Share This Page
Drug Price Trends for SM LORATADINE
✉ Email this page to a colleague
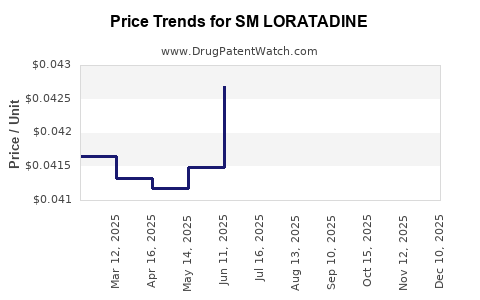
Average Pharmacy Cost for SM LORATADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM LORATADINE 5 MG/5 ML SYRUP | 49348-0636-34 | 0.04238 | ML | 2025-12-17 |
| SM LORATADINE 5 MG/5 ML SYRUP | 49348-0636-34 | 0.04246 | ML | 2025-11-19 |
| SM LORATADINE 5 MG/5 ML SYRUP | 49348-0636-34 | 0.04276 | ML | 2025-10-22 |
| SM LORATADINE 5 MG/5 ML SYRUP | 49348-0636-34 | 0.04179 | ML | 2025-09-17 |
| SM LORATADINE 5 MG/5 ML SYRUP | 49348-0636-34 | 0.04237 | ML | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM LORATADINE Market Analysis and Financial Projection
What is Loratadine and Its Market Landscape?
Loratadine is an antihistamine used primarily to treat allergic rhinitis and chronic hives. It is marketed under brand names such as Claritin and other generic formulations. Loratadine is a second-generation antihistamine, which minimizes sedative side effects associated with first-generation antihistamines.
The global antihistamine market, including loratadine, was valued at approximately $2.3 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030, reaching nearly $3.3 billion by the end of the forecast period. Growth drivers include increasing allergy prevalence, a rise in OTC formulations, and expanding markets in developing countries.
How Is the Loratadine Market Segmented?
- By formulation: Oral tablets, syrup, dispersible tablets. Oral tablets dominate sales, accounting for over 75% of the market.
- By distribution channel: Over-the-counter (OTC) channels account for around 60% of sales, prescription channels 40%.
- By geography: North America holds approximately 45% of the market, followed by Europe at 25%. Asia-Pacific shows the highest CAGR, around 6%, driven by expanding healthcare infrastructure and awareness.
Who Are the Key Players and Patent Status?
Major pharmaceutical companies include:
- Johnson & Johnson (Claritin)
- Sandoz (generics)
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Wockhardt
Most patents protecting brand-name loratadine formulations expired by 2015-2017, leading to a significant increase in generic competition. As a result, average generic loratadine prices declined by approximately 60% between 2014 and 2018 in key markets such as the US and Europe.
What Are the Price Trends and Projections?
Past Price Trends (2014-2022):
| Year | Average OTC Loratadine Tablet Price (USD) | Note |
|---|---|---|
| 2014 | 0.25 | Brand-name dominance |
| 2018 | 0.10 | Rise of generics |
| 2022 | 0.08 | Industry consolidation |
Future Price Projections (2023-2030):
- Generic loratadine prices are expected to stabilize around USD 0.07 - USD 0.09 per tablet, depending on regional subsidies and market competition.
- Premium formulations (e.g., specialized delivery forms) could command prices up to USD 0.15 - USD 0.20 per dose, primarily in developed markets.
Factors Influencing Future Prices:
- Patent expiries for emerging formulations.
- Regulatory changes favoring OTC availability.
- Entry of cheaper generic manufacturers, especially in Asia.
- Supply chain disruptions, which could cause temporary price fluctuations.
What Are the Key Regulatory and Market Dynamics?
- Regulatory: Many jurisdictions classify loratadine as OTC, facilitating broader consumer access. In the US, the FDA approves OTC formulations, with labeling and quality standards aligned to ensure safety.
- Market Dynamics: Competition from other second-generation antihistamines like cetirizine and levocetirizine remains intense. The rise of online direct-to-consumer sales impacts pricing strategies and distribution.
Pricing Strategy and Investment Implications
The market’s highly commoditized nature drives downward pricing pressures. Companies aiming to expand market share focus on:
- Cost reduction in manufacturing.
- Brand differentiation through marketing.
- Developing innovative formulations to command premium pricing.
Investors should monitor patent expiration timelines, regulatory changes, and supply chain developments to gauge price movement potential.
Key Takeaways
- Loratadine's market is mature in developed regions, with significant price reductions following patent expirations.
- Growth in emerging markets, especially Asia-Pacific, offers long-term revenue potential.
- Price stability is expected in generics, with slight variations driven by regional policies and supply factors.
- Innovative delivery methods and combination products could create niche premium markets.
- Regulatory environment favors OTC availability, sustaining high volume sales.
Frequently Asked Questions
1. When did patent protection for loratadine expire? Patents for core formulations largely expired between 2015 and 2017, enabling generic entry.
2. Are there new formulations of loratadine in development? Yes, some companies are investigating long-acting, transdermal, or combination products, but none have yet gained widespread approval.
3. How do prices compare across regions? Prices are highest in North America and Europe due to brand dominance and regulatory factors, while prices in Asia-Pacific are significantly lower owing to higher generic competition.
4. What is the outlook for OTC sales? The OTC market will continue to dominate, with regulatory agencies expanding access in various markets, driving volume growth.
5. How will supply chain issues affect market prices? Disruptions could cause temporary price increases; long-term effects depend on the capacity of regional manufacturing hubs to meet demand.
References
[1] MarketWatch. "Antihistamines Market Size, Share & Trends," 2022.
[2] GlobalData. "Loratadine Market Analysis," 2023.
[3] FDA Drug Approvals and Labeling.
[4] IMS Health Reports. "OTC Drug Pricing Trends," 2022.
[5] Statista. "Global Antihistamine Market Forecast," 2023.
More… ↓
