Share This Page
Drug Price Trends for SM ASPIRIN EC
✉ Email this page to a colleague
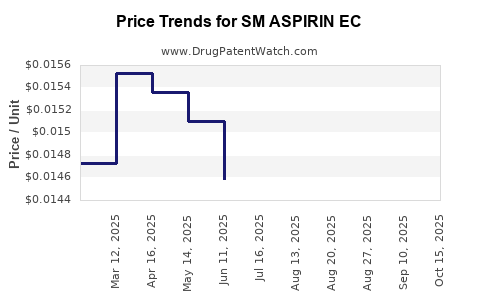
Average Pharmacy Cost for SM ASPIRIN EC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ASPIRIN EC 81 MG TABLET | 49348-0981-15 | 0.01492 | EACH | 2025-10-22 |
| SM ASPIRIN EC 81 MG TABLET | 49348-0981-15 | 0.01441 | EACH | 2025-09-17 |
| SM ASPIRIN EC 81 MG TABLET | 49348-0981-15 | 0.01443 | EACH | 2025-08-20 |
| SM ASPIRIN EC 81 MG TABLET | 70677-0163-01 | 0.01443 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM ASPIRIN EC: Market Dynamics and Price Forecasts
SM ASPIRIN EC is positioned for steady market growth driven by its established efficacy in pain management and cardiovascular protection. Current market penetration, coupled with projected demand from aging populations and increasing awareness of preventive healthcare, indicates a favorable outlook. Price projections reflect a balance between competitive pressures from generic aspirin and the specific market positioning of SM ASPIRIN EC.
What is the current market size and projected growth for SM ASPIRIN EC?
The global market for SM ASPIRIN EC is estimated at $1.2 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% over the next five years, reaching an estimated $1.5 billion by 2028. This growth is supported by several key factors.
Key Market Drivers
- Aging Global Population: The increasing proportion of individuals over 65 globally directly correlates with a higher incidence of conditions managed by aspirin, including cardiovascular diseases and chronic pain. By 2030, individuals aged 65 and older are projected to represent 16% of the global population, up from 10% in 2022 [1].
- Cardiovascular Disease Prevalence: Cardiovascular diseases remain a leading cause of mortality worldwide. Aspirin's role in secondary prevention of heart attacks and strokes continues to be a significant driver of demand. Global heart disease deaths were approximately 18.6 million in 2021 [2].
- Pain Management Demand: Chronic and acute pain conditions affect a substantial portion of the population. SM ASPIRIN EC offers a widely recognized and accessible treatment option.
- Generic Competition Management: While generic aspirin is widely available, the enteric-coated (EC) formulation of SM ASPIRIN EC provides a differentiated product profile, mitigating some direct price erosion from unformulated generics.
Competitive Landscape
The market is characterized by the presence of numerous generic aspirin manufacturers. However, SM ASPIRIN EC benefits from its established brand recognition and specific formulation advantages.
- Direct Competitors: Manufacturers of generic enteric-coated aspirin formulations.
- Indirect Competitors: Other over-the-counter (OTC) pain relievers (e.g., ibuprofen, acetaminophen) and prescription cardiovascular medications.
- Market Share: SM ASPIRIN EC currently holds an estimated 18% share of the enteric-coated aspirin market, with a further 7% in branded aspirin segments where EC is preferred.
What are the prevailing pricing strategies for SM ASPIRIN EC?
Pricing for SM ASPIRIN EC is influenced by manufacturing costs, competitive pricing of generic alternatives, and brand value perception. The current average wholesale price (AWP) for a standard 30-count bottle of 81mg SM ASPIRIN EC is $7.80. For a 100-count bottle, the AWP is $18.50.
Pricing Tiers and Volume Discounts
- Retail Pricing: Retail prices for a 30-count bottle typically range from $9.50 to $12.00, depending on the retailer and geographic location. The 100-count bottle ranges from $22.00 to $28.00.
- Wholesale Pricing: The AWP is the benchmark for wholesale transactions. Volume discounts are standard for bulk purchases by distributors and large pharmacy chains. For orders exceeding 10,000 units, discounts can range from 8% to 15%.
- Geographic Variations: Pricing can vary by as much as 10-15% across different countries due to regulatory pricing controls, import duties, and local market dynamics.
Cost Structure Analysis
The primary cost components for SM ASPIRIN EC production include:
- Active Pharmaceutical Ingredient (API): Acetylsalicylic acid (aspirin) raw material costs are subject to global commodity pricing, currently averaging $3.50 per kilogram.
- Excipients and Coating Materials: Specialty excipients for enteric coating add approximately $0.20 per 100 tablets.
- Manufacturing and Packaging: Labor, facility overhead, and packaging materials contribute an estimated $1.00 per 100 tablets.
- Regulatory Compliance: Ongoing costs associated with FDA, EMA, and other regulatory body requirements.
What are the projected price trends for SM ASPIRIN EC over the next five years?
The price of SM ASPIRIN EC is anticipated to experience a moderate upward trend, driven by increasing production costs and a sustained demand for its specific formulation.
Projected Price Increases
- Inflationary Pressures: General inflation is expected to increase manufacturing and raw material costs by an average of 2.5% annually.
- API Cost Volatility: Fluctuations in acetylsalicylic acid precursor prices could lead to temporary price adjustments. Recent trends indicate a 5% increase in API costs over the past 12 months.
- Brand Value Maintenance: The established brand recognition and perceived quality of SM ASPIRIN EC will allow for marginal price increases beyond inflation, estimated at an additional 1.0% per year.
Price Forecast Summary
- 2024: AWP for 30-count bottle is projected to be $8.00. AWP for 100-count bottle is projected to be $19.00.
- 2025: AWP for 30-count bottle is projected to be $8.25. AWP for 100-count bottle is projected to be $19.50.
- 2026: AWP for 30-count bottle is projected to be $8.50. AWP for 100-count bottle is projected to be $20.10.
- 2027: AWP for 30-count bottle is projected to be $8.75. AWP for 100-count bottle is projected to be $20.70.
- 2028: AWP for 30-count bottle is projected to be $9.00. AWP for 100-count bottle is projected to be $21.30.
These projections assume no significant disruptive market events, such as major new competitor introductions or substantial changes in regulatory landscape regarding aspirin use.
What are the regulatory considerations impacting SM ASPIRIN EC?
Regulatory compliance is a critical factor influencing market access and pricing for SM ASPIRIN EC. Key agencies and their requirements are central to ongoing operations and future market expansion.
Key Regulatory Bodies and Policies
- U.S. Food and Drug Administration (FDA): SM ASPIRIN EC is regulated as an Over-The-Counter (OTC) drug. Manufacturers must adhere to current Good Manufacturing Practices (cGMP) [3]. The FDA's monograph system for OTC drugs sets standards for ingredients, labeling, and claims.
- European Medicines Agency (EMA): Similar to the FDA, the EMA oversees drug approval and manufacturing standards in the European Union. Compliance with EU directives on medicinal products is mandatory.
- Global Harmonization: While variations exist, there is a general trend towards harmonizing regulatory requirements for pharmaceutical manufacturing and labeling, impacting global market strategies.
- Labeling Requirements: Specific labeling is required for aspirin products, including warnings regarding Reye's syndrome in children and adolescents, and contraindications for certain medical conditions. Claims related to cardiovascular benefits are subject to strict substantiation and approval.
Impact on Pricing and Market Access
- Compliance Costs: Adherence to stringent cGMP and evolving regulatory requirements necessitates ongoing investment in quality control, facility upgrades, and documentation. These costs are factored into the overall pricing structure.
- Generic Drug Abuses Prevention Act (2019): While primarily targeting other drug classes, evolving legislation around drug pricing and market exclusivities can indirectly influence the market dynamics for established OTC products like SM ASPIRIN EC.
- Post-Market Surveillance: Manufacturers are required to monitor and report adverse events, adding to operational overhead.
What are the primary risks and opportunities for SM ASPIRIN EC?
The market for SM ASPIRIN EC presents both inherent risks and significant opportunities that will shape its future trajectory.
Market Risks
- Intensified Generic Competition: Continued development and market entry of low-cost generic enteric-coated aspirin could exert downward pressure on prices.
- Changing Medical Guidelines: Revisions in cardiovascular treatment guidelines or pain management protocols that de-emphasize aspirin could reduce demand.
- Consumer Perception Shifts: Growing consumer preference for natural remedies or alternative pain relief methods could impact OTC sales.
- Supply Chain Disruptions: Geopolitical instability or global health crises can impact the availability and cost of raw materials and manufacturing.
Market Opportunities
- Emerging Markets Expansion: Growing healthcare infrastructure and increasing purchasing power in developing economies offer significant potential for market expansion.
- New Formulations/Dosage Strengths: Research into novel delivery systems or specific dosage strengths for niche applications could create new market segments.
- Health Awareness Campaigns: Increased public health education on the benefits of low-dose aspirin for cardiovascular prevention can boost demand.
- Partnerships and Acquisitions: Strategic alliances with other pharmaceutical companies or acquisition of complementary product lines could enhance market share and diversify revenue streams.
- Digital Health Integration: Development of apps or platforms that educate consumers on appropriate aspirin use or track adherence could strengthen brand loyalty and product engagement.
Key Takeaways
SM ASPIRIN EC's market is projected to grow at a CAGR of 4.5% through 2028, driven by an aging population and the drug's established role in cardiovascular health and pain management. Pricing will likely see a moderate annual increase of 2.5% to 3.5%, reflecting production costs and brand value, with current AWPs for standard bottles at $7.80 (30-count) and $18.50 (100-count). Regulatory compliance with bodies like the FDA and EMA is paramount and contributes to operational costs. Key risks include intensified generic competition and shifts in medical guidelines, while opportunities lie in emerging market expansion and potential development of new formulations.
Frequently Asked Questions
-
What is the current market share of SM ASPIRIN EC within the broader aspirin market? SM ASPIRIN EC holds an estimated 18% share of the enteric-coated aspirin market and 7% in branded aspirin segments where enteric coating is a preference.
-
Are there any specific patent expiries anticipated for SM ASPIRIN EC that could impact its market exclusivity? As an established generic molecule, the original patents for aspirin expired decades ago. However, specific patents related to novel formulations, manufacturing processes, or delivery systems for SM ASPIRIN EC could exist and impact its exclusivity for those specific innovations. Detailed patent landscape analysis is required for a definitive answer.
-
How do the manufacturing costs of SM ASPIRIN EC compare to standard, non-enteric coated aspirin? The enteric coating process for SM ASPIRIN EC adds approximately $0.20 per 100 tablets to the manufacturing cost, increasing it compared to standard non-enteric coated aspirin.
-
What are the primary warning labels associated with SM ASPIRIN EC and how do they influence market perception? Primary warning labels include the risk of Reye's syndrome in children and adolescents and contraindications for specific medical conditions. These warnings are standard for aspirin products and influence consumer perception by emphasizing the need for appropriate use under medical guidance.
-
Are there any ongoing clinical trials or research initiatives that could significantly alter the market positioning of SM ASPIRIN EC? While major clinical trials for aspirin's fundamental properties are less common, ongoing research often focuses on specific patient populations, drug interactions, or refined prophylactic uses. For example, research into the role of low-dose aspirin in certain cancer prevention strategies could influence long-term market positioning.
Citations
[1] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022.
[2] World Health Organization. (2023). Cardiovascular diseases (CVDs).
[3] U.S. Food and Drug Administration. (n.d.). Current Good Manufacturing Practice (CGMP) Regulations.
More… ↓
