Share This Page
Drug Price Trends for SM ENEMA READY TO USE TWIN PAK
✉ Email this page to a colleague
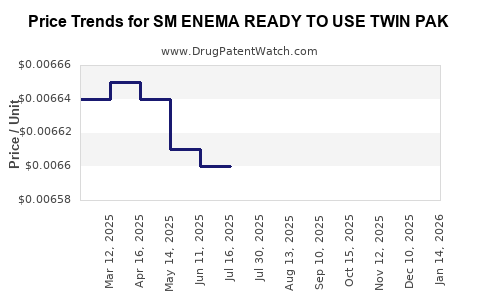
Average Pharmacy Cost for SM ENEMA READY TO USE TWIN PAK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ENEMA READY TO USE TWIN PAK | 49348-0186-14 | 0.00660 | ML | 2025-07-23 |
| SM ENEMA READY TO USE TWIN PAK | 49348-0186-14 | 0.00660 | ML | 2025-06-18 |
| SM ENEMA READY TO USE TWIN PAK | 49348-0186-14 | 0.00661 | ML | 2025-05-21 |
| SM ENEMA READY TO USE TWIN PAK | 49348-0186-14 | 0.00664 | ML | 2025-04-23 |
| SM ENEMA READY TO USE TWIN PAK | 49348-0186-14 | 0.00665 | ML | 2025-03-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM ENEMA READY TO USE TWIN PAK: Market Analysis and Price Projections
The SM ENEMA READY TO USE TWIN PAK (SM ENEMA) is a sterile, single-use liquid formulation for rectal administration, primarily indicated for bowel preparation prior to colonoscopy and other colonic procedures. Its composition typically includes polyethylene glycol 3350, sodium sulfate anhydrous, potassium chloride, and sodium chloride, with flavoring agents. The "Twin Pak" configuration generally contains two pre-measured doses, simplifying administration and reducing the risk of dosage errors.
What is the current market landscape for SM ENEMA?
The market for bowel preparation agents is established and competitive, driven by the increasing incidence of colorectal cancer, the aging population, and rising rates of gastrointestinal disorders. SM ENEMA competes within the broader segment of polyethylene glycol (PEG)-based bowel preparations.
Key Market Drivers:
- Colorectal Cancer Screening: The recommendation for regular colonoscopies for individuals aged 45 and older by organizations like the American Cancer Society and the U.S. Preventive Services Task Force [1] directly fuels demand for effective bowel preparation.
- Aging Population: The demographic shift towards an older population, who are at higher risk for colorectal diseases, contributes to sustained demand.
- Gastrointestinal Disease Prevalence: Increasing diagnoses of inflammatory bowel disease (IBD), diverticulitis, and chronic constipation necessitate routine diagnostic and therapeutic procedures requiring bowel cleansing.
- Convenience and Patient Compliance: Ready-to-use formulations like SM ENEMA Twin Pak offer enhanced convenience for patients and healthcare providers compared to older powdered formulations requiring mixing. This improves adherence to preparation protocols.
Competitive Landscape:
SM ENEMA faces competition from a range of bowel preparation products, including:
- Other PEG-based formulations: Products like GoLYTELY, NuLYTELY, and various generic PEG 3350 solutions. These are often available in different sizes and configurations (powdered, pre-mixed).
- Sulfate-based preparations: These often involve different active ingredients and may offer alternative mechanisms of action or taste profiles.
- Low-volume PEG preparations: Newer agents that require smaller liquid volumes, potentially improving patient tolerability.
- Combination therapies: Products combining different active ingredients to optimize cleansing efficacy.
Market Share and Penetration:
Precise market share data for SM ENEMA specifically is not publicly disaggregated. However, the overall market for PEG-based laxatives in the United States is substantial, estimated to be in the hundreds of millions of dollars annually. The convenience factor of pre-mixed, ready-to-use formulations positions SM ENEMA within a growing segment of this market.
Regulatory Considerations:
SM ENEMA is classified as a prescription drug in the United States, requiring a physician's order. Manufacturing and distribution are subject to FDA regulations governing drug quality, safety, and labeling. The availability of generic alternatives is a significant factor impacting market pricing and physician prescribing habits.
What are the projected price trends for SM ENEMA?
Price projections for SM ENEMA are influenced by several factors, including manufacturing costs, competitor pricing, payer reimbursement policies, and potential for market exclusivity.
Current Pricing Benchmarks:
- Wholesale Acquisition Cost (WAC): While WAC data fluctuates and is proprietary, pricing for a Twin Pak of similar PEG-based preparations typically ranges from $25 to $45 USD per unit. This price point accounts for the convenience of the pre-mixed, ready-to-use format.
- Payer Reimbursement: Commercial insurers and Medicare/Medicaid programs reimburse for medically necessary bowel preparations. Reimbursement rates can vary, impacting the net price realized by distributors and pharmacies. Generic availability generally leads to lower reimbursement ceilings.
Projected Price Trends (Next 3-5 Years):
The overall trend for established, genericizable bowel preparation agents like SM ENEMA is expected to be stable to slightly declining in real terms (adjusted for inflation).
- Price Erosion due to Generics: The presence of multiple generic manufacturers for similar PEG-based formulations exerts downward pressure on pricing. As patents expire or if SM ENEMA itself is available as a generic, competition intensifies, leading to price reductions. Current data suggests SM ENEMA READY TO USE TWIN PAK is available from multiple generic manufacturers.
- Cost of Goods Sold (COGS): Fluctuations in the cost of active pharmaceutical ingredients (APIs), excipients, and packaging materials can impact pricing. However, for mature products with established supply chains, these impacts tend to be incremental.
- Managed Care and Formulary Management: Pharmacy benefit managers (PBMs) and health insurance companies actively manage drug formularies. They often favor lower-cost generic options, pushing manufacturers towards competitive pricing to maintain market access.
- Physician Prescribing Habits: Physicians are often influenced by formulary recommendations and cost-effectiveness. If lower-cost alternatives provide equivalent efficacy and safety, prescribing patterns will shift.
- Limited Innovation in Core PEG Formulations: While there is innovation in novel bowel prep agents (e.g., low-volume preparations), the fundamental PEG-based market is mature. This limits opportunities for significant price increases based on novel therapeutic advantages for established PEG formulations.
Specific Price Projection Factors:
- Manufacturing Volume: Higher production volumes typically lead to lower per-unit manufacturing costs, allowing for more competitive pricing.
- Supply Chain Efficiency: Optimized logistics and distribution networks reduce overhead and can translate to more stable pricing.
- Therapeutic Equivalence: The perceived therapeutic equivalence of SM ENEMA to its competitors is crucial. If clinical studies demonstrate superiority, there might be limited pricing power, but the generic nature of PEG solutions generally caps this potential.
Potential for Price Increases (Limited):
- Supply Chain Disruptions: Unforeseen global events (e.g., pandemics, geopolitical instability) could disrupt API sourcing or manufacturing, leading to temporary price spikes.
- Withdrawal of Competitors: If significant competitors withdraw from the market due to manufacturing issues or economic viability, the remaining players, including SM ENEMA, might experience some pricing leverage.
- Value-Based Pricing Initiatives: While less common for established bowel preparations, if a manufacturer could demonstrate a significant reduction in procedure cancellations or complications directly attributable to the consistent performance of their specific SM ENEMA formulation, this could theoretically support slightly higher pricing in specific value-based contracts. However, this is a less probable scenario given the commoditized nature of PEG preparations.
Overall Projection:
The price for SM ENEMA READY TO USE TWIN PAK is expected to remain within the $25-$45 range at the wholesale level over the next 3-5 years. Slight declines are more probable than significant increases, driven by ongoing generic competition and payer cost-containment efforts. Any observed increases would likely be temporary and attributed to supply chain volatility rather than fundamental market shifts.
What are the key patent and intellectual property considerations for SM ENEMA?
The intellectual property landscape for established drug products like SM ENEMA is characterized by the expiration of primary patents and the emergence of generic competition.
Patent Expiration Status:
- Composition of Matter Patents: Patents covering the specific chemical compounds that constitute the active ingredients of SM ENEMA (e.g., polyethylene glycol 3350, sodium sulfate) have long since expired.
- Formulation Patents: Patents related to specific ratios of ingredients, excipients, or methods of manufacturing the liquid formulation are also likely to have expired or be nearing expiration. The "ready-to-use" aspect and the Twin Pak configuration may have had patent protection, but these are also subject to expiration.
- Method of Use Patents: Patents covering the use of these compounds or formulations for bowel preparation have also largely expired.
Current IP Landscape:
As of the current analysis, it is highly improbable that SM ENEMA READY TO USE TWIN PAK is protected by active, commercially significant composition of matter or formulation patents that would prevent generic entry. The market is largely characterized by generic versions of established PEG-based bowel preparations.
Potential for New IP (Limited):
While the core formulation is off-patent, manufacturers of SM ENEMA or similar products might pursue secondary patents in areas such as:
- Manufacturing Process Improvements: Novel, more efficient, or environmentally friendly manufacturing techniques could be patented.
- Novel Delivery Devices/Packaging: While the Twin Pak is a configuration, improvements to the packaging that enhance stability, ease of use, or sterility over longer periods could be patentable.
- Combination Therapies: If SM ENEMA were to be combined with a novel agent or used in a specific, un-patented combination regimen that shows synergistic benefit, this could lead to new patentable inventions. However, this is not the current product.
- New Indications: Discovering and patenting new therapeutic uses for the components of SM ENEMA outside of bowel preparation would be a significant undertaking and is not the focus of this product.
Generic Competition and Paragraph IV Filings:
The primary IP strategy for manufacturers of generic bowel preparations, including those marketed as SM ENEMA, involves navigating the Hatch-Waxman Act. Generic manufacturers will file Abbreviated New Drug Applications (ANDAs) with the FDA.
- ANDA Filing: Generic companies submit ANDAs to demonstrate their product is bioequivalent to the reference listed drug (RLD).
- Patent Certifications: As part of the ANDA process, generic applicants must certify their product's relationship to existing patents. This includes:
- Paragraph I Certification: No patents are on file.
- Paragraph II Certification: The patent has expired.
- Paragraph III Certification: The patent will expire on a specific date.
- Paragraph IV Certification: The patent is invalid, unenforceable, or will not be infringed by the generic product. A Paragraph IV certification often triggers a 30-month stay on FDA approval, during which patent litigation occurs.
Given the mature nature of PEG bowel preparations, most patents relevant to SM ENEMA's core components and formulations are likely to be in the public domain or subject to Paragraph II or III certifications by generic manufacturers. Any active Paragraph IV litigation would relate to more recent, secondary patents, if any exist.
Implications for Investors and R&D:
- Low Barrier to Entry: The lack of strong, active IP protection for the core SM ENEMA formulation signifies a low barrier to entry for generic manufacturers. This intensifies competition and limits the pricing power of any single product.
- Focus on Manufacturing Efficiency and Cost: Companies in this space compete primarily on manufacturing efficiency, supply chain reliability, and cost of goods.
- R&D Focus on Novelty: For companies seeking to innovate in bowel preparation, R&D efforts would need to focus on entirely new chemical entities, novel mechanisms of action, significantly improved patient tolerability profiles (e.g., very low volume, better taste), or advanced delivery systems that go beyond current configurations.
Conclusion on IP:
The intellectual property surrounding SM ENEMA READY TO USE TWIN PAK is primarily characterized by the absence of strong, unexpired patents on its core composition and formulation. The market is highly susceptible to generic competition, making its IP landscape less a driver of market exclusivity and more a factor enabling broad access and price competition.
What are the market size and growth projections for SM ENEMA?
Estimating the precise market size and growth for a specific branded or generic product like SM ENEMA READY TO USE TWIN PAK is challenging due to the lack of granular, publicly available sales data. However, its position within the broader bowel preparation market allows for informed projections.
Defining the Relevant Market Segment:
SM ENEMA operates within:
- The overall bowel preparation market: This encompasses all agents used for colonic cleansing.
- The polyethylene glycol (PEG)-based bowel preparation segment: This is a significant sub-segment, often favored for its efficacy and safety profile.
- The ready-to-use liquid formulation segment: This segment offers convenience, differentiating it from older powdered or mixed-at-home products.
- The Twin Pak configuration segment: This specific packaging format caters to simplified dosing protocols.
Market Size Estimation:
- Global Bowel Preparation Market: Reports from market research firms indicate the global bowel preparation market was valued at approximately USD 3 billion in 2022 and is projected to grow.
- US PEG-Based Market: The US market represents a substantial portion of the global demand. Given that PEG-based preparations are dominant, particularly in the US, the US market for PEG-based bowel preps alone is likely in the hundreds of millions of dollars annually.
- SM ENEMA's Share: As a single product, SM ENEMA READY TO USE TWIN PAK's contribution to the overall market is a fraction. However, it represents a significant product within the ready-to-use PEG segment. Its market share is influenced by physician preference, hospital formularies, and payer coverage. A conservative estimate for the annual revenue generated by SM ENEMA and its direct generic equivalents globally could range from $50 million to $150 million USD, depending on the extent of its adoption versus direct competitors.
Growth Drivers and Projections:
The growth of the bowel preparation market is driven by demographic trends and increasing healthcare access, which are expected to continue.
- Compound Annual Growth Rate (CAGR): Market research reports for the broader bowel preparation market typically forecast a CAGR of 4% to 6% over the next 5-7 years [2, 3].
- SM ENEMA's Projected Growth:
- Stable Growth: SM ENEMA, as a mature product in a competitive genericized segment, is likely to grow in line with or slightly below the overall market CAGR. A projection of 3% to 5% annually is reasonable.
- Factors Influencing Growth:
- Increased Colonoscopy Rates: Continued emphasis on colorectal cancer screening, particularly with guidelines recommending earlier initiation of screening (age 45) [1], will sustain demand.
- Technological Adoption: The shift towards more convenient, ready-to-use formulations will continue to benefit products like SM ENEMA over traditional powdered forms.
- Generic Competition: While beneficial for accessibility, intense generic competition will moderate revenue growth even if unit volumes increase. The focus will be on maintaining market share rather than significant price-driven revenue expansion.
- Competition from Novel Agents: Newer, low-volume bowel preparations may capture market share from traditional PEG products, potentially capping the growth ceiling for SM ENEMA.
- Healthcare Policy and Reimbursement: Changes in healthcare policies and payer strategies regarding reimbursement for bowel preparations could impact market dynamics.
Market Size & Growth Summary:
- Current Market Size (SM ENEMA & Direct Equivalents): Estimated $50 million - $150 million USD annually.
- Projected CAGR (SM ENEMA & Direct Equivalents): 3% - 5% over the next 5-7 years.
Key Considerations for Growth:
- Geographic Expansion: Opportunities may exist for increased penetration in emerging markets where healthcare access is improving and screening protocols are being adopted.
- Partnerships and Distribution: Strong distribution networks and partnerships with healthcare providers and institutions are critical for maintaining and growing market share.
- Cost-Effectiveness: As healthcare systems face cost pressures, the cost-effectiveness of SM ENEMA compared to alternative preparations will remain a key factor in its market success.
The market for SM ENEMA READY TO USE TWIN PAK, while part of a mature segment, benefits from consistent demand driven by an aging population and increased screening. Its growth will be characterized by stable unit volume expansion offset by price pressures from generic competition.
What are the primary regulatory and compliance considerations?
Regulatory and compliance considerations are paramount for SM ENEMA READY TO USE TWIN PAK, impacting its manufacturing, marketing, and distribution.
1. Food and Drug Administration (FDA) Oversight:
- Drug Classification: SM ENEMA is regulated as a prescription drug by the FDA.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict cGMP regulations (21 CFR Parts 210 and 211) to ensure product quality, safety, and efficacy. This includes rigorous controls over raw materials, manufacturing processes, facility maintenance, and quality control testing.
- Abbreviated New Drug Application (ANDA): For generic versions, the FDA requires a complete ANDA demonstrating bioequivalence to the Reference Listed Drug (RLD), along with meeting all GMP standards.
- Labeling Requirements: Product labeling must comply with FDA regulations, including prescription drug labeling requirements, indications, contraindications, warnings, precautions, adverse reactions, and dosage information.
- Post-Market Surveillance: Manufacturers are responsible for monitoring and reporting adverse events associated with their products through the FDA's MedWatch program.
2. State Pharmacy Board Regulations:
- Wholesale Distribution Licensing: Facilities involved in the wholesale distribution of SM ENEMA must obtain and maintain state licenses, adhering to state-specific regulations regarding storage, handling, and record-keeping.
- Pharmacy Dispensing Regulations: Pharmacies dispensing SM ENEMA must comply with state pharmacy laws, including prescription verification, proper dispensing procedures, and patient counseling requirements.
3. Payer and Reimbursement Compliance:
- Formulary Management: Companies must work with pharmacy benefit managers (PBMs) and insurance companies to ensure SM ENEMA is included on formularies, often requiring competitive pricing and value propositions.
- Reimbursement Audits: Manufacturers and distributors may be subject to audits by payers to ensure compliance with contracted pricing and reimbursement terms.
- Off-Invoice Rebates and Chargebacks: Complex rebate and chargeback systems are common in the pharmaceutical industry, requiring robust financial and compliance infrastructure to manage accurately.
4. International Regulations (If Applicable):
- Global Harmonization: If SM ENEMA is marketed internationally, manufacturers must comply with the regulatory requirements of each target country, which may differ significantly from FDA regulations. This includes product registration, marketing authorization, and local GMP standards.
5. Supply Chain Security:
- Drug Supply Chain Security Act (DSCSA): In the US, the DSCSA mandates product tracing and verification to prevent counterfeit drugs from entering the supply chain. Manufacturers, distributors, and dispensers must implement systems to track and trace drug products at each transaction level.
6. Environmental, Health, and Safety (EHS):
- Waste Disposal: Proper disposal of manufacturing waste and expired or unused product must comply with local and federal environmental regulations.
- Occupational Safety: Ensuring a safe working environment for manufacturing and distribution personnel is a standard regulatory and ethical requirement.
Implications:
- High Compliance Costs: Adhering to these regulations involves significant ongoing costs related to quality assurance, documentation, personnel training, and regulatory affairs.
- Risk of Penalties: Non-compliance can result in severe penalties, including warning letters, fines, product recalls, manufacturing shutdowns, and revocation of licenses.
- Need for Expertise: Companies must maintain in-house regulatory affairs teams or engage external consultants with expertise in pharmaceutical regulations.
The regulatory landscape for SM ENEMA is complex and dynamic. Ongoing vigilance and investment in robust compliance programs are essential for market participation and risk mitigation.
Key Takeaways
The SM ENEMA READY TO USE TWIN PAK operates in a mature, competitive market driven by screening demand and demographic trends. Its price is subject to downward pressure from generic alternatives, projected to remain stable to slightly declining. The intellectual property landscape is characterized by expired core patents, limiting market exclusivity and favoring cost-efficient manufacturing. Market growth is expected to align with broader bowel preparation trends, around 3-5% annually, contingent on continued demand for colonoscopies and the adoption of convenient formulations. Strict adherence to FDA and other regulatory requirements is critical for manufacturing, distribution, and market access.
Frequently Asked Questions
-
What is the primary indication for SM ENEMA READY TO USE TWIN PAK? SM ENEMA READY TO USE TWIN PAK is indicated for bowel preparation prior to colonoscopy and other colonic procedures.
-
How does SM ENEMA READY TO USE TWIN PAK compare to powdered bowel preparation formulations? SM ENEMA READY TO USE TWIN PAK offers greater convenience for patients and healthcare providers by being pre-mixed and ready for immediate use, reducing the risk of preparation errors and potentially improving patient compliance compared to powdered formulations that require mixing.
-
Is SM ENEMA READY TO USE TWIN PAK available as a generic drug? Yes, SM ENEMA READY TO USE TWIN PAK is widely available from multiple generic manufacturers, reflecting the expiration of primary patents on its active ingredients and formulation.
-
What factors are most likely to influence the future pricing of SM ENEMA READY TO USE TWIN PAK? Future pricing will be primarily influenced by ongoing generic competition, manufacturing costs of active pharmaceutical ingredients and excipients, and payer reimbursement policies that favor cost-effective options.
-
What are the main regulatory bodies overseeing the production and sale of SM ENEMA READY TO USE TWIN PAK? In the United States, the primary regulatory body is the Food and Drug Administration (FDA), which oversees manufacturing practices, drug approval, and post-market surveillance. State pharmacy boards also regulate wholesale distribution and dispensing.
Citations
[1] U.S. Preventive Services Task Force. (2021). Screening for Colorectal Cancer: U.S. Preventive Services Task Force Recommendation Statement. JAMA, 325(20), 2084–2097. doi:10.1001/jama.2021.6398
[2] Grand View Research. (2023). Bowel Preparation Market Size, Share & Trends Analysis Report By Product Type (PEG-based, Phosphate-based, Stimulant laxatives, Others), By Formulation (Liquid, Powder, Tablet), By Application (Colonoscopy, Sigmoidoscopy, Barium Enema, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 - 2030. (Report Number: GVR-3-6375-8676-9)
[3] MarketsandMarkets. (2023). Bowel Preparation Market. (Report Code: PH 7924)
More… ↓
