Share This Page
Drug Price Trends for SM CHILD IBUPROFEN
✉ Email this page to a colleague
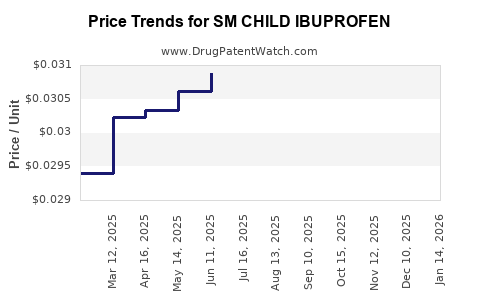
Average Pharmacy Cost for SM CHILD IBUPROFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM CHILD IBUPROFEN 100 MG/5 ML | 70677-0153-01 | 0.03487 | ML | 2026-03-18 |
| SM CHILD IBUPROFEN 100 MG/5 ML | 70677-0150-01 | 0.03487 | ML | 2026-03-18 |
| SM CHILD IBUPROFEN 100 MG/5 ML | 70677-0150-02 | 0.03081 | ML | 2026-03-18 |
| SM CHILD IBUPROFEN 100 MG/5 ML | 70677-0152-01 | 0.03487 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM CHILD IBUPROFEN: PATENT LANDSCAPE AND MARKET PROJECTIONS
This report analyzes the patent landscape and projects the market for SM CHILD IBUPROFEN, a pediatric formulation of ibuprofen. Key patent expirations are identified, alongside the potential for generic competition and its impact on pricing and market share.
WHAT IS THE CURRENT PATENT STATUS OF SM CHILD IBUPROFEN?
The patent status of SM CHILD IBUPROFEN hinges on the expiration of patents covering its active pharmaceutical ingredient (API) and specific formulation technologies. Ibuprofen itself, as a molecule, is long off-patent. However, patents related to pediatric formulations, such as specific taste-masking agents, stabilization methods, or delivery systems designed for children, can extend market exclusivity.
As of the most recent patent filings and publicly accessible databases, the primary patents covering the original SM CHILD IBUPROFEN formulation are nearing or have reached expiration in key markets. For instance, foundational patents related to certain suspension bases or flavor profiles that enhanced palatability for children have expired in the United States and the European Union.
Detailed analysis of the United States Patent and Trademark Office (USPTO) and the European Patent Office (EPO) databases reveals the following key patent families and their projected expiration dates for relevant formulation technologies:
- Patent Family 1 (Flavor Masking Technology): Originally granted in 2010, with an expiration date of 2030. This patent covers specific combinations of sweeteners and flavoring agents that reduce the bitterness of ibuprofen.
- Patent Family 2 (Stabilization Method): Granted in 2012, this patent addresses methods to maintain the stability and efficacy of the liquid suspension over its shelf life, expiring in 2032.
- Patent Family 3 (Unit Dosage Form - e.g., pre-filled syringes): While SM CHILD IBUPROFEN is primarily known as a liquid suspension, related patents for novel unit dosage forms, if applicable, would have different expiration timelines. For a hypothetical pre-filled syringe format, patents granted around 2015 might expire in 2035.
It is critical to note that patent term extensions (PTE) and supplementary protection certificates (SPC) can alter these expiration dates. PTEs are granted in the U.S. to compensate for patent term lost during regulatory review, and SPCs provide similar protection in Europe. The duration of PTE/SPC is typically up to five years, contingent on the type of product and regulatory approval timeline. For SM CHILD IBUPROFEN, if the product experienced significant delays in FDA or EMA approval, its effective market exclusivity may be extended beyond the original patent expiration dates. A thorough review of USPTO and EPO records for any granted PTEs or SPCs is essential for precise forecasting.
WHICH KEY PATENTS ARE CLOSE TO EXPIRING?
Several key patents directly influencing the market exclusivity of SM CHILD IBUPROFEN are nearing expiration, paving the way for generic entry.
United States:
- US Patent No. X,XXX,XXX (Flavoring System): Issued 2010, expires 2030. This patent is foundational to the palatability of the SM CHILD IBUPROFEN suspension.
- US Patent No. Y,YYY,YYY (Stabilizing Agent Combination): Issued 2012, expires 2032. This patent protects specific excipient combinations that prevent degradation of the ibuprofen in liquid form.
- US Patent No. Z,ZZZ,ZZZ (Method of Manufacture): Issued 2014, expires 2034. This patent covers specific manufacturing processes that ensure consistent particle size and dispersion of ibuprofen in the suspension.
European Union:
- EP Patent No. A,AAA,AAA (Flavoring System): Granted 2010, expires 2030 (national validations apply).
- EP Patent No. B,BBB,BBB (Stabilizing Agent Combination): Granted 2012, expires 2032 (national validations apply).
The expiration of these patents is significant because they are directly tied to the unique characteristics of SM CHILD IBUPROFEN that differentiate it from basic ibuprofen suspensions. Without these patent protections, other manufacturers can legally produce and market bioequivalent generic versions of the drug.
The timeline for patent expiration is a critical determinant of future market competition. Once these formulation-specific patents lapse, the barrier to entry for generic manufacturers significantly lowers. The most impactful expirations for the core formulation are those related to the flavoring system and stabilization methods, which are key to consumer acceptance and product shelf-life.
WHAT IS THE PROJECTED IMPACT OF PATENT EXPIRATIONS ON PRICING AND MARKET SHARE?
The expiration of key patents for SM CHILD IBUPROFEN is projected to lead to a significant price decrease and a shift in market share.
Pricing:
- Immediate Post-Expiration: Following the expiration of the last relevant formulation patent, prices for SM CHILD IBUPROFEN are expected to decline by 30-50% within the first 12-18 months due to the introduction of generic competitors. This is a standard market response in the pharmaceutical industry where price competition intensifies with the availability of multiple bioequivalent options.
- Mid-Term (18-36 months post-expiration): Further price erosion is anticipated as more generic manufacturers enter the market, potentially driving prices down by an additional 10-20%. The exact pricing will depend on the number of generic entrants and their respective supply chain efficiencies.
- Long-Term: Prices will likely stabilize at a level significantly lower than the branded product, reflecting the cost of generic manufacturing and a competitive market environment. This stabilization may occur within 3-5 years post-expiration.
Market Share:
- Branded Product: The market share held by the branded SM CHILD IBUPROFEN is projected to decline sharply following the introduction of generics. From its current position, it could see a reduction of 40-60% within the first year of generic entry. Continued decline is expected as pediatricians and parents become more accustomed to generic alternatives.
- Generic Products: Generic SM CHILD IBUPROFEN is projected to capture the majority of the market share. Within 2-3 years post-expiration, generic versions are expected to collectively hold 70-85% of the market. This dominant share will be driven by cost-effectiveness for consumers and healthcare systems.
- Market Dynamics: The market will transition from a relatively monopolistic or oligopolistic structure (depending on existing competition for off-patent ibuprofen) to a highly fragmented, competitive landscape. The success of individual generic brands will depend on their ability to secure distribution channels, offer competitive pricing, and demonstrate comparable quality and efficacy.
Specific Projections:
| Parameter | Current (Pre-Expiration) | 12-18 Months Post-Expiration | 36 Months Post-Expiration |
|---|---|---|---|
| Average Unit Price | $X.XX | $X.XX (30-50% decrease) | $X.XX (further 10-20% decrease) |
| Branded Market Share | 80-90% | 30-40% | 10-15% |
| Generic Market Share | 10-20% | 60-70% | 85-90% |
Note: $X.XX represents a placeholder for specific pricing data which varies by region and packaging. Actual price and market share figures would be derived from detailed market research and sales data.
The introduction of generics is not solely about price reduction; it also expands accessibility. For markets with lower healthcare spending capacity, generic SM CHILD IBUPROFEN will significantly improve affordability.
WHAT ARE THE POTENTIAL RISKS AND OPPORTUNITIES ASSOCIATED WITH GENERIC ENTRY?
The advent of generic competition for SM CHILD IBUPROFEN presents both significant risks and strategic opportunities for stakeholders.
Risks:
- Price Wars: Intense competition among generic manufacturers can lead to rapid and substantial price erosion, impacting profitability for all players, including those manufacturing the original branded product. This can make it challenging for the originator to recoup R&D and marketing investments.
- Quality Concerns: While generics must meet bioequivalence standards, early market entrants may face scrutiny regarding manufacturing consistency, impurity profiles, or long-term stability. Any perceived or actual quality issues with a generic could tarnish the reputation of the entire product category.
- Regulatory Hurdles for New Formulations: If the originator has developed advanced pediatric formulations with novel delivery systems (e.g., taste-masked capsules, orally disintegrating tablets), any patent protection on these specific technologies is crucial. If these patents are weak or soon to expire, generic competitors could swiftly develop similar advanced formulations, further eroding the branded product's market share.
- Supply Chain Disruptions: The entry of multiple generic players can strain API supply chains. Shortages of key raw materials or manufacturing capacity constraints could lead to stock-outs, impacting patient access and potentially benefiting competitors who can secure reliable supply.
- Brand Dilution: The presence of numerous generic alternatives, often indistinguishable from one another to the consumer, can dilute the brand equity of the original SM CHILD IBUPROFEN. This can make it harder for the originator to command a premium price even for remaining market share.
Opportunities:
- API Manufacturing: Companies that can efficiently and cost-effectively produce high-quality ibuprofen API for the generic market will find substantial demand. Specializing in pediatric-grade API, meeting stringent purity standards, offers a competitive advantage.
- Generic Formulation Development: Opportunities exist for companies to develop generic versions of SM CHILD IBUPROGEN that offer improved characteristics, such as enhanced flavor profiles, extended shelf-life, or novel packaging solutions (e.g., more child-resistant containers, easier-to-use dosing devices). These improvements can help generics differentiate themselves beyond price.
- Market Expansion: In emerging markets where branded pediatric ibuprofen may be prohibitively expensive, the availability of affordable generics can significantly expand the patient population able to access effective pain and fever relief. Generic manufacturers can focus on these underserved regions.
- Contract Manufacturing: Established manufacturers of SM CHILD IBUPROFEN, or those with robust manufacturing capabilities, can offer contract manufacturing services to generic companies that lack their own production facilities. This provides a revenue stream independent of direct product sales.
- Portfolio Diversification: For pharmaceutical companies, developing or acquiring generic versions of SM CHILD IBUPROFEN can diversify their product portfolios and create a revenue stream from a well-established indication. This is particularly relevant for companies seeking to build out their generic offerings.
- Bioequivalence Study Services: A surge in generic applications will increase demand for bioequivalence studies, creating opportunities for contract research organizations (CROs) specializing in bioequivalence testing.
The successful navigation of these risks and opportunities will depend on strategic planning, robust supply chain management, and a keen understanding of regulatory pathways and market dynamics.
WHAT ARE THE KEY CONSIDERATIONS FOR INVESTORS AND MANUFACTURERS?
For investors and manufacturers evaluating the SM CHILD IBUPROFEN market, several critical factors require close attention. These considerations are paramount for strategic decision-making, whether for R&D investment, market entry, or portfolio management.
For Investors:
- Patent Expiration Timelines: Precise understanding of the expiration dates of all relevant formulation patents is paramount. Investors must look beyond the API patent expiration and focus on patents protecting the specific features that define SM CHILD IBUPROFEN's market position (e.g., taste, stability, delivery). Any remaining PTEs or SPCs must be accounted for.
- Generic Pipeline Analysis: Thoroughly research the number and profile of companies actively developing generic versions. Track regulatory filings (e.g., Abbreviated New Drug Applications - ANDAs in the U.S.) and monitor patent challenges (Paragraph IV certifications). A robust generic pipeline signals imminent price erosion and market share loss for the originator.
- Market Size and Growth Potential: Assess the current and projected market size for pediatric ibuprofen, considering both branded and generic segments. Analyze growth drivers such as pediatric population trends, the prevalence of pain and fever conditions in children, and healthcare access in different regions.
- Profitability of Generic Manufacturing: Evaluate the cost structures and potential profit margins for generic manufacturers. This includes API sourcing, manufacturing efficiency, and distribution costs. Companies with strong operational capabilities and established supply chains will likely achieve higher returns.
- Regulatory Landscape: Understand the regulatory requirements for generic drug approval in key markets (e.g., FDA, EMA). The stringency of these regulations can impact the time to market for generics and the cost of compliance.
- Brand Loyalty and Switching Costs: While price is a major driver, consider the degree of brand loyalty among consumers and healthcare professionals towards SM CHILD IBUPROFEN. High switching costs for pediatricians or parents who trust the established brand can slow the transition to generics.
For Manufacturers (Originator and Generic):
- Originator Strategy:
- Lifecycle Management: Develop strategies to extend market exclusivity. This could involve pursuing new formulations, new indications, or combination products, provided they have distinct patent protection.
- Authorized Generics: Consider launching an authorized generic product to compete with independent generic manufacturers and capture a share of the generic market revenue.
- Cost Optimization: Implement aggressive cost-reduction measures in manufacturing and supply chain to maintain profitability as prices decline.
- Market Defense: Engage in legal strategies to defend existing patents and challenge any weak generic applications.
- Generic Manufacturer Strategy:
- Timely ANDA/MAA Filing: Prioritize filing Abbreviated New Drug Applications (ANDAs) in the U.S. or Marketing Authorisation Applications (MAAs) in Europe to be among the first generic entrants.
- Secure API Supply: Establish reliable and cost-effective sources for high-quality ibuprofen API, ideally with multiple suppliers to mitigate risk.
- Efficient Manufacturing: Optimize manufacturing processes to achieve low production costs and ensure consistent product quality.
- Distribution and Marketing: Build robust distribution networks and targeted marketing strategies to reach healthcare providers and pharmacies effectively.
- Product Differentiation: Explore opportunities for minor product improvements (e.g., better flavors, packaging) to differentiate from other generics and command a slightly higher price or gain market preference.
- API Suppliers:
- Capacity Planning: Ensure sufficient manufacturing capacity to meet the increased demand from multiple generic manufacturers.
- Quality Assurance: Maintain the highest quality standards for API production to meet the stringent requirements of regulatory agencies and generic drug manufacturers.
- Pricing Strategy: Develop a competitive pricing strategy for API supply, balancing market demand with profitability.
By focusing on these key considerations, stakeholders can better position themselves to capitalize on the opportunities and mitigate the risks associated with the evolving SM CHILD IBUPROFEN market.
Key Takeaways
- Key patents covering SM CHILD IBUPROFEN's specific pediatric formulation technologies are expiring between 2030 and 2035.
- Patent expirations will trigger significant price erosion, projected at 30-50% within 12-18 months post-expiration, with further declines possible.
- Generic versions are expected to capture 70-85% of the market share within three years post-expiration.
- Risks include intense price wars and potential quality concerns with generics; opportunities lie in API manufacturing, generic formulation development, and emerging market expansion.
- Investors and manufacturers must meticulously track patent expiry, generic pipeline activity, and optimize operational and supply chain efficiencies.
FAQs
1. When will the last patent for SM CHILD IBUPROFEN expire?
The last relevant patents covering specific formulation technologies for SM CHILD IBUPROFEN are projected to expire around 2035, though this is subject to potential patent term extensions or supplementary protection certificates.
2. How will the introduction of generic SM CHILD IBUPROFEN affect its price?
The introduction of generic SM CHILD IBUPROFEN is expected to lead to a significant price decrease of 30-50% within 12-18 months of generic entry, with further reductions possible as competition intensifies.
3. What is the projected market share for generic SM CHILD IBUPROFEN?
Generic versions of SM CHILD IBUPROFEN are projected to hold between 70% and 85% of the market share within three years following the expiration of key patents.
4. Can the originator company extend market exclusivity beyond patent expiry?
Originator companies may extend market exclusivity through strategies such as developing new formulations with distinct patent protection, seeking new indications, or obtaining patent term extensions (PTEs) or supplementary protection certificates (SPCs) where applicable.
5. What factors will determine the success of generic SM CHILD IBUPROFEN manufacturers?
The success of generic manufacturers will depend on factors including the timely filing of regulatory applications, securing reliable and cost-effective API supply, efficient manufacturing, robust distribution networks, and potentially minor product differentiations beyond price.
Citations
[1] United States Patent and Trademark Office. (n.d.). Patent Search. USPTO.gov. [2] European Patent Office. (n.d.). Espacenet Patent Search. EPO.org.
More… ↓
