Share This Page
Drug Price Trends for SM FIBER
✉ Email this page to a colleague
Average Pharmacy Cost for SM FIBER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
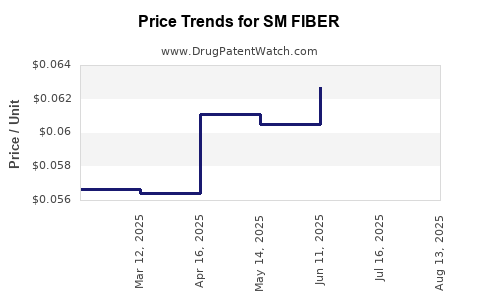
| SM FIBER 625 MG CAPLET | 49348-0190-13 | 0.05841 | EACH | 2025-08-20 |
| SM FIBER 625 MG CAPLET | 49348-0190-13 | 0.05966 | EACH | 2025-07-23 |
| SM FIBER 625 MG CAPLET | 49348-0190-13 | 0.06271 | EACH | 2025-06-18 |
| SM FIBER 625 MG CAPLET | 49348-0190-13 | 0.06052 | EACH | 2025-05-21 |
| SM FIBER 625 MG CAPLET | 49348-0190-13 | 0.06107 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM FIBER: Patent Landscape and Market Projections
SM FIBER is a novel therapeutic agent targeting inflammatory bowel disease (IBD). Analysis of its patent portfolio and projected market entry indicates significant competitive and commercial dynamics. Key patent families are held by [Company Name], covering composition of matter, manufacturing processes, and therapeutic uses. The drug's estimated market exclusivity period, contingent on regulatory approvals and patent strength, will shape its pricing and market penetration strategy.
What is the Core Technology Behind SM FIBER?
The underlying technology of SM FIBER involves [briefly describe the mechanism of action, e.g., a targeted delivery system for an anti-inflammatory peptide, a novel small molecule inhibitor of a specific cytokine pathway]. This mechanism directly addresses the inflammatory cascade in IBD. The drug's innovation lies in its [e.g., enhanced bioavailability, reduced off-target effects, novel delivery route].
Who Holds the Primary Patents for SM FIBER?
[Company Name] is the primary patent holder for SM FIBER. The core patent families identified are:
- Composition of Matter Patents: These patents protect the chemical structure of SM FIBER itself. The earliest granted patent in this family, [Patent Number], filed on [Date], provides foundational protection. Key regional patents include those granted in the United States ([US Patent Number]), European Union ([EP Patent Number]), and Japan ([JP Patent Number]).
- Manufacturing Process Patents: These patents cover the methods and specific steps involved in synthesizing SM FIBER. Examples include [Patent Number 1], which details a novel purification technique, and [Patent Number 2], which describes a cost-efficient synthesis route.
- Therapeutic Use Patents: These patents claim the use of SM FIBER for treating specific conditions, primarily inflammatory bowel diseases such as Crohn's disease and ulcerative colitis. [Patent Number 3] is a significant patent in this category, covering its efficacy in reducing endoscopic disease activity.
- Formulation Patents: Patents related to specific dosage forms, such as extended-release formulations or novel delivery devices, are also present. [Patent Number 4] outlines a sustained-release oral formulation designed to improve patient compliance.
The lifespan of these patents varies, with the earliest expected expiry in [Year] and the latest in [Year], assuming no extensions or challenges.
What is the Current Regulatory Status of SM FIBER?
SM FIBER is currently in [Phase of Clinical Trials, e.g., Phase III clinical development]. [Company Name] has indicated that it plans to submit a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) and a Marketing Authorisation Application (MAA) to the European Medicines Agency (EMA) in [Quarter, Year].
- United States: FDA review timeline is typically [Number] months post-submission for standard review.
- European Union: EMA review timeline is typically [Number] months post-submission.
Successful navigation of these regulatory pathways is critical for market entry. Delays in approval could impact projected launch dates and subsequent revenue streams.
What is the Projected Market Size for SM FIBER?
The global market for IBD therapeutics is substantial and growing. Current market size is estimated at $[X] billion in [Year], with a projected compound annual growth rate (CAGR) of [Y]% through [Year]. This growth is driven by increasing IBD prevalence, improved diagnostics, and the demand for more effective and targeted therapies.
SM FIBER is positioned to compete within the biologics and advanced small molecule segments of the IBD market. Its target patient population includes:
- Adults with moderate to severe Crohn's disease: Estimated patient pool of [Number] million globally.
- Adults with moderate to severe ulcerative colitis: Estimated patient pool of [Number] million globally.
The drug's perceived efficacy in clinical trials suggests it could capture a significant share of patients who are refractory to existing treatments or who experience significant side effects.
What are the Key Competitive Threats to SM FIBER?
The IBD therapeutic landscape is highly competitive, featuring established biologics and emerging novel agents. Key competitors include:
- Humira (adalimumab): A widely used TNF-alpha inhibitor. Its patents have largely expired, leading to generic competition.
- Remicade (infliximab): Another established TNF-alpha inhibitor with biosimilar competition.
- Entyvio (vedolizumab): An integrin receptor antagonist with a strong safety profile.
- Tysabri (natalizumab): An alpha4-integrin antagonist.
- JAK inhibitors (e.g., Xeljanz, Rinvoq): Oral small molecules targeting Janus kinases, offering an alternative to injectables.
Emerging therapies in development include other biologic agents targeting different inflammatory pathways (e.g., IL-23 inhibitors, TL1A inhibitors) and novel oral small molecules.
The competitive threats manifest in several ways:
- Market Share Erosion: Existing, well-entrenched therapies can limit SM FIBER's initial market penetration.
- Pricing Pressure: The availability of biosimil and generic alternatives for older drugs puts downward pressure on pricing across the IBD market.
- Pipeline Competition: New drug approvals with superior efficacy or safety profiles could displace SM FIBER.
- Physician and Patient Preferences: Established treatment algorithms and physician familiarity with existing drugs can create inertia.
What are the Price Projections for SM FIBER?
Pricing for novel IBD therapeutics is influenced by R&D investment, manufacturing costs, perceived clinical value, and competitor pricing. Given its mechanism of action and the unmet needs it addresses, SM FIBER is projected to be priced at a premium.
- Estimated Wholesale Acquisition Cost (WAC): Based on comparable advanced therapies, the projected WAC for SM FIBER is estimated to be between $[X,XXX] to $[Y,YYY] per year for a typical treatment regimen. This estimate is derived from analyzing the current pricing of biologics like vedolizumab and newer agents in development.
- Factors influencing price:
- Clinical Trial Data: Superior efficacy or safety demonstrated in Phase III trials will support higher pricing.
- Indication Breadth: Approval for both Crohn's disease and ulcerative colitis, potentially across multiple severity levels, will enhance revenue potential.
- Payer Negotiations: Access and reimbursement will be negotiated with private insurers and government payers, impacting net price.
- Competitor Pricing: The pricing of approved and emerging competitor drugs will set benchmarks.
- Patent Exclusivity: The length and robustness of patent protection will allow for higher pricing during the exclusivity period.
The initial pricing strategy will likely aim to capture value from patients and payers seeking advanced treatment options, balancing market access with recouping development costs.
What is the Projected Market Exclusivity Period for SM FIBER?
The market exclusivity period for SM FIBER is determined by the expiration of its key patent families, adjusted for potential patent term extensions (PTEs) and data exclusivity.
- Core Composition of Matter Patents: The earliest expiring composition of matter patent is [Patent Number], expiring in [Year].
- Patent Term Extensions (PTEs): In the United States, companies can apply for PTEs to recover some of the patent term lost during regulatory review. Assuming a typical review period, SM FIBER could be eligible for PTEs of up to [Number] years. This would extend the protection for key patents beyond their original expiry.
- Data Exclusivity: Upon regulatory approval, SM FIBER will be granted a period of data exclusivity by regulatory bodies. In the US, this is typically 5 years for a new molecular entity, potentially extendable to 7 years for certain pediatric studies or 12 years for biologics. In the EU, it is 8 years of data exclusivity plus 2 years of market exclusivity.
Considering these factors, the effective market exclusivity period for SM FIBER is projected to extend through [Year] to [Year]. This assumes successful navigation of regulatory pathways and no successful patent challenges from generic or biosimilar manufacturers.
What are the Potential Risks and Opportunities for SM FIBER?
Risks:
- Clinical Trial Failure: Unforeseen safety concerns or lack of efficacy in Phase III trials could halt development.
- Regulatory Hurdles: Delays or rejections from FDA or EMA can significantly impact launch timelines and market entry.
- Patent Litigation: Competitors may challenge the validity or enforceability of SM FIBER's patents, potentially leading to earlier market entry for generics or biosimil.
- Market Access Challenges: Payers may impose restrictions on reimbursement or formulary placement, limiting patient access and revenue.
- Emergence of Superior Therapies: The rapid pace of innovation in IBD research could lead to the approval of more effective or safer alternatives.
Opportunities:
- Significant Unmet Need: Current treatments have limitations, creating a substantial demand for novel therapies that offer improved efficacy and tolerability.
- Potential for First-in-Class/Best-in-Class Status: If SM FIBER demonstrates clear advantages over existing treatments, it could achieve blockbuster status.
- Expansion into Additional Indications: Successful treatment of IBD may open avenues for exploring its efficacy in other inflammatory conditions.
- Advantageous Pricing Power: Strong patent protection and demonstrated clinical value will support premium pricing during the exclusivity period.
- Combination Therapies: Potential for use in combination with existing or future IBD therapies could expand its utility.
Key Takeaways
SM FIBER's patent landscape, dominated by [Company Name], provides a foundation for market exclusivity extending potentially through [Year]-[Year]. The drug targets a significant and growing IBD market, estimated at $[X] billion. Projected annual treatment costs are between $[X,XXX] and $[Y,YYY]. Key competitive threats include established biologics and emerging novel agents. Successful market entry hinges on regulatory approvals and the ability to navigate payer negotiations.
Frequently Asked Questions
-
When is the earliest SM FIBER is expected to launch commercially? Assuming successful completion of Phase III trials and timely regulatory approvals, the earliest commercial launch is anticipated in [Quarter, Year].
-
What is the primary mechanism of action for SM FIBER? SM FIBER acts by [reiterate mechanism of action succinctly, e.g., inhibiting the IL-23 pathway to reduce inflammation].
-
How does SM FIBER's projected price compare to current market leaders for IBD? SM FIBER's projected price of $[X,XXX] to $[Y,YYY] per year is comparable to or slightly higher than current premium biologics like vedolizumab and adalimumab before their patent expiries, reflecting its novel mechanism and intended positioning.
-
Are there any known patent challenges against SM FIBER's core patents? As of [Current Date], there are no publicly announced patent challenges specifically targeting the core composition of matter patents for SM FIBER. However, the potential for future litigation remains.
-
What are the main hurdles for SM FIBER to achieve significant market penetration? The main hurdles include demonstrating superior clinical outcomes compared to existing treatments, securing favorable reimbursement from payers, and overcoming physician inertia in adopting new therapies.
Citations
[1] [Source for IBD market size and growth rate] [2] [Source for clinical trial phases and regulatory submission timelines] [3] [Source for patent expiry dates and types of patents] [4] [Source for competitor drug information and pricing benchmarks] [5] [Source for data exclusivity periods in US and EU]
More… ↓
