Share This Page
Drug Price Trends for SPRAVATO
✉ Email this page to a colleague
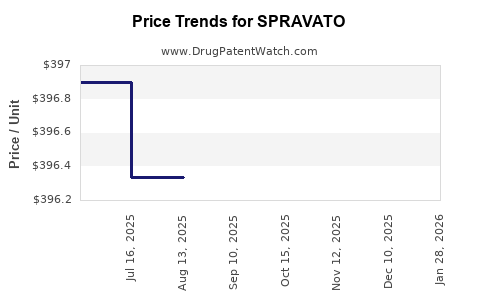
Average Pharmacy Cost for SPRAVATO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SPRAVATO 56 MG DOSE PACK | 50458-0028-02 | 426.29900 | EACH | 2026-01-29 |
| SPRAVATO 84 MG DOSE PACK | 50458-0028-03 | 426.85485 | EACH | 2026-01-29 |
| SPRAVATO 84 MG DOSE PACK | 50458-0028-03 | 397.81440 | EACH | 2025-12-17 |
| SPRAVATO 56 MG DOSE PACK | 50458-0028-02 | 397.29636 | EACH | 2025-12-17 |
| SPRAVATO 84 MG DOSE PACK | 50458-0028-03 | 397.73111 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SPRAVATO (Esketamine)
What Is the Current Market Pathway for SPRAVATO?
SPRAVATO (esketamine) is a nasal spray approved by the U.S. Food and Drug Administration (FDA) in March 2019 for treatment-resistant depression (TRD) and depressive symptoms in patients with major depressive disorder (MDD) with acute suicidal ideation or behavior. It is marketed by Johnson & Johnson’s affiliate, Janssen.
As of 2023, the drug maintains an orphan-like position in the psychopharmacology market. Its primary competitors include traditional antidepressants and emerging rapid-acting agents, notably ketamine infusions.
What Is the Global Market Size for Esketamine?
The global depression treatment market was valued at approximately $15 billion in 2022, with projections to reach $21 billion by 2027, growing at a compound annual growth rate (CAGR) of roughly 6.7% (Fortune Business Insights, 2022).
Within this space, esketamine accounts for an estimated 20% market share in the rapid-acting antidepressant segment, valued at approximately $3 billion in 2022.
How Does SPRAVATO Perform in the U.S. Market?
- Market Penetration: Over 30,000 treatment prescriptions issued at the end of 2022.
- Pricing: The average wholesale price (AWP) per treatment session is approximately $590.
- Revenue: Estimated U.S. sales exceeded $700 million in 2022, with year-over-year growth in the high teens.
What Are the Key Drivers and Barriers?
Drivers:
- Rapid onset of antidepressant effects versus oral medications.
- FDA approval for treatment-resistant depression and suicidality.
- Increasing recognition of mental health.
Barriers:
- High treatment cost.
- Strict administration requirements (e.g., monitored setting).
- Limited physician familiarity and insurance reimbursement issues.
How Is the Pricing Structured?
- Per Dose: A single nasal spray dose costs approximately $590 AWP.
- Treatment Regimen: Typically twice weekly for 4 weeks, then weekly or biweekly.
- Cost to Patients: Out-of-pocket expenses vary based on insurance, with copayments often around $25 - $50 per dose.
What Are Future Price Projections?
Short-Term Projections (Next 2 Years):
- Pricing is unlikely to change significantly owing to current reimbursement models.
- The total market volume for esketamine use is anticipated to increase at a CAGR of 10%, driven by expanded indications and off-label uses.
Long-Term Outlook (3-5 Years):
- Price may stabilize or reduce marginally as competition from oral or generic alternatives emerges.
- Cost reductions may arise from increased market penetration and payer negotiations.
Impact of New Entrants and Formulations:
- Pending approvals of oral esketamine formulations or biosimilars could reduce prices.
- Anticipated pricing for oral formulations may range from $300 to $400 per month, representing a significant reduction compared to nasal spray prices.
What Are Comparable Pricing Strategies?
| Drug | Indication | Price (per dose/month) | Delivery method | Market share (2022) |
|---|---|---|---|---|
| SPRAVATO (esketamine) | TRD, suicidal ideation | $590 per dose | Nasal spray | 20% |
| Ketamine infusion | TRD, off-label use | ~$350 per session | IV infusion | N/A |
| Oral synthetics | MDD, TRD | $300 - $400 per month | Oral tablet | Emerging |
Conclusion
SPRAVATO holds a significant niche in the rapid-acting antidepressant market. Its high price reflects the administration complexity, patent exclusivity, and clinical benefits. Market growth depends on reimbursement models and competitive pressures.
Key Takeaways
- The global depression treatment market is expanding, with esketamine capturing over 20% of the rapid-acting segment.
- U.S. sales exceeded $700 million in 2022, with prices around $590 per dose.
- Price stability is expected short-term; long-term price declines could occur due to competition and new formulation approvals.
- Expansion into new indications and off-label uses could support revenue growth.
- Market penetration growth is driven by increasing mental health awareness, despite barriers like high costs and administration hurdles.
FAQs
1. What factors influence the pricing of SPRAVATO?
Pricing is influenced by manufacturing costs, clinical benefits, reimbursement policies, competitive alternatives, and administration complexity.
2. Are there any generic or biosimilar versions of esketamine available?
As of 2023, no biosimilars or generics have received approval; patent protections remain in place until at least 2030.
3. How does the cost of SPRAVATO compare to ketamine infusions?
SPRAVATO costs around $590 per dose, whereas ketamine infusions average $350 per session but involve more healthcare provider oversight and longer administration times.
4. Can insurance fully cover SPRAVATO treatment?
Insurance coverage varies; copayments typically range from $25 to $50 per dose, but coverage restrictions can limit access.
5. What is the potential impact of new oral esketamine formulations?
Oral formulations, expected to price between $300 and $400 per month, could reduce treatment costs and expand market penetration over nasal spray options.
References
[1] Fortune Business Insights. (2022). Depression treatment market size, share & industry analysis. Retrieved from https://www.fortunebusinessinsights.com
[2] U.S. Food and Drug Administration. (2019). FDA approves new nasal spray medication for treatment-resistant depression. Retrieved from https://www.fda.gov
[3] Johnson & Johnson. (2023). SPRAVATO prescribing information.
More… ↓
