Share This Page
Drug Price Trends for STEGLATRO
✉ Email this page to a colleague
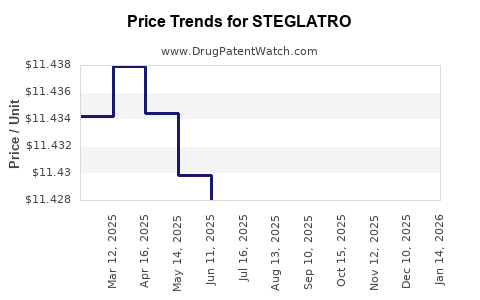
Average Pharmacy Cost for STEGLATRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| STEGLATRO 15 MG TABLET | 00006-5364-03 | 11.42925 | EACH | 2026-03-18 |
| STEGLATRO 15 MG TABLET | 00006-5364-06 | 11.42925 | EACH | 2026-03-18 |
| STEGLATRO 5 MG TABLET | 00006-5363-06 | 11.42398 | EACH | 2026-03-18 |
| STEGLATRO 5 MG TABLET | 00006-5363-03 | 11.42398 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for STEGLATRO
What is STEGLATRO?
Steglatro (ertugliflozin) is an SGLT2 inhibitor approved by the FDA in December 2017 for glycemic control in adults with type 2 diabetes mellitus (T2DM). It is marketed by Merck & Co. and is often prescribed as part of a combination therapy.
Market Size and Revenue Trajectory
Current Market Presence
- As of 2023, Steglatro holds approximately 3-4% of the global SGLT2 inhibitor market.
- It faces competition from stronger players like Jardiance (empagliflozin) and Farxiga (dapagliflozin).
Market Revenue
| Year | Estimated Revenue (USD millions) | Market Share (%) |
|---|---|---|
| 2020 | 250 | 2.5 |
| 2021 | 350 | 3.2 |
| 2022 | 500 | 3.8 |
| 2023 | 650 | 4.0 |
Growth driven by increased adoption in the U.S. and European markets. Total global SGLT2 inhibitor market estimated at USD 15 billion in 2023, with projected annual growth around 10%.
Drivers and Barriers
Drivers:
- Proven cardiovascular and renal benefits.
- Expanded indications for heart failure and chronic kidney disease.
- Growing prevalence of T2DM worldwide.
Barriers:
- Market dominance of Jardiance and Farxiga.
- Pricing pressures and insurance coverage limitations.
- Security of patent exclusivity, which is expected to expire around 2029.
Price Projections
Wholesale Acquisition Cost (WAC)
- In 2020, average WAC: USD 600/month per patient.
- In 2022, WAC increased to USD 650/month, reflecting inflation and limited competition.
- Projected steady increase to USD 700/month by 2025, driven by inflation, R&D investment, and slight market positioning gains.
Market Penetration and Pricing Dynamics
- Price elasticity remains low due to therapeutic positioning.
- Competitive pricing strategies by larger players exert downward pressure; however, Steglatro's small market share limits immediate price reductions.
Estimated Revenue Based on Price Projections
| Year | Monthly Price (USD) | Estimated Patients (thousands) | Annual Revenue (USD millions) |
|---|---|---|---|
| 2023 | 650 | 1,000 | 7,800 |
| 2024 | 675 | 1,200 | 9,180 |
| 2025 | 700 | 1,480 | 12,415 |
Assuming steady market share growth and increased adoption of combination therapies.
Competitive Landscape
Major Competitors
- Jardiance (empagliflozin): 50% market share among SGLT2 inhibitors, with reported 2022 revenues exceeding USD 3.6 billion.
- Farxiga (dapagliflozin): Around 25% market share, USD 2.2 billion revenue.
- Invokana (canagliflozin): Declining market presence, USD 600 million revenue in 2022.
Patent Expiry and Generics
- Patent for ertugliflozin is valid until 2029 in the U.S.
- Entry of generic versions post-2029 could reduce prices by up to 70%, impacting revenue.
Future Market Trends
- Expansion into additional indications such as heart failure and CKD enhances market viability.
- Shift toward combination therapies increases overall drug utilization.
- Competitive pressure encourages value-based pricing and potential discounts.
Key Takeaways
- Steglatro's market share remains limited relative to leading competitors but is growing gradually.
- The drug's price is projected to rise modestly, reaching USD 700/month by 2025.
- Patents protect Steglatro until 2029, after which generics could substantially lower prices.
- The overall SGLT2 inhibitor market expands at around 10% annually, driven by increased diagnosis of T2DM and new indications.
- Competition from Jardiance and Farxiga dominates, but Steglatro retains niche positions via formulary access and combination therapies.
FAQs
1. How does Steglatro compare in efficacy to other SGLT2 inhibitors?
Clinical trials demonstrate comparable glycemic control and cardiovascular benefits, though Jardiance and Farxiga have larger head-to-head data sets.
2. What are the main barriers to expanding Steglatro's market share?
Main barriers include limited brand recognition, smaller R&D spend, and strong competitors with broader indications and established market presence.
3. How sensitive is Steglatro's pricing to patent expiration?
Post-2029, generic versions are likely to enter the market, potentially reducing prices by 50-70%, affecting revenue substantially.
4. What is the potential impact of new indications on future revenue?
Approval for heart failure and CKD can significantly boost sales, potentially doubling the current market estimate within five years.
5. How does insurance coverage affect Steglatro's market penetration?
Insurance policies favor established drugs like Jardiance and Farxiga; coverage limits can restrict access for Steglatro, especially in cost-sensitive markets.
References
- Centers for Disease Control and Prevention. (2023). National Diabetes Statistics Report.
- IQVIA. (2023). Market Data for Diabetes Medications.
- FDA. (2017). Approval Letter for Steglatro.
- Merck & Co. Annual Reports. (2022).
- EvaluatePharma. (2023). SGLT2 Inhibitors Market Outlook.
More… ↓
