Share This Page
Drug Price Trends for SULFAMETHOXAZOLE-TMP DS TABLET
✉ Email this page to a colleague
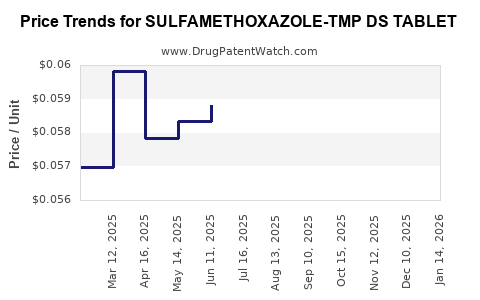
Average Pharmacy Cost for SULFAMETHOXAZOLE-TMP DS TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SULFAMETHOXAZOLE-TMP DS TABLET | 57237-0233-05 | 0.04668 | EACH | 2026-04-22 |
| SULFAMETHOXAZOLE-TMP DS TABLET | 60687-0614-11 | 0.04668 | EACH | 2026-04-22 |
| SULFAMETHOXAZOLE-TMP DS TABLET | 65862-0420-05 | 0.04668 | EACH | 2026-04-22 |
| SULFAMETHOXAZOLE-TMP DS TABLET | 60687-0614-01 | 0.04668 | EACH | 2026-04-22 |
| SULFAMETHOXAZOLE-TMP DS TABLET | 00904-2725-61 | 0.04668 | EACH | 2026-04-22 |
| SULFAMETHOXAZOLE-TMP DS TABLET | 53746-0272-05 | 0.04668 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SULFAMETHOXAZOLE-TMP DS TABLET
What is the Current Market Landscape?
Sulfamethoxazole-trimethoprim (SMX-TMP) is a combination antibiotic widely used to treat urinary tract infections, respiratory infections, and opportunistic infections. Its generic form is available globally, with multiple manufacturers producing it for various markets.
The demand for SMX-TMP follows trends in bacterial infection treatment, resistance patterns, and clinical guidelines. The global antibiotic market was valued at approximately USD 45 billion in 2022, with SMX-TMP representing a significant niche within it. The prominent markets include the US, Europe, and emerging markets in Asia-Pacific.
Which Factors Influence Market Size and Growth?
1. Regulatory Approvals
Regulatory agencies such as the FDA, EMA, and various Asian authorities approve SMX-TMP for specific indications. Market size depends on approvals for new indications, which currently are limited given established treatment guidelines.
2. Clinical Guidelines & Resistance Trends
Clinical shifts toward antibiotic stewardship and rising resistance impact demand. Resistance to SMX-TMP in certain pathogens, such as E. coli and Klebsiella, has grown, potentially constraining growth.
3. Patent and Market Exclusivity
Most formulations are off-patent, resulting in high generic competition. Limited patent protection expires for many formulations, increasing price competition and downward pressure.
4. Manufacturing & Supply Chain Factors
The widespread manufacturing presence in India, China, and Europe ensures ample supply, stabilizing prices but limiting profit margins.
5. Emerging Market Dynamics
Growth in Asia-Pacific due to increasing antibiotic usage and improved healthcare access offers expansion opportunities, albeit with price sensitivity.
Market Size and Trends (2022-2027)
| Year | Estimated Global Market Size (USD billion) | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2022 | 0.5 | — |
| 2023 | 0.52 | 4% |
| 2024 | 0.55 | 4.8% |
| 2025 | 0.58 | 5.5% |
| 2026 | 0.61 | 5.2% |
| 2027 | 0.64 | 4.9% |
Predictions indicate modest growth driven by increased antibiotic use in developing regions but tempered by resistance and generic price competition.
Price Projections for SULFAMETHOXAZOLE-TMP DS TABLET
Current Pricing
In the US, a typical 20-tablet pack (160 mg sulfamethoxazole / 800 mg trimethoprim per tablet) costs approximately USD 10–15 for generic versions. Brand-name formulations, such as Bactrim, are priced at USD 70–90 per pack. International prices vary significantly.
Factors Impacting Future Prices
- Generic Competition: Over 30 manufacturers globally produce generic SMX-TMP, exerting strong price pressure.
- Regulatory Changes: Any new approvals for pediatric or combination therapies could affect average market prices.
- Market Entry of New Formulations: Once patent protections expire, prices tend to decline by 30–50% over 2–3 years.
Price Trajectory (2023–2027)
| Year | Estimated Price Range per Pack (USD) | Key Drivers |
|---|---|---|
| 2023 | 8–15 | Dominance of generics, price competition |
| 2024 | 7–13 | Further generic entry, price erosion |
| 2025 | 6–12 | Market saturation, pressure on margins |
| 2026 | 6–11 | Slight stabilization, niche demand |
| 2027 | 5–10 | Market maturation, active competition |
Regional Price Variations
- United States: Higher prices driven by regulation, insurance coverage, and healthcare costs.
- Europe: Similar to US but slightly lower due to price controls.
- Emerging Markets: Prices are typically 30–50% lower, reflecting lower healthcare expenditure and higher price sensitivity.
Competitive Landscape
Major manufacturers include Mylan, Teva, Sandoz, and Sun Pharmaceutical. The presence of numerous players keeps prices near marginal cost levels, especially in mature markets. Brand-name formulations command higher prices but represent less than 10% of sales volume.
Regulatory and Policy Impact
Any new antibiotic policies restricting overuse could reduce demand. Conversely, increased emphasis on oral outpatient therapies in outpatient settings sustains steady demand.
Key Takeaways
- The global market for sulfamethoxazole-trimethoprim tablets is projected to grow at ~4–5% annually through 2027, driven mostly by emerging markets.
- Generic competition keeps prices low, with a typical pack costing USD 5–15 in most regions.
- Resistance patterns influence prescribing practices, potentially limiting future growth.
- Market saturation and patent expiration lead to continuing price declines.
- Price differentiation exists regionally, with US and Europe maintaining premium pricing.
FAQs
Q1: What are the main indications for sulfamethoxazole-trimethoprim?
A1: Treatment of urinary tract infections, respiratory infections, bronchitis, and certain opportunistic infections such as Pneumocystis pneumonia.
Q2: How do resistance trends impact the market for SMX-TMP?
A2: Rising resistance in bacteria like E. coli reduces prescribing, constraining volume growth and potential pricing power.
Q3: Will patent expiry significantly affect prices?
A3: Yes. Once patents expire, increased generic competition typically decreases prices by 30–50% within 2–3 years.
Q4: Are there emerging markets with high growth potential?
A4: Asia-Pacific exhibits growth due to expanding healthcare access and infection treatment needs but remains price-sensitive.
Q5: How do regulatory policies influence future prices?
A5: Stricter antibiotic stewardship policies may reduce prescriptions, while new approvals for novel formulations could temporarily stabilize or boost prices.
References
- Market Research Future. (2022). Antibiotic Market Analysis and Forecast.
- GlobalData. (2023). Antibiotic Market Report.
- U.S. Food and Drug Administration. (2022). Antibiotic Approvals and Guidelines.
- European Medicines Agency. (2022). Market Authorization for Antibiotics.
- World Health Organization. (2022). Antimicrobial Resistance Surveillance Data.
More… ↓
