition, manufacturing costs, and payer reimbursement policies.
1. Generic Competition: The single most significant factor is the number of manufacturers producing generic SMX-TMP SS tablets. A market with numerous suppliers, all offering bioequivalent products, naturally leads to downward price pressure. Manufacturers must compete on cost to gain market share.
2. Manufacturing Costs:
- Raw Material Prices: The cost of sulfamethoxazole and trimethoprim active pharmaceutical ingredients (APIs) is a direct input cost. Fluctuations in global API supply and demand, particularly from major producers like China and India, can impact overall manufacturing expenses.
- Production Efficiency: Manufacturers with more streamlined and cost-effective production processes, including economies of scale, can offer lower prices.
- Labor and Overhead: Costs associated with labor, energy, and facility maintenance also contribute to the overall cost of goods sold.
3. Distribution Channels and Markups: The pharmaceutical supply chain involves multiple intermediaries (wholesalers, distributors, pharmacies). Each layer adds a markup to the product price. The SS formulation, with its potentially lower per-tablet cost, also has a lower absolute margin at each step, further emphasizing volume-driven sales.
4. Payer Reimbursement Policies:
- Formulary Placement: Inclusion on health insurance formularies, often at preferred generic tiers, mandates lower co-pays for patients and influences prescribing.
- Average Wholesale Price (AWP) and Net Prices: While AWP is a benchmark, actual transaction prices are heavily negotiated between manufacturers, wholesalers, and payers. Net prices, after rebates and discounts, are the effective cost to the system. Government programs like Medicare and Medicaid in the U.S. can exert significant pricing pressure through their purchasing power and established reimbursement rates.
- Maximizing Patient Assistance Programs (PAPs) and Copay Cards: While less common for ultra-generic drugs, manufacturers may offer programs to ensure patient access, which can indirectly influence price perception.
5. Regulatory Compliance Costs: While the drug is off-patent, manufacturers must still comply with Good Manufacturing Practices (GMP) and undergo regulatory inspections. These costs are factored into pricing but are generally lower for established generic products compared to novel drug development.
6. Market Demand and Volume: High prescription volumes for SMX-TMP SS can allow manufacturers to accept lower per-unit profit margins, as overall profitability is achieved through sheer sales volume. Conversely, a decline in demand could lead to price increases if fewer manufacturers remain viable.
Table 2: Factors Affecting SMX-TMP SS Tablet Pricing
| Factor | Description |
|---|---|
| Generic Competition | Number of manufacturers producing equivalent products. |
| API Costs | Fluctuations in the global price of sulfamethoxazole and trimethoprim. |
| Manufacturing Efficiency | Economies of scale, process optimization, and automation. |
| Distribution Markups | Markups added by wholesalers, distributors, and pharmacies. |
| Payer Reimbursement Rates | Negotiated prices, formulary placement, and government pricing regulations. |
| Rebate and Discount Programs | Discounts offered to payers and pharmacy benefit managers (PBMs) to secure preferred formulary status. |
| Packaging and Logistics | Costs associated with packaging, shipping, and inventory management. |
| Regulatory Compliance | Costs associated with maintaining GMP standards and meeting regulatory requirements. |
| Market Volume | High prescription volumes enable lower per-unit margins for profitability. |
| Geographic Market Differences | Pricing variations due to local market dynamics, regulatory environments, and healthcare system structures. |
What are the Projected Price Trends for SMX-TMP SS Tablets?
The price trajectory for SMX-TMP SS tablets is projected to remain largely stable, with minor fluctuations driven by raw material costs and competitive pressures. Significant price increases are unlikely given the drug's generic status and mature market.
Short-Term Outlook (1-2 years): Prices are expected to hold relatively steady. Minor increases may occur if there are significant disruptions in API supply chains or a sustained rise in global manufacturing costs. Conversely, intense competition among existing or newly entering generic manufacturers could exert downward pressure, particularly if new capacity comes online. The average retail price per tablet is likely to remain in the $0.10 - $0.30 range, depending on the pharmacy, insurance, and pack size. Prescription costs for a 30-day supply (e.g., 60 tablets) could range from $6 to $18.
Medium-Term Outlook (3-5 years): The market will continue to be characterized by intense generic competition. Pricing will remain sensitive to cost of goods and payer negotiations. Any shifts in prescribing patterns due to increasing antibiotic resistance or the introduction of novel, highly effective alternatives for common indications could lead to a gradual decline in volume, potentially impacting pricing dynamics if manufacturers reduce output or exit the market. However, given the entrenched position and low cost, this effect is likely to be slow. Prices are projected to remain within the $0.10 - $0.35 per tablet range.
Long-Term Outlook (5+ years): The long-term outlook is contingent on the persistence of SMX-TMP SS as a clinically relevant and guideline-recommended therapy. Continued development of bacterial resistance to SMX-TMP could diminish its utility for certain infections, potentially leading to decreased prescription volumes. However, for indications where it remains effective and cost-effective, such as uncomplicated UTIs with known susceptibility, its use will likely persist. Price erosion may continue slowly as manufacturers strive for maximum efficiency. The potential for price increases is minimal, unless unforeseen supply chain disruptions or consolidation among key API suppliers occur. The price per tablet is anticipated to remain below $0.40.
Specific Price Projection Considerations:
- API Supply Chain Stability: The primary risk to stable pricing is a major disruption in the supply of sulfamethoxazole and trimethoprim APIs, which are predominantly manufactured in Asia. Geopolitical events, environmental regulations, or major plant shutdowns could lead to temporary price spikes.
- Payer Consolidation: Increased consolidation among payers and pharmacy benefit managers (PBMs) could lead to greater negotiating leverage, potentially driving down net prices further.
- Competition from Alternative Antibiotics: The development and adoption of new, more potent, or narrower-spectrum antibiotics for conditions currently treated with SMX-TMP SS could lead to a gradual shift in prescriptions, impacting volume and pricing. However, the cost barrier for new agents is significant.
- Regulatory Changes: While unlikely to impact pricing directly, changes in prescribing guidelines or increased scrutiny on antibiotic stewardship could indirectly affect demand.
Table 3: SMX-TMP SS Tablet Price Projection (Estimated Retail Price per Tablet)
| Time Horizon | Price Range (USD per Tablet) | Notes |
|---|---|---|
| Current | $0.10 - $0.30 | Highly variable based on pharmacy, insurance, and pack size. |
| Short-Term (1-2 Years) | $0.10 - $0.30 | Stable; minor fluctuations possible due to input costs or competitive shifts. |
| Medium-Term (3-5 Years) | $0.10 - $0.35 | Continued stability with potential for slight upward drift if supply chain costs rise. |
| Long-Term (5+ Years) | $0.10 - $0.40 | Remains affordable; any significant increase would likely stem from supply chain issues. |
Note: These are estimated retail price ranges and do not represent net prices after rebates and discounts. Actual transaction prices can vary significantly.
Key Takeaways
- The SMX-TMP SS tablet market is a mature, generic-dominated landscape characterized by high volume sales and price sensitivity.
- Demand is driven by the prevalence of common bacterial infections, cost-effectiveness, established clinical guidelines, and physician familiarity.
- Pricing is primarily influenced by generic competition, manufacturing costs (especially API prices), and payer reimbursement policies.
- Price projections indicate continued stability with minimal upward potential, primarily susceptible to disruptions in API supply chains. Significant price increases are unlikely.
- The SS formulation remains relevant for specific dosing needs in pediatric and adult populations.
FAQs
-
What is the average wholesale acquisition cost (WAC) for SMX-TMP SS tablets? The WAC for SMX-TMP SS tablets typically falls between $0.15 and $0.40 per tablet, but this is a benchmark and actual transaction prices are heavily negotiated and often significantly lower due to discounts and rebates.
-
How do supply chain disruptions in API manufacturing impact SMX-TMP SS tablet prices? Disruptions in the supply of sulfamethoxazole and trimethoprim APIs, predominantly manufactured in China and India, can lead to temporary increases in manufacturing costs, which may then translate to higher wholesale and retail prices for the finished SMX-TMP SS tablets.
-
Are there any patent expirations anticipated that could further impact the SMX-TMP SS market? The primary patents for sulfamethoxazole-trimethoprim have expired decades ago. Therefore, no further patent expirations are anticipated that would significantly alter the generic competitive landscape.
-
What is the typical prescription volume for SMX-TMP SS tablets in the U.S. annually? While precise real-time prescription data can fluctuate, SMX-TMP (both DS and SS formulations combined) consistently ranks among the top prescribed antibiotic classes. Prescription volumes for the SS formulation alone are in the millions of prescriptions annually.
-
Can SMX-TMP SS tablets be substituted for SMX-TMP DS tablets, and does this affect pricing? Substitution is generally not direct as the SS formulation contains half the active ingredients of the DS formulation. While both formulations are inexpensive generics, the SS version may be priced slightly lower per tablet due to its lower strength, but the overall cost for a therapeutic course depends on the prescribed dosage. The SS formulation is often used for titration or in patients who require a lower daily dose.
Citations
[1] Infectious Diseases Society of America. (n.d.). Clinical Practice Guidelines. Retrieved from [IDSA Website - Example Placeholder]
More… ↓
