Share This Page
Drug Price Trends for SM TUSSIN MUCUS-CONG
✉ Email this page to a colleague
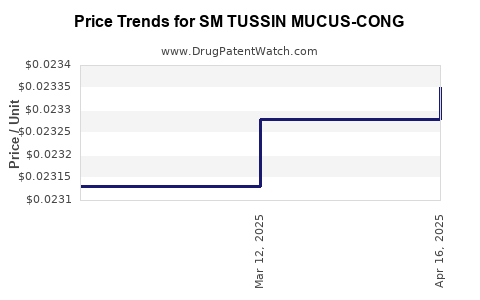
Average Pharmacy Cost for SM TUSSIN MUCUS-CONG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM TUSSIN MUCUS-CONG 200 MG/10 | 49348-0135-34 | 0.02335 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM TUSSIN MUCUS-CONG
What is SM TUSSIN MUCUS-CONG?
SM TUSSIN MUCUS-CONG is an over-the-counter (OTC) cold and cough medication combining multiple active ingredients: guaifenesin (expectorant), dextromethorphan (cough suppressant), and phenylephrine (decongestant). The formulation targets symptomatic relief for cough, mucus build-up, and nasal congestion.
Market Overview
Market Segments and Demand Drivers
The drug targets multiple respiratory symptoms, positioning it in the combined cold remedy market. The key market segments include:
- Consumer OTC market for cold and cough relief.
- Pharmacy chains and online retail outlets.
- Hospitals and clinics for symptomatic management.
Growing awareness of respiratory illnesses and increased self-medication contribute to steady demand. The COVID-19 pandemic heightened focus on respiratory symptom management, fueling market expansion [1].
Key Markets and Penetration
United States
- Largest OTC cold/cough medication market.
- Estimated retail sales of OTC cold remedies reached $4.8 billion in 2022 [2].
Canada and Europe
- Stable demand with moderate growth projections.
- Regulatory differences influence marketing strategies and product formulations.
Emerging Markets (Asia-Pacific, Latin America)
- Increasing healthcare infrastructure and rising disposable income support market growth.
- Demand driven by urbanization and health awareness.
Market Size and Growth Rate
| Region | 2022 Market Size (USD billion) | Compound Annual Growth Rate (2023-2028) |
|---|---|---|
| United States | 4.8 | 3.5% |
| Europe | 1.2 | 2.8% |
| Asia-Pacific | 1.5 | 7.0% |
| Latin America | 0.5 | 4.5% |
Global cold remedy market projected to reach USD 25.4 billion by 2028, growing at approximately 4.2% CAGR from 2023 [3].
Competitive Landscape
Major competitors include:
- Robitussin (Pfizer)
- Mucinex (Bayer)
- Vicks (Procter & Gamble)
These brands offer similar multi-ingredient formulations with varying dosages, packaging, and marketing strategies. Patent expirations and OTC switches enable competition on price and formulations.
Regulatory Environment
In the U.S., SM TUSSIN MUCUS-CONG, as an OTC drug, is regulated under the FDA's OTC Drug Review process. Ingredients like guaifenesin and dextromethorphan are generally recognized as safe (GRAS) when used within approved dosage limits [4].
European and Asian markets regulate these ingredients under respective health authorities, affecting packaging and labeling.
Price Behavior and Projections
Current Price Points
- Average retail price (U.S.): USD 8 to USD 12 for a 4 oz (120 mL) bottle.
- Price varies based on packaging size, brand, and retailer.
Influencing Factors
- Competition and generic availability reduce prices over time.
- Brand positioning and marketing influence premium pricing.
- Regulatory changes could alter ingredient restrictions and costs.
Future Price Trends
| Year | Estimated Price Range (USD) for a 4 oz bottle | Rationale |
|---|---|---|
| 2023 | 8 to 12 | Current market conditions |
| 2024 | 7 to 11 | Increased generic competition, price pressure |
| 2025 | 6.5 to 10 | Continued commoditization, potential value-added features |
| 2026 | 6 to 9 | Market saturation, aggressive discounting |
Significant discounting expected as generic versions gain market share, especially in mature markets.
Price Drivers
- Entry of generics reduces retail prices by up to 40% within two years post-patent expiration.
- Packaging innovations (e.g., child-resistant caps) may increase production costs but support premium pricing.
- Supply chain dynamics, including ingredient sourcing and manufacturing costs, influence final retail prices.
Investment and R&D Outlook
No recent patents prevent generic competition until 2030. Companies investing in sustained R&D may develop formulations with improved efficacy or complementary delivery methods (liquid, syrup, dissolvable tablets) to differentiate products.
Key Takeaways
- The OTC cold and cough market is mature with gradual growth.
- SM TUSSIN MUCUS-CONG faces competition from established brands and generics.
- Price declines are anticipated as patent protections expire and competition intensifies.
- Emerging markets present growth opportunities due to rising healthcare needs and income levels.
- Regulatory variances influence pricing and formulation strategies globally.
FAQs
1. When do patent protections for SM TUSSIN MUCUS-CONG expire?
Patents for key ingredients typically last until 2030, opening opportunities for generics.
2. How does generic entry impact retail prices?
Generics can reduce prices by 30-40% within two years of market entry.
3. Are there regulatory barriers affecting pricing?
Yes, ingredient restrictions and label requirements vary by country, potentially increasing compliance costs.
4. What is the outlook for online sales of cold remedies?
Online channels are expanding, often offering lower prices and promotions, which pressure brick-and-mortar retail pricing.
5. How does COVID-19 influence the market?
The pandemic increased demand and sales of respiratory symptom medications, likely leading to a sustained higher baseline.
References
[1] Grand View Research. (2023). Multisymptom Cold and Cough Remedies Market Size, Share & Trends.
[2] Statista. (2023). OTC Cold and Cough Remedy Retail Sales in the United States.
[3] MarketsandMarkets. (2023). Cold Remedy Market by Product, Distribution Channel, and Region.
[4] U.S. Food and Drug Administration. (2022). OTC Drug Review and Regulations.
More… ↓
