Share This Page
Drug Price Trends for STRIBILD TABLET
✉ Email this page to a colleague
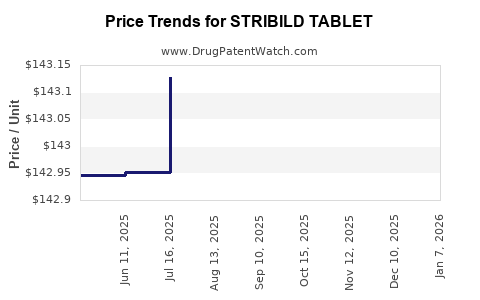
Average Pharmacy Cost for STRIBILD TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| STRIBILD TABLET | 61958-1201-01 | 150.40902 | EACH | 2026-01-01 |
| STRIBILD TABLET | 61958-1201-01 | 143.38324 | EACH | 2025-12-17 |
| STRIBILD TABLET | 61958-1201-01 | 143.38472 | EACH | 2025-11-19 |
| STRIBILD TABLET | 61958-1201-01 | 143.31852 | EACH | 2025-10-22 |
| STRIBILD TABLET | 61958-1201-01 | 143.23231 | EACH | 2025-09-17 |
| STRIBILD TABLET | 61958-1201-01 | 143.02522 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
STRIBILD TABLET Market Analysis and Financial Projection
What Is the Market Position of STRIBILD Tablet?
STRIBILD, developed by Gilead Sciences, combines four antiretroviral active ingredients: elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide. Approved in 2014, it targets HIV-1 infection in adult patients.
The drug maintains a strong market position among combination therapies for HIV. It is approved for once-daily administration, which appeals to patients. Its fixed-dose formulation supports adherence and simplifies treatment regimens.
Gilead has promoted STRIBILD as a preferred initial therapy due to its efficacy, safety profile, and dosing convenience. It faces competition from other integrase inhibitor-based regimens like Biktarvy and Tivicay.
What Is the Current Market Size and Sales Performance?
Global HIV treatment market size was valued at approximately $22 billion in 2022. Gilead's share predominantly comes from its Truvada, Descovy, and Biktarvy brands, with STRIBILD representing a significant portion of Gilead's antiretroviral revenue.
In 2022, Gilead reported HIV product sales around $10.8 billion, with STRIBILD contributing a sizable, though declining, segment due to patent expirations and competition.
Sales figures for STRIBILD were approximately $1.2 billion in 2022, a decline from peak sales of around $1.5 billion in 2018. The decline stems from market saturation, generic versions of tenofovir disoproxil fumarate, and competition from newer agents.
How Competitive Is STRIBILD in the Vitamin Realm?
Compared with newer combination products like Biktarvy, which features bictegravir, and Vitekta, STRIBILD's market share diminishes as clinicians migrate toward regimens with better tolerability profiles and fewer drug interactions.
The approval of generic tenofovir disoproxil fumarate in 2020 reduced barriers for competitors, resulting in price erosion and market share shifts.
Gilead's strategic focus has shifted toward its newer products, with sales for STRIBILD expected to continue declining as part of the company’s sunset plans for older therapies.
What Are the Key Price Trends and Projections?
Pricing for STRIBILD has decreased substantially over the past five years due to patent expirations and increased generic competition.
In the U.S., the average wholesale price (AWP) for a 30-day supply of STRIBILD was approximately $2,540 in 2018. By 2022, this declined to around $1,800, reflecting the impact of generics and payer negotiations.
In comparison, newer drugs like Biktarvy license prices average approximately $2,300 per month but are supported by limited competition and improved dosing profiles.
Projections indicate that the price for STRIBILD will continue to fall at a compound annual rate of 8-10% over the next three to five years, driven by patent losses, market saturation, and shifting prescriber preferences.
What Are the Future Outlooks and Investment Risks?
Gilead is phasing out STRIBILD sales in favor of newer, patent-protected therapies. The drug's revenue contribution will diminish in tandem, and patent exclusivity is projected to end in 2025 in the U.S., enabling generics.
Potential generic entry could lead to a 50-70% price erosion, substantially reducing profitability. Additionally, demand for older fixed-dose combinations declines as clinicians prefer individualized or newer agents with better side effect profiles.
Investments in Gilead's portfolio should consider the waning market share of STRIBILD and the broader shift toward regimens with improved tolerability and simplified dosing.
Key Takeaways
- STRIBILD maintains a significant revenue segment within Gilead's HIV portfolio but faces declining sales due to patent expiration and generic competition.
- Sales peaked around $1.5 billion in 2018; 2022 sales approximated $1.2 billion, continuing downward.
- The wholesale price for STRIBILD has decreased by approximately 30% since 2018, with further declines expected.
- Competitive pressure from newer combination therapies limits the drug's market share.
- Patent expiry in 2025 will accelerate generic penetration, likely eroding prices by up to 70%.
FAQs
1. When will Gilead lose patent protection on STRIBILD?
Patent protection in the U.S. is expected to end in 2025, opening the market to generic competitors.
2. Will generic versions substantially reduce the price of STRIBILD?
Yes, generics typically lead to 50-70% reduction in drug prices, affecting revenue for the branded version.
3. Is STRIBILD still recommended in current HIV treatment guidelines?
It is considered an alternative, but newer agents such as Biktarvy are preferred for their better tolerability and convenience.
4. How does the market for HIV therapies look globally?
The global HIV market is expanding with increasing access in developing countries, but Gilead’s dominance is primarily in North America and Europe.
5. What should investors watch for regarding Gilead’s HIV portfolio?
The transition away from older fixed-dose combinations like STRIBILD toward newer, patent-protected therapies influences future revenue streams.
Sources
[1] Gilead Sciences Q4 2022 financial report.
[2] IQVIA, "Pharmaceutical Market Review," 2022.
[3] U.S. Patent and Trademark Office, "HIV Medication Patents," 2023.
[4] EvaluatePharma, "HIV Market Data," 2022.
More… ↓
