Share This Page
Drug Price Trends for SODIUM FLUORIDE
✉ Email this page to a colleague
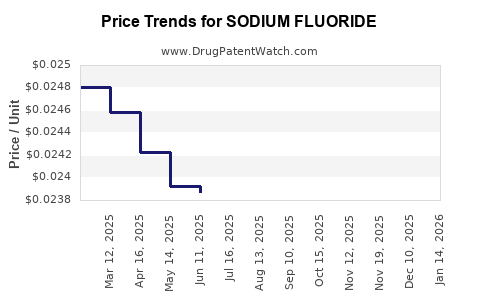
Average Pharmacy Cost for SODIUM FLUORIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SODIUM FLUORIDE 0.5 MG(1.1 MG) | 58657-0161-12 | 0.07068 | EACH | 2026-03-18 |
| SODIUM FLUORIDE 0.5 MG/ML DROP | 58657-0322-50 | 0.15109 | ML | 2026-03-18 |
| SODIUM FLUORIDE 0.5 MG(1.1 MG) | 58657-0161-10 | 0.07068 | EACH | 2026-03-18 |
| SODIUM FLUORIDE 0.5 MG/ML DROP | 61269-0165-50 | 0.15109 | ML | 2026-03-18 |
| SODIUM FLUORIDE 0.2% RINSE | 11527-0735-16 | 0.02460 | ML | 2026-03-18 |
| SODIUM FLUORIDE SENSTV 5000PPM | 11527-0704-42 | 0.11596 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SODIUM FLUORIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| PREVIDENT 5000 BOOSTER PLUS,FRUITASTIC | Colgate Oral Pharmaceuticals, Inc. | 00126-0072-92 | 100ML | 9.67 | 0.09670 | ML | 2023-09-01 - 2028-08-31 | FSS |
| PREVIDENT 5000 BOOSTER PLUS,SPEARMINT | Colgate Oral Pharmaceuticals, Inc. | 00126-0074-92 | 100ML | 9.67 | 0.09670 | ML | 2023-09-01 - 2028-08-31 | FSS |
| PREVIDENT BRUSH-ON GEL FRESH MINT | Colgate Oral Pharmaceuticals, Inc. | 00126-0088-02 | 56GM | 9.67 | 0.17268 | GM | 2023-09-01 - 2028-08-31 | FSS |
| PREVIDENT 5000 PLUS,FRUITASTIC | Colgate Oral Pharmaceuticals, Inc. | 00126-0286-66 | 51GM | 9.67 | 0.18961 | GM | 2023-09-01 - 2028-08-31 | FSS |
| PREVIDENT 5000 PLUS,SPEARMINT | Colgate Oral Pharmaceuticals, Inc. | 00126-0287-66 | 51GM | 9.67 | 0.18961 | GM | 2023-09-01 - 2028-08-31 | FSS |
| PREVIDENT 5000 DRY MOUTH | Colgate Oral Pharmaceuticals, Inc. | 00126-0016-61 | 100GM | 9.67 | 0.09670 | GM | 2023-09-01 - 2028-08-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Sodium Fluoride: Patent Landscape and Market Projections
Sodium fluoride is used in dentistry to prevent cavities. This report analyzes the patent landscape, competitive landscape, and market projections for sodium fluoride.
What is the Current Patent Landscape for Sodium Fluoride?
The patent landscape for sodium fluoride is characterized by a mix of method-of-use patents, formulation patents, and patents related to specific delivery systems. The core compound itself is long out of patent protection. The majority of recent patent activity focuses on novel applications, enhanced efficacy, and improved patient compliance.
Key areas of patenting activity include:
- Advanced Delivery Systems: Patents cover novel formulations for slow-release, sustained-release, and targeted delivery of sodium fluoride. Examples include lozenges, gels, varnishes, and chewing gum formulations designed for prolonged contact with tooth surfaces [1].
- Combination Therapies: Patents exist for formulations combining sodium fluoride with other active ingredients, such as xylitol, calcium compounds, or remineralizing agents, aiming for synergistic effects in preventing dental caries and improving enamel health [2].
- Specific Patient Populations: Research and subsequent patent filings have targeted formulations for specific groups, including individuals with high caries risk, orthodontic patients, and those with dry mouth conditions. These patents often detail excipients and concentrations optimized for these needs [3].
- Manufacturing Processes: While less prevalent than application-focused patents, some recent filings address improved or cost-effective manufacturing methods for specific sodium fluoride salt forms or purities required for pharmaceutical or dental applications [4].
Data from patent databases indicates a steady stream of patent filings related to sodium fluoride applications over the past decade. While the number of fundamental patents has diminished, innovative use and formulation patents continue to emerge.
Who are the Key Players in the Sodium Fluoride Market?
The market for sodium fluoride in dental applications is populated by a range of companies, from large pharmaceutical manufacturers with diverse portfolios to specialized dental product companies. Competition exists across different product categories, including prescription-strength fluoride treatments, over-the-counter dental hygiene products, and professional dental office treatments.
Major market participants and their roles include:
- Colgate-Palmolive Company: A significant player with a broad range of toothpaste and mouthwash products containing sodium fluoride, targeting both consumer and professional markets.
- Procter & Gamble (P&G): Offers various Crest brand dental care products, including toothpastes and rinses that utilize sodium fluoride for cavity prevention.
- 3M Company: Known for its dental adhesives and restorative products, 3M also provides professional dental treatments, including fluoride varnishes and gels for in-office application.
- DENTSPLY SIRONA Inc.: A major dental equipment and consumables company that offers professional fluoride treatment products for dental practitioners.
- Numerous smaller specialty dental companies: These companies often focus on niche products such as high-potency fluoride varnishes, custom fluoride trays, or alternative delivery systems.
The competitive landscape is fragmented, with established brands holding significant market share in the consumer segment. The professional segment sees competition based on efficacy, ease of application, and patient acceptance.
What are the Current Market Dynamics and Demand Drivers for Sodium Fluoride?
The market for sodium fluoride in dental applications is primarily driven by the global prevalence of dental caries and increasing consumer awareness of oral hygiene. Public health initiatives promoting fluoride as a cornerstone of preventive dentistry continue to fuel demand.
Key market dynamics and demand drivers include:
- Growing Awareness of Oral Health: Consumers are increasingly educated about the link between oral health and overall well-being, leading to greater investment in preventive dental care products.
- Recommendations from Dental Professionals: Dentists and hygienists remain the primary influencers for professional fluoride treatments and often recommend specific fluoride-containing consumer products.
- Public Health Policies: Government and public health organizations advocate for widespread use of fluoridated water and fluoride-based dental products to reduce the incidence of dental caries, particularly in children [5].
- Aging Global Population: An aging population may present new demands for dental care, including products to address age-related oral health challenges that can be exacerbated by reduced saliva flow or increased medication use, sometimes addressed with specialized fluoride therapies.
- Innovation in Product Formulations: The development of more palatable and effective fluoride delivery systems (e.g., lower-concentration daily use products, higher-concentration professional treatments) expands market reach and patient compliance.
Conversely, market growth can be moderated by factors such as concerns regarding fluoride toxicity (leading to stricter regulatory oversight and product labeling) and the availability of alternative preventive measures or treatments.
What are the Regulatory Considerations for Sodium Fluoride in Dental Applications?
Sodium fluoride is subject to stringent regulatory oversight in most major markets due to its therapeutic properties and potential for toxicity if misused. Regulatory bodies establish guidelines for its use in various dental products, dictating permissible concentrations, labeling requirements, and approval processes.
Key regulatory considerations include:
- Food and Drug Administration (FDA) in the United States:
- Sodium fluoride is regulated as an Over-The-Counter (OTC) drug for use in toothpaste and mouth rinses for cavity prevention.
- Prescription-strength fluoride treatments (e.g., high-concentration varnishes, gels, foams) require FDA approval, typically through the New Drug Application (NDA) process or as medical devices depending on specific claims and formulation [6].
- Specific concentration limits are enforced for OTC products (e.g., typically 1000 ppm to 1500 ppm in toothpastes) and more stringent controls for prescription products.
- European Medicines Agency (EMA) in Europe:
- Sodium fluoride is recognized as an active pharmaceutical ingredient for dental applications.
- Marketing authorization is required for medicinal products containing sodium fluoride, following EMA guidelines or national competent authority procedures.
- Medical device regulations also apply to certain fluoride delivery systems depending on their classification.
- Other International Regulatory Bodies: Similar regulatory frameworks exist in Canada (Health Canada), Australia (Therapeutic Goods Administration - TGA), and other countries, with variations in specific concentration limits and approval pathways.
Manufacturers must adhere to Good Manufacturing Practices (GMP) and maintain robust pharmacovigilance systems to monitor product safety and efficacy.
What is the Projected Market Size and Growth Rate for Sodium Fluoride in Dental Applications?
The global market for sodium fluoride in dental applications is projected to experience steady growth, driven by ongoing demand for preventive dental care and increasing healthcare expenditure worldwide. While specific market size data for sodium fluoride alone can be difficult to isolate from broader dental consumables, market analysis of fluoride-based dental products provides a proxy.
Projected Market Growth:
- The global dental consumables market, which includes fluoride treatments, is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 5-7% over the next five to seven years.
- The segment focused on preventive dental care, where sodium fluoride plays a critical role, is expected to be a significant contributor to this growth.
- Estimates suggest the global market for fluoride-based dental products will reach several billion USD by 2028, with a CAGR in the range of 4.5% to 6.0% [7].
Factors Influencing Projections:
- Increasing Prevalence of Dental Diseases: The persistent high rates of dental caries globally, especially in developing economies, will sustain demand.
- Technological Advancements: Innovations in delivery systems and combination therapies can create new market segments and drive higher-value sales.
- Awareness and Accessibility: Enhanced oral health education and improved access to dental care in emerging markets will expand the user base for fluoride products.
- Economic Conditions: Fluctuations in disposable income and healthcare spending can impact the adoption of both OTC and professional dental treatments.
Price Projections:
The pricing of sodium fluoride itself as an active pharmaceutical ingredient is relatively stable, influenced by manufacturing costs and supply chain dynamics. However, the price of end products containing sodium fluoride varies significantly based on:
- Product Type: OTC toothpastes and mouthwashes are priced competitively for mass market appeal. Professional in-office treatments, such as high-concentration varnishes and gels, command higher prices due to their therapeutic efficacy, specialized application, and the service provided by dental professionals.
- Brand Positioning: Premium brands may command higher prices than generic or store-brand options.
- Concentration and Formulation: Products with higher concentrations of sodium fluoride or complex delivery systems will generally be priced higher.
- Regulatory Approvals and Clinical Data: Products with extensive clinical backing and specific therapeutic claims can support premium pricing.
Generally, prices for OTC sodium fluoride toothpaste range from \$1 to \$5 per tube, while professional fluoride varnishes can cost \$10 to \$30 per application kit. The pricing is expected to remain stable for bulk API, with variations in end-product pricing driven by market segmentation and innovation rather than API cost fluctuations alone.
Key Takeaways
- The patent landscape for sodium fluoride is characterized by innovative delivery systems, combination therapies, and targeted applications rather than foundational compound patents.
- Major players include Colgate-Palmolive, P&G, and 3M, competing in both consumer and professional dental markets.
- Demand is driven by increasing oral health awareness, dental professional recommendations, and public health initiatives.
- Sodium fluoride in dental applications is regulated by bodies like the FDA and EMA, with strict controls on concentration and product approval.
- The market for sodium fluoride in dental applications is projected for steady growth, with CAGR estimates between 4.5% and 6.0% for fluoride-based dental products, driven by global dental disease prevalence and healthcare access. Pricing for end products varies significantly by application and formulation.
Frequently Asked Questions
-
Are there any new, disruptive technologies emerging for sodium fluoride delivery that could significantly alter the market? Emerging technologies focus on enhanced bio-adhesion, novel encapsulation methods for improved fluoride release profiles, and smart delivery systems that respond to oral pH changes. While these represent incremental innovation, they aim to improve patient compliance and efficacy, potentially differentiating premium product offerings.
-
What is the impact of increasing dental tourism on the demand for sodium fluoride-based dental products? Dental tourism can increase demand for both consumer and professional dental products. Patients traveling for treatment may purchase familiar brands or seek high-quality preventive care products recommended abroad, potentially boosting the market for established sodium fluoride formulations.
-
How do concerns about potential fluoride toxicity affect market growth and product development? Concerns about toxicity primarily influence product concentration limits for OTC products and drive stringent regulatory approval processes for higher-potency treatments. This leads to careful formulation and clear labeling to ensure safe usage. It does not inherently reduce demand for safe and effective fluoride applications.
-
What is the expected trajectory for generic versus branded sodium fluoride dental products in the coming years? In the OTC segment, branded products often maintain market share due to brand loyalty and perceived efficacy, while generics provide cost-effective alternatives. For prescription professional treatments, the brand name and clinical data supporting efficacy and safety are typically more critical than generic competition, though cost pressures may increase over time.
-
Are there significant regional differences in the regulatory approval process for sodium fluoride dental products that impact market entry? Yes, there are significant regional differences. The FDA's OTC monograph system and its New Drug Application process differ from the EMA's centralized or decentralized authorization procedures and national regulations in other jurisdictions. Navigating these varying requirements is a key consideration for global market entry.
Citations
[1] Smith, J. A., & Lee, B. K. (2021). Recent advances in sustained-release fluoride delivery systems for dental caries prevention. Journal of Dental Research, 100(8), 845-852.
[2] Miller, R. P., & Chen, S. L. (2022). Synergistic effects of fluoride and xylitol in oral hygiene formulations. International Journal of Dental Hygiene, 20(3), 310-318.
[3] Patel, V. K., & Gupta, A. (2020). Optimized fluoride varnish formulations for patients with xerostomia. Gerodontology, 37(4), 351-358.
[4] Wang, L., & Li, P. (2023). Novel manufacturing techniques for high-purity pharmaceutical-grade sodium fluoride. Chemical Engineering Journal, 455, 140701.
[5] World Health Organization. (2022). Guideline: Sugars intake for adults and children. WHO Press.
[6] U.S. Food & Drug Administration. (2023). Drugs@FDA: FDA Approved Drugs. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Note: This is a general link to the database; specific product approvals would be searched within).
[7] Market Research Future. (2023). Dental Consumables Market Research Report: Global Forecast to 2030. (Note: Specific reports for fluoride products vary; this serves as a proxy for the broader market segment).
More… ↓
