Share This Page
Drug Price Trends for STELARA
✉ Email this page to a colleague
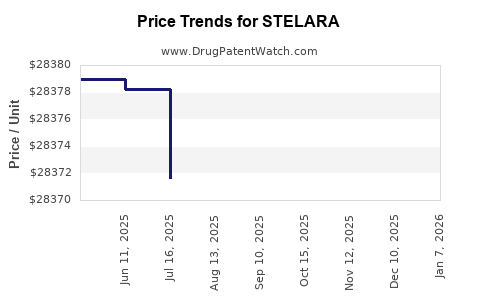
Average Pharmacy Cost for STELARA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| STELARA 90 MG/ML SYRINGE | 57894-0061-03 | 29794.54472 | ML | 2026-01-02 |
| STELARA 45 MG/0.5 ML SYRINGE | 57894-0060-03 | 29841.64800 | ML | 2026-01-02 |
| STELARA 45 MG/0.5 ML SYRINGE | 57894-0060-03 | 28420.61714 | ML | 2025-12-17 |
| STELARA 90 MG/ML SYRINGE | 57894-0061-03 | 28375.75688 | ML | 2025-12-17 |
| STELARA 90 MG/ML SYRINGE | 57894-0061-03 | 28372.62933 | ML | 2025-11-19 |
| STELARA 45 MG/0.5 ML SYRINGE | 57894-0060-03 | 28420.61714 | ML | 2025-11-19 |
| STELARA 90 MG/ML SYRINGE | 57894-0061-03 | 28364.36966 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
STELARA Market Analysis and Price Projections
STELARA (ustekinumab) is an interleukin-12 and -23 inhibitor approved for the treatment of moderate to severe plaque psoriasis, psoriatic arthritis, and Crohn's disease. Its market trajectory is influenced by patent expirations, biosimilar competition, and evolving treatment landscapes for its target indications.
What is the current market status of STELARA?
STELARA is a well-established biologic with significant global sales. As of 2023, it remains a top-selling drug, generating billions in annual revenue for Janssen Biotech, a subsidiary of Johnson & Johnson. Its broad therapeutic applications and proven efficacy contribute to its market dominance. However, the looming patent cliff presents a critical inflection point for its market performance.
Key Market Indicators:
- 2022 Global Sales: Approximately $9.7 billion [1].
- Primary Indications: Plaque psoriasis, psoriatic arthritis, Crohn's disease, ulcerative colitis.
- Mechanism of Action: Monoclonal antibody targeting the p40 subunit shared by interleukin-12 (IL-12) and interleukin-23 (IL-23) [2].
- Market Share: Holds a substantial share in the immunology biologic market.
When will STELARA face patent expiration and biosimilar entry?
The primary patents protecting STELARA have been subject to litigation and negotiation, leading to staggered patent expirations and anticipated biosimilar launches. The most significant patent expiries impacting the U.S. market are expected around late 2023 and early 2024, paving the way for biosimilar entrants. European patent expiries have a similar timeline, with some regions seeing earlier or later dates depending on individual patent strength and market exclusivity.
U.S. Patent Expiration Timeline (Anticipated):
- Key Composition of Matter Patents: Expired or with limited remaining exclusivity in the U.S. by late 2023 [3].
- Formulation and Method of Use Patents: May offer some residual protection, but generally considered weaker barriers to biosimilar entry.
- Anticipated Biosimilar Launch: U.S. biosimilar launches are expected to commence in late 2023/early 2024.
European Patent Expiration Timeline (Indicative):
- Varies by Country: Exact dates depend on national patent grants and supplementary protection certificates (SPCs).
- General Trend: Expirations in major European markets are largely aligning with U.S. timelines for biosimilar entry, with some variations.
Which biosimilar competitors are poised to enter the market?
Several pharmaceutical companies have developed biosimilar versions of ustekinumab. These companies have actively pursued U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) approval. The introduction of these biosimilars will introduce price competition and diversify treatment options.
Notable Biosimilar Developers:
- Celltrion Healthcare: Has developed CT-P43, a ustekinumab biosimilar with pending regulatory review.
- Samsung Bioepis: Has a ustekinumab biosimilar candidate in development, aiming for market entry post-patent expiry.
- Amgen: Has also been involved in the development of ustekinumab biosimilars.
- Organon: Through a partnership or internal development, may also be a competitor.
(Note: Specific market entry dates for individual biosimilars are contingent on regulatory approvals and manufacturing readiness.)
How will biosimilar entry impact STELARA's pricing and market share?
The introduction of biosimilars is expected to lead to a significant price reduction for ustekinumab products. Biosimilars typically launch at a discount to the reference biologic, and this trend is projected to continue. This price erosion will directly impact STELARA's revenue. Market share will also be redistributed as payers and providers adopt lower-cost biosimilar alternatives.
Projected Price Impact:
- Initial Discount: Biosimilars are anticipated to launch at discounts ranging from 15% to 30% compared to the list price of STELARA [4].
- Long-Term Erosion: As competition intensifies and more biosimilars gain approval, price discounts could deepen to 40% or more over time.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance companies will likely incentivize the use of biosimilars through formulary placement and rebate negotiations, accelerating price declines.
Market Share Dynamics:
- STELARA's Share Decline: J&J's STELARA revenue is projected to decrease by over 50% within a few years of biosimilar entry [5].
- Biosimilar Market Growth: The ustekinumab biosimilar market is forecast to reach multi-billion-dollar valuations rapidly after launch.
- Geographic Variations: The pace of biosimilar adoption and subsequent price erosion may vary across different countries and healthcare systems.
What are the long-term market projections for ustekinumab?
The long-term market for ustekinumab will be characterized by a bifurcated landscape: the reference product STELARA and its biosimilars. While STELARA's originator revenue will decline, the total market for ustekinumab (including biosimilars) is expected to remain robust, driven by its continued clinical utility and the expanding indications for interleukin inhibitors.
Future Market Landscape:
- Originator Product Decline: STELARA sales will see a steep drop, with J&J likely shifting focus to lifecycle management or other therapeutic areas.
- Biosimilar Market Expansion: The collective market share of ustekinumab biosimilars will grow substantially, becoming the dominant source of ustekinumab prescriptions.
- Therapeutic Area Evolution: The growth of IL-12/23 inhibitors, including ustekinumab biosimilars, will continue, potentially facing competition from newer biologic classes or oral small molecules targeting similar or different pathways.
Key Factors for Long-Term Growth:
- Demonstrated Efficacy: Continued real-world data supporting ustekinumab's efficacy across its approved indications.
- Payer Acceptance: Favorable formulary access and reimbursement policies for approved biosimilars.
- Physician Adoption: Clinician comfort and confidence in prescribing biosimilar ustekinumab products.
- Biosimilar Quality: Confirmation of high analytical, clinical, and manufacturing similarity by regulatory agencies.
What is the outlook for STELARA's price post-biosimilar entry?
Following the entry of multiple biosimilars, the price of ustekinumab is expected to decrease substantially. The reference product, STELARA, will likely see a phased price reduction to remain competitive, while biosimilar prices will establish a new, lower price benchmark for the molecule.
Price Projection Scenarios:
- Year 1 Post-Entry: STELARA list price potentially reduced by 20-35%. Biosimilar prices at 15-30% discount to the original STELARA list price.
- Year 3-5 Post-Entry: STELARA list price could be reduced by 40-60% from its pre-biosimilar peak. Biosimilar prices potentially at 40-55% discount to the original STELARA list price, creating a significant overall cost reduction for the drug.
- Negotiated Prices: Actual transaction prices will be heavily influenced by negotiations between manufacturers, PBMs, and payers, leading to variable net prices.
What are the potential risks and opportunities associated with STELARA's market transition?
The transition to a biosimilar market presents both risks and opportunities for stakeholders. For the originator, the primary risk is revenue erosion. For biosimilar manufacturers, the opportunity lies in capturing significant market share. Payers benefit from cost savings, while patients may gain improved access to treatment.
Risks:
- Originator Revenue Loss: Significant and rapid decline in STELARA sales for Johnson & Johnson.
- Market Access Challenges: Difficulties in securing favorable formulary placement for biosimilars amidst intense competition.
- Interchangeability Status: Lack of automatic interchangeability designation in some regions could slow biosimilar uptake compared to generic drugs.
- Supply Chain Disruptions: Potential for manufacturing or distribution issues impacting biosimilar availability.
Opportunities:
- Biosimilar Market Entry: Substantial revenue potential for biosimilar manufacturers.
- Cost Savings for Healthcare Systems: Reduced overall drug expenditure for payers and governments.
- Increased Patient Access: Lower drug costs may enable broader patient access to ustekinumab therapy.
- Innovation in Biologics: Resources freed up by cost savings could be reinvested in R&D for novel therapies.
Key Takeaways
- STELARA's market is entering a critical phase with anticipated biosimilar launches in late 2023/early 2024 in the U.S. and similar timelines in Europe.
- The introduction of biosimilar competitors will drive significant price erosion for ustekinumab, with projected discounts of 15-55% depending on market maturity and competition.
- Johnson & Johnson's STELARA revenue is expected to decline sharply post-biosimilar entry, while the overall ustekinumab molecule market (including biosimilars) is projected to remain substantial.
- Multiple biosimilar developers are poised to enter the market, intensifying competition and offering a wider range of treatment options.
- Long-term market success will depend on payer acceptance, physician adoption, and continued demonstration of ustekinumab's clinical efficacy and safety.
Frequently Asked Questions
-
What is the primary driver for the projected price decrease of STELARA? The primary driver is the anticipated entry of multiple biosimilar versions of ustekinumab, which are legally and therapeutically equivalent to the reference biologic and typically launch at a lower price point to compete.
-
Will STELARA be fully withdrawn from the market once biosimilars are available? It is unlikely that STELARA will be fully withdrawn. Johnson & Johnson may continue to market the originator product, potentially at a reduced price, to compete with biosimilars and leverage established brand loyalty.
-
How will biosimilar interchangeability status affect market adoption? In regions where a biosimilar is designated as interchangeable, pharmacists can substitute it for the reference biologic without prescriber intervention. This can accelerate adoption and increase market share compared to non-interchangeable biosimilars.
-
What are the key regulatory hurdles for ustekinumab biosimilars? Biosimilars must undergo rigorous review by regulatory bodies like the FDA and EMA to demonstrate high similarity to the reference product in terms of quality, safety, and efficacy. This includes analytical, pharmacokinetic, and pharmacodynamic studies, and potentially clinical trials.
-
Beyond price, what other factors will influence physician prescribing of ustekinumab biosimilars? Physician prescribing will be influenced by clinical trial data showing comparable efficacy and safety, manufacturing quality and consistency, supply chain reliability, and educational resources provided by biosimilar manufacturers.
Citations
[1] Johnson & Johnson. (2023). 2022 Annual Report. [2] Currentbiologics. (n.d.). Ustekinumab. Retrieved from https://currentbiologics.com/ustekinumab/ [3] Fierce Pharma. (2023, July 26). J&J’s Stelara patent fight continues as biosimilar rivals line up. [4] GlobalData. (2023). Ustekinumab Biosimilar Market Analysis. [5] Evaluate Pharma. (2023). STELARA® (ustekinumab) Drug Profile.
More… ↓
