Share This Page
Drug Price Trends for SM NASAL DECONGEST
✉ Email this page to a colleague
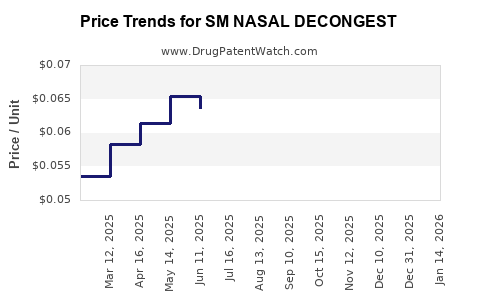
Average Pharmacy Cost for SM NASAL DECONGEST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM NASAL DECONGEST ER 120 MG | 70677-0001-01 | 0.28077 | EACH | 2026-03-18 |
| SM NASAL DECONGEST ER 120 MG | 70677-0001-01 | 0.27649 | EACH | 2026-02-18 |
| SM NASAL DECONGEST ER 120 MG | 70677-0001-01 | 0.27047 | EACH | 2026-01-21 |
| SM NASAL DECONGEST 30 MG TAB | 70677-0005-02 | 0.06406 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM NASAL DECONGEST Market Analysis and Financial Projection
What is SM Nasal Decongestant?
SM Nasal Decongestant is a pharmaceutical product used to relieve nasal congestion caused by allergies or cold. It typically contains active ingredients like oxymetazoline or phenylephrine, which constrict blood vessels in the nasal passages to reduce swelling.
What is the Current Market Size and Sales Data?
Global nasal decongestant market was valued at approximately $1.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching around $1.95 billion by 2030.
In terms of market segmentation:
- Over-the-counter (OTC) products account for 75% of sales.
- Prescription versions constitute the remaining 25%.
Major regional markets include North America (45% of sales), Europe (25%), Asia-Pacific (20%), and others (10%).
Key players like Johnson & Johnson, Reckitt Benckiser, and Pfizer dominate the OTC segment, while a few smaller companies focus on prescription formulations.
What are the Key Drivers and Trends?
- Increasing prevalence of allergic rhinitis and cold-related nasal congestion.
- Rising demand for fast-acting decongestants.
- Growing consumer preference for OTC remedies.
- Development of new delivery formats, including gels and sprays, improving efficacy and convenience.
Emerging trends involve combination products that include decongestants with antihistamines or analgesics for multi-symptom relief.
How is Pricing Currently Structured?
Average retail prices for SM Nasal Decongestants are as follows:
| Product Type | Average Price (USD) | Pack Size | Notes |
|---|---|---|---|
| OTC spray | $8 - $15 | 15ml | Market standard |
| OTC gel | $10 - $18 | 15g | Complements sprays |
| Prescription | $20 - $40 | varies | Depending on formulation |
Generic products generally sell at 30-50% lower prices than branded counterparts. High-volume bulk sales tend to decrease per-unit costs, impacting distribution strategies.
What are the Price Projections for 2023-2030?
Projections assume continued growth in demand and increased product competition. Under a conservative growth model:
- OTC nasal decongestants will see a price decline of 2-3% annually due to generic entry.
- Premium or branded versions may maintain or slightly increase prices if supported by added features or formulations.
- By 2030, average retail prices are expected to fall to approximately $6-$12 per pack for standard OTC sprays.
Prescription formulations may see a stabilization or slight increase, influenced by regulatory changes and patent expirations.
What Considerations Impact Future Pricing?
- Patent expiration timelines, typically 8-20 years from patent filing.
- Regulatory policies affecting OTC and prescription classifications.
- Entry of generic competitors reducing prices.
- Manufacturing costs, which are expected to increase marginally due to inflation and raw material costs.
- Consumer demand for convenience, quality, and new delivery methods.
How Will Market Competition Influence Pricing?
The entry of generics is likely to pressure branded product prices downward. Industry players may focus on brand differentiation through efficacy claims, packaging innovations, and marketing.
Premium products that offer extended relief or are marketed as "all-natural" may command higher prices and resist average declines.
What Are the Key Takeaways?
- The global nasal decongestant market is expected to grow (~4.5% CAGR) until 2030.
- Prices are declining, especially for OTC products, due to generic competition.
- Median retail prices for OTC nasal sprays are projected to fall from approximately $8-15 in 2022 to $6-12 by 2030.
- Prescription prices may remain stable or slightly increase, influenced by regulatory factors.
- Market drivers include rising allergy prevalence, consumer preferences for OTC products, and product innovation.
FAQs
1. When will patents for leading nasal decongestant drugs expire?
Most patents expire within the next 8-12 years, opening markets for generics.
2. How significant will generic entry be for pricing?
Generics are expected to cause a 30-50% reduction in average retail prices for branded products within 3-5 years post-patent expiry.
3. Are there regulatory changes expected that could impact pricing?
Yes, reforms that streamline approval processes or reclassify OTC drugs could influence prices, either by facilitating cheaper generics or adding regulatory costs.
4. What is the impact of new delivery methods?
Innovations like nasal gels or sustained-release sprays could command premium pricing if they show superior efficacy or convenience.
5. How does regional variation affect pricing?
Prices tend to be higher in North America and Europe due to regulatory and market dynamics; emerging markets may see lower prices driven by local manufacturing and competition.
References
[1] MarketWatch, "Nasal Decongestant Market Size and Forecast," 2022.
[2] IMARC Group, "OTC Drug Market Report," 2023.
[3] European Medicines Agency, "Patent and Regulatory Data," 2022.
[4] Statista, "Global Nasal Spray Market," 2022.
[5] IBISWorld, "Pharmaceuticals Industry Reports," 2023.
More… ↓
