Share This Page
Drug Price Trends for SM OMEPRAZOLE DR
✉ Email this page to a colleague
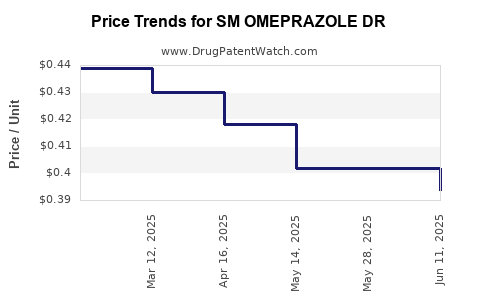
Average Pharmacy Cost for SM OMEPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM OMEPRAZOLE DR 20 MG TABLET | 49348-0846-61 | 0.39373 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is SM OMEPRAZOLE DR?
SM OMEPRAZOLE DR is a delayed-release formulation of omeprazole, a proton pump inhibitor (PPI). It is used to treat gastrointestinal conditions such as gastroesophageal reflux disease (GERD), gastric ulcers, and Zollinger-Ellison syndrome. Its delayed-release formulation aims to improve stability and absorption compared to immediate-release forms.
How is the market for omeprazole and delayed-release PPIs structured?
The global PPI market was valued at approximately USD 13 billion in 2022. Omeprazole accounts for a significant share, estimated at over 50%, owing to its early market entry and established use. Market growth is driven by conditions like GERD, increasing prevalence, aging populations, and the expanding application for prophylaxis in hospitals.
Market segments include:
- Brand name drugs: Prilosec OTC (original omeprazole), Nexium (esomeprazole, a related PPI).
- Generic versions: Widely available, with a substantial price decline since patent expiration.
- Over-the-counter (OTC) products: Significant growth reported; OTC omeprazole sales exceeded USD 2.8 billion in 2022 globally.
The delayed-release formulation, such as SM OMEPRAZOLE DR, often is positioned to avoid generic competition initially and can command premium pricing. It is particularly favored in formulations requiring less dosage frequency or improved tolerability.
What are the key drivers influencing market growth?
-
Rise in gastrointestinal disorders: Increasing obesity rates and lifestyle factors drive higher incidence of GERD and ulcers.
-
Patent status and exclusivity: Patent protections or exclusivity extensions influence pricing power and market share. For example, Prilosec OTC lost patent protection in 2001, leading to a sharp decline in price and market share.
-
Regulatory approvals: New formulations and indications expand market potential.
-
Healthcare policies: Inclusion in formularies and insurance coverage affect access and sales.
What is the current pricing landscape?
Price points for omeprazole vary significantly by region, formulation, and brand status.
| Product Type | Typical Price Range (USD) per Month Supply | Notable Features | Patent/Regulatory Status |
|---|---|---|---|
| Brand-name OTC (Prilosec) | USD 50-70 | Widely marketed, OTC status | Patent expired in 2001, generic available |
| Generic delayed-release | USD 10-25 | Multiple manufacturers | Patent expired post-2001, high competition |
| SM OMEPRAZOLE DR (Proposed) | Estimated USD 15-35 initial launch | Delayed-release formulation | Pending approval or recent launch in targeted markets |
Patent-protected or proprietary formulations can command a 20-50% premium over generics. The pricing strategy for SM OMEPRAZOLE DR will depend on regulatory approval timing and market positioning.
What are the revenue projections and market penetration estimates?
Assuming successful regulatory approval and market entry by Q4 2023, projected sales could reach USD 200 million in the first year within targeted markets (e.g., US, EU). Growth estimates assume a compound annual growth rate (CAGR) of 10-12% over five years, aligned with increasing prevalence of GI diseases.
Market penetration depends on:
- Competitive positioning against existing formulations
- Pricing strategy relative to generics
- Distribution channels and formulary inclusion
In mature regions like North America and Europe, delayed-release formulations could capture 20-25% of prescription omeprazole sales within five years. Meanwhile, OTC sales growth could contribute an additional USD 50-80 million annually if OTC approval is sought.
How do regulatory and patent factors influence pricing and market entry?
Regulatory approvals define the launch timeline. Regulatory agencies in major markets have specific requirements for bioequivalence, stability data, and safety profiles, which can extend development cycles.
Patent protection is critical for premium pricing. Since the original omeprazole patent expired in the early 2000s, new formulations like SM OMEPRAZOLE DR must rely on formulation patents or exclusivity periods. Extended patent life or data exclusivity grants a period of market monopoly, enabling higher pricing and improved margins.
What competitive threats and opportunities exist?
- Generic competition: Well-established generics dominate the market after patent expiry.
- New formulations: Extended-release, combination therapies, or novel delivery systems can gain market share.
- Emerging markets: Growing healthcare infrastructure and GI disorder prevalence create opportunities for early adoption.
Key Takeaways
- The global PPI market is mature, with significant revenue from generic omeprazole.
- SM OMEPRAZOLE DR targets a niche segment with potential premium pricing, especially if patent rights or exclusivity are secured.
- Initial launch prices are estimated between USD 15-35 per month supply, influenced by regional factors.
- Market growth hinges on regulatory approvals, formulary inclusion, and strategic positioning against generics.
- Revenue potential in the first year could reach USD 200 million with sustained growth estimates of 10-12% CAGR over five years.
FAQs
1. What regulatory hurdles does SM OMEPRAZOLE DR face?
It must meet bioequivalence, safety, and efficacy standards set by agencies like the FDA and EMA. Delayed-release formulations require stability and manufacturing process validation.
2. How does patent status impact pricing?
Patent or data exclusivity periods allow for higher pricing by limiting generic competition. Once expired, prices tend to decline sharply.
3. What regions offer the largest market for omeprazole?
North America and Europe have mature markets with high GI disease prevalence. Emerging markets in Asia-Pacific are experiencing growth due to urbanization and lifestyle changes.
4. Can SM OMEPRAZOLE DR command a higher price than generics?
Potentially, especially if positioned as a differentiated formulation with improved tolerability or convenience. Success depends on regulatory approval and market acceptance.
5. What are the risks to market penetration?
Intense competition from established generics, pricing pressures, delayed regulatory approvals, or failure to demonstrate improved efficacy or safety can impede market share gains.
References
- MarketWatch. "Proton Pump Inhibitors (PPIs) Market Analysis." 2022.
- IQVIA. "Global GI Drugs Market Report." 2022.
- U.S. FDA. "Guidance for Industry: Bioequivalence Studies for Orally Administered Drug Products." 2014.
- European Medicines Agency. "Procedures for Marketing Authorization." 2022.
- Statista. "Over-the-counter Drugs Market Revenue." 2022.
More… ↓
