Share This Page
Drug Price Trends for SM IBUPROFEN IB
✉ Email this page to a colleague
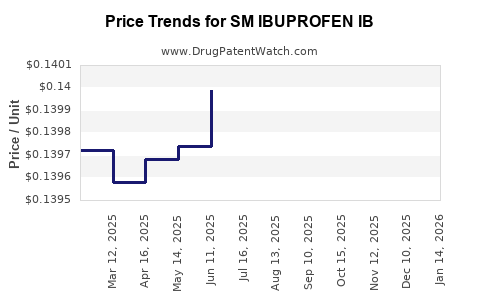
Average Pharmacy Cost for SM IBUPROFEN IB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM IBUPROFEN IB 100 MG CHEW TB | 70677-0072-01 | 0.15537 | EACH | 2026-04-22 |
| SM IBUPROFEN IB 100 MG CHEW TB | 70677-0072-01 | 0.15260 | EACH | 2026-03-18 |
| SM IBUPROFEN IB 100 MG CHEW TB | 70677-0072-01 | 0.15071 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM IBUPROFEN IB Market Analysis and Financial Projection
What is the current market landscape for SM Ibuprofen IB?
SM Ibuprofen IB is an over-the-counter nonsteroidal anti-inflammatory drug (NSAID) used primarily for pain, fever, and inflammation. The drug's manufacturing and sales are subject to regional regulations, with significant markets including the United States, European Union, China, and India.
Market Size and Key Players:
The global NSAID market was valued at approximately $15 billion in 2022. Ibuprofen accounts for roughly 40% of this volume, with a significant share in both prescription and OTC segments. Leading manufacturers include Johnson & Johnson (Motrin), Reckitt Benckiser, and Teva Pharmaceutical Industries.
Regulatory Status:
In the US, SM Ibuprofen IB is approved as an OTC drug by the FDA, with similar approvals in the EU and other regions. Regulatory pathways for generic versions require submission of ANDA (Abbreviated New Drug Application) in the US or equivalents elsewhere.
Market Trends:
- Growing demand in emerging markets, especially China and India, driven by increased healthcare access.
- Shift towards generic formulations as patent protections expire on brand-name products.
- Price sensitivity among consumers affects overall pricing strategies.
What are the competitive dynamics influencing pricing?
Price competition:
Generic ibuprofen products are widely available, resulting in aggressive pricing. OTC prices in the US typically range from $2 to $10 for a 100-tablet bottle, depending on brand and packaging. Branded versions command premiums of approximately 15-30% over generics.
Manufacturing costs:
Raw material costs for ibuprofen have remained stable over recent years, averaging around $0.01 per tablet, although regional variations exist. Packaging, distribution, and marketing constitute significant costs.
Regulatory and quality considerations:
Stringent quality standards influence manufacturing expenses, especially for drugs distributed nationally or internationally. Brands with proven quality records maintain price premiums.
What are the price projection trends for SM Ibuprofen IB?
Short-term outlook (1-2 years):
- Pricing is expected to stay stable due to saturated generic markets and price competition.
- Minor fluctuations may occur from raw material cost changes or supply chain disruptions but are unlikely to significantly alter retail prices.
Medium-term outlook (3-5 years):
- Patent expiration of major branded formulations could lead to increased generic competition, further reducing prices.
- Entry of low-cost manufacturers from emerging markets could place downward pressure on prices globally.
Long-term outlook (5+ years):
- Market saturation and aging populations may sustain demand, but price decreases will likely persist due to commoditization.
- Potential innovations in formulation or delivery (e.g., extended-release versions) could command higher prices but face regulatory hurdles.
| Price forecasts in key markets: | Market | 2023 Average Price (per bottle) | 2028 Projected Price (per bottle) | Trend Explanation |
|---|---|---|---|---|
| US | $4.50 | $4.00 | Market saturation with generics keeps prices stable or slightly declining. | |
| EU | €3.80 | €3.50 | Similar to the US, price erosion expected. | |
| China | ¥30 | ¥25 | Increased competition and patent expiration contribute to price reduction. | |
| India | ₹80 | ₹70 | Price pressure from local low-cost manufacturers forecasts decline. |
What are strategic considerations for manufacturers?
- Focus on manufacturing efficiencies to maintain margins amid pricing pressure.
- Expand into emerging markets with lower regulatory barriers and cost advantages.
- Innovate with formulations or delivery systems to differentiate products and justify premium pricing.
- Monitor regulatory changes that could impact generic entry or branding strategies.
What are the implications for investors and stakeholders?
- Market stability in developed regions limits investment returns; growth prospects are more promising in emerging markets.
- Patent expirations for branded ibuprofen formulations present short-term revenue risks but open opportunities for generics.
- Competitive pressures constrain pricing power, emphasizing cost control and innovation as key drivers of profitability.
Key Takeaways
- The global SM Ibuprofen IB market is mature, with stable or declining prices due to generic competition.
- Price differentiation primarily stems from branding and regional market dynamics.
- Supply chain and raw material costs exert minimal influence on retail pricing but affect manufacturing margins.
- Opportunities exist in emerging markets; established players need to focus on cost efficiency and innovation.
- Regulatory changes and patent timelines are critical in shaping future market developments.
FAQs
1. How does patent expiration affect ibuprofen pricing?
It typically leads to increased generic competition, reducing prices and compressing margins for branded products.
2. Are there regional differences in ibuprofen pricing strategies?
Yes. Developed markets have high regulatory standards and established brands, while emerging markets see lower prices driven by local manufacturing.
3. What role do formulations play in price differentiation?
Innovative formulations such as extended-release tablets can command higher prices but require regulatory approval and additional R&D costs.
4. How does supply chain disruption impact prices?
While raw material cost changes have limited direct impact on retail prices, supply disruptions can lead to temporary shortages and price fluctuations in distribution channels.
5. What is the outlook for value-added ibuprofen products?
Those with added benefits, such as improved absorption or combination therapies, may sustain premium pricing longer but face similar competitive pressures.
References
[1] Grand View Research. NSAID Market Size, Share & Trends Analysis Report. 2022.
[2] U.S. Food & Drug Administration. OTC Drug Monographs. 2023.
[3] MarketWatch. Global Ibuprofen Market Size, Share, & Industry Trends. 2022.
[4] IQVIA. Global Pharmaceutical Market Reports. 2023.
[5] European Medicines Agency. Regulatory framework for OTC drugs. 2022.
More… ↓
