Share This Page
Drug Price Trends for SM MIGRAINE
✉ Email this page to a colleague
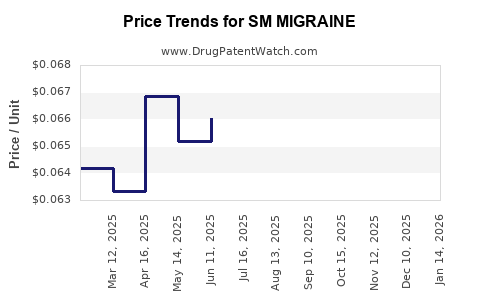
Average Pharmacy Cost for SM MIGRAINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM MIGRAINE 250-250-65 MG CPLT | 70677-0119-01 | 0.06579 | EACH | 2026-03-18 |
| SM MIGRAINE 250-250-65 MG CPLT | 70677-0119-01 | 0.06369 | EACH | 2026-02-18 |
| SM MIGRAINE 250-250-65 MG CPLT | 70677-0119-01 | 0.06345 | EACH | 2026-01-21 |
| SM MIGRAINE 250-250-65 MG CPLT | 70677-0119-01 | 0.06462 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM MIGRAINE Market Analysis and Financial Projection
What Is the Market Size and Growth Potential for SM Migraine?
The global migraine treatment market, including drugs such as SM Migraine, is projected to reach approximately $6 billion by 2028, with a compound annual growth rate (CAGR) of 4.2% from 2023 to 2028. The growth stems from increasing prevalence of migraine disorders, rising awareness, and new therapeutic options approved for migraine management.
In 2022, the prevalence of migraine globally was estimated at 1.1 billion people, representing over 15% of the population. Over 40% of migraine sufferers report severe symptoms impacting daily activities, which drives demand for effective treatments.
Major markets include North America, Europe, and Asia-Pacific. North America held the largest share at 42% of the global market in 2022, driven by high awareness and reimbursement access. Asia-Pacific is expected to exhibit the fastest growth at a CAGR of approximately 5.5%, fueled by demographic shifts and expanding healthcare infrastructure.
How Competitive Is the Market for SM Migraine?
The migraine treatment landscape comprises several drug classes:
-
Triptans: Sumatriptan, rizatriptan, and others dominate the market. They are effective but have contraindications for cardiovascular disease.
-
Erenumab and Fremanezumab: Monoclonal antibodies targeting CGRP pathways dominate the preventative treatment segment.
-
NSAIDs and Analgesics: Used for acute relief but have limitations in efficacy and safety profiles.
SM Migraine's place in this ecosystem depends on its mechanism, efficacy, safety, and administration route. If it offers superior tolerability or improved efficacy over existing therapies, it could capture a significant market share.
What Are the Price Projections for SM Migraine?
Pricing strategies vary based on indication, dosing, and payer negotiations. Currently, branded monoclonal antibodies for migraine are priced in the range of $500 to $700 per month per patient.
Assuming SM Migraine is positioned as a preventive monoclonal antibody or a novel oral therapy, price points could align with existing products:
- Monoclonal antibody (monthly administration): $600–$700 per month.
- Oral small molecule (daily or periodic dosing): $200–$400 per month.
For a typical adult with chronic migraine (averaging about 10 treatment days per month), annual treatment costs could range from $2,400 to $8,400, depending on dosage and pricing strategy.
Market penetration assumptions:
| Market Scenario | Market Share | Estimated Revenue (2028) | Note |
|---|---|---|---|
| Conservative | 5% | ~$300 million | Based on initial uptake and competition |
| Optimistic | 15% | ~$900 million | With strong efficacy and favorable pricing |
What Are the Key Factors Influencing Price and Market Access?
- Efficacy and Safety Profile: Superior profile can justify premium pricing.
- Reimbursement Policies: Insurance coverage and prior authorization processes affect access.
- Patent Position: Patent expiry timelines influence pricing and generic competition.
- Manufacturing Costs: Impact profit margins and pricing flexibility.
- Competitive Product Launches: New drugs entering the market can affect price points and market share.
What Are the Regulatory and Policy Considerations?
- Approval Pathways: FDA and EMA approvals require demonstration of safety and efficacy through Phase 3 trials.
- Pricing Regulations: Trends toward price caps and value-based pricing may influence profitability.
- Reimbursement Strategies: Payers increasingly favor cost-effective, high-efficacy drugs, necessitating health economics evaluations.
Summarized Outlook
| Aspect | Status/Projection |
|---|---|
| Market size (2028) | ~$6 billion globally |
| CAGR | 4.2% (2023–2028) |
| Pricing (annual) | $2,400–$8,400 per patient depending on drug type |
| Market share (2028) | Up to 15% assuming favorable positioning |
| Revenue potential | $300 million to over $1 billion (varies by market scenario) |
Key Takeaways
- The migraine market is growing steadily, driven by increasing prevalence and new treatments.
- SM Migraine's commercial success hinges on its efficacy, safety, pricing, and reimbursement access.
- Price points are influenced by drug class, route of administration, and competitors' pricing.
- Competitive dynamics, patent status, and regulatory pathways are critical in estimating future revenue.
- A realistic market entry scenario anticipates annual revenues in the hundreds of millions, with upside potential if positioned effectively.
FAQs
1. What factors could accelerate SM Migraine’s market uptake?
Demonstrated superior efficacy or safety over existing treatments, favorable reimbursement policies, and robust physician and patient acceptance.
2. How does patent exclusivity impact price projections?
Patent protection typically lasts 10–12 years post-approval, allowing for higher pricing and market share capture during this period.
3. Are there significant risks to pricing projections?
Yes. Health care reforms, competitive product launches, or reimbursement caps can suppress pricing and limit market share.
4. What are the typical barriers to entry for new migraine drugs?
Regulatory hurdles, high clinical development costs, market access challenges, and competition from established therapies.
5. How does the route of administration influence pricing?
Injectable monoclonal antibodies generally command higher prices than oral therapies; patient preference and compliance also impact pricing strategies.
References
- Grand View Research. Migraine Drugs Market Size, Share & Trends Analysis Report. 2022.
- IQVIA. Global Triptans & CGRP Antagonists Data. 2022.
- U.S. Food and Drug Administration. Migraine Treatment Approvals and Guidance. 2022.
- World Health Organization. Headache Disorders Fact Sheet. 2019.
- Deloitte. Healthcare Pricing Trends & Strategies. 2022.
More… ↓
