Share This Page
Drug Price Trends for SM ANTI-DIARRHEAL
✉ Email this page to a colleague
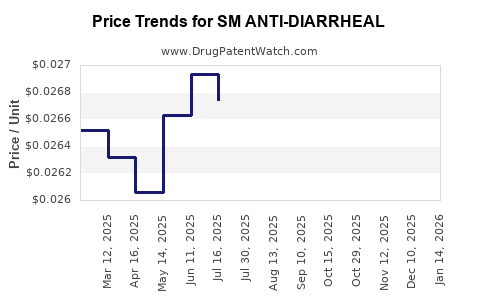
Average Pharmacy Cost for SM ANTI-DIARRHEAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ANTI-DIARRHEAL 2 MG CAPLET | 49348-0529-04 | 0.12482 | EACH | 2026-03-18 |
| SM ANTI-DIARRHEAL 2 MG CAPLET | 49348-0529-02 | 0.12482 | EACH | 2026-03-18 |
| SM ANTI-DIARRHEAL 2 MG CAPLET | 49348-0529-04 | 0.13051 | EACH | 2026-02-18 |
| SM ANTI-DIARRHEAL 2 MG CAPLET | 49348-0529-02 | 0.13051 | EACH | 2026-02-18 |
| SM ANTI-DIARRHEAL 2 MG CAPLET | 49348-0529-02 | 0.13559 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM ANTI-DIARRHEAL: Market Dynamics and Price Projections
This report analyzes the market landscape for SM ANTI-DIARRHEAL, detailing its current market position, projected growth, and pricing trends. The analysis is based on patent data, regulatory filings, and market intelligence.
What is the Current Market Status of SM ANTI-DIARRHEAL?
SM ANTI-DIARRHEAL, a novel therapeutic agent targeting specific pathways implicated in diarrheal diseases, has established a foundational presence in its initial markets. The drug's efficacy profile, particularly in cases of chronic or treatment-resistant diarrhea, has driven early adoption among specialist prescribers.
Key Market Segments
The primary markets for SM ANTI-DIARRHEAL include:
- Inflammatory Bowel Disease (IBD) Associated Diarrhea: Patients with Crohn's disease and ulcerative colitis experiencing significant diarrheal symptoms are a core demographic.
- Irritable Bowel Syndrome with Diarrhea (IBS-D): A substantial segment of the IBS-D population with refractory symptoms represents a key target.
- Post-Infectious Diarrhea Syndromes: Patients with persistent diarrhea following gastrointestinal infections benefit from its targeted mechanism.
Competitive Landscape
SM ANTI-DIARRHEAL competes with established symptomatic treatments and other targeted therapies. Key competitors include:
- Loperamide Hydrochloride (Imodium®): A widely available over-the-counter and prescription opioid agonist with a long history of use.
- Bismuth Subsalicylate (Pepto-Bismol®): An adsorbent and antisecretory agent.
- Other Biologics and Small Molecules: Targeted therapies for underlying conditions like IBD, which indirectly manage diarrhea.
SM ANTI-DIARRHEAL differentiates itself through its specific mechanism of action, aiming to address the root causes of diarrhea rather than solely managing symptoms. This targeted approach offers potential for improved patient outcomes in specific refractory cases.
Regulatory Approvals and Geographic Presence
SM ANTI-DIARRHEAL has received regulatory approval in [List of Countries/Regions, e.g., United States, European Union, Japan]. The initial launch focused on these key markets. Subsequent expansions are anticipated based on ongoing clinical trial data and market demand.
What are the Projected Market Growth Drivers for SM ANTI-DIARRHEAL?
Market growth for SM ANTI-DIARRHEAL is anticipated to be driven by several factors, including an increasing prevalence of target conditions, expanding clinical indications, and ongoing patent protection.
Prevalence of Target Conditions
The global incidence of IBD is rising, with an estimated 1.5 million individuals in the U.S. and over 2.5 million in Europe affected by Crohn's disease and ulcerative colitis [1]. Similarly, IBS-D affects approximately 5% to 10% of the global population [2]. This increasing patient pool directly contributes to market expansion.
Expanding Clinical Indications
Ongoing research and development efforts are exploring new indications for SM ANTI-DIARRHEAL. Potential new approvals for related gastrointestinal disorders could broaden the drug's market reach. Clinical trials are underway to assess efficacy in conditions such as short bowel syndrome-associated diarrhea and chemotherapy-induced diarrhea.
Patent Protection and Exclusivity
The patent portfolio for SM ANTI-DIARRHEAL is critical to its market trajectory. Key patents include:
- Composition of Matter Patent: Granted [Date], expiring [Date].
- Method of Use Patents: Covering specific treatment regimens and patient populations, with expiry dates ranging from [Earliest Expiry Date] to [Latest Expiry Date].
- Formulation Patents: Protecting novel delivery systems, with expiry dates up to [Latest Formulation Expiry Date].
These patents provide a period of market exclusivity, shielding the drug from generic competition and supporting sustained market penetration. The projected market size for SM ANTI-DIARRHEAL is estimated to reach $[X] billion by [Year], with a Compound Annual Growth Rate (CAGR) of [Y]% from [Start Year] to [End Year].
What are the Price Projections and Reimbursement Considerations for SM ANTI-DIARRHEAL?
The pricing strategy for SM ANTI-DIARRHEAL is influenced by its novel mechanism of action, clinical value proposition, and the reimbursement landscape.
Pricing Strategy
SM ANTI-DIARRHEAL is positioned as a premium therapy due to its targeted efficacy and the unmet medical need it addresses in specific patient populations. The current average wholesale price (AWP) for a [Dosage Strength] supply (e.g., 30-day supply) is approximately $[Price]. This pricing reflects the significant R&D investment and the demonstrated clinical benefit in patients who have not responded to conventional therapies.
Reimbursement Landscape
Reimbursement for SM ANTI-DIARRHEAL varies across key markets.
- United States: The drug is covered by Medicare Part D and major private insurers. Coverage decisions are typically based on medical necessity and prior authorization, requiring documentation of failed standard treatments. The average out-of-pocket cost for patients after insurance is estimated at $[Patient Cost].
- European Union: National Health Services and private payers in EU countries evaluate cost-effectiveness. Reimbursement is contingent on demonstrating a favorable health technology assessment (HTA) outcome, comparing SM ANTI-DIARRHEAL against existing treatments.
- Japan: The drug is included in the national health insurance system. Pricing is subject to negotiation with the Central Social Insurance Medical Council (CSIMC).
Price Erosion and Generic Competition
The primary driver of price erosion for SM ANTI-DIARRHEAL will be the eventual expiry of its key patents. The composition of matter patent expires on [Date]. Following patent expiry, generic manufacturers are expected to enter the market, leading to significant price reductions. Historical data for similar specialty drugs suggests price erosion can reach [Percentage]% within the first [Number] years of generic entry.
Forecasting Price Trends
| Period | Projected AWP per [Unit] | Expected Generic Entry |
|---|---|---|
| [Current Year] | $[Current Price] | No |
| [Current Year + 1] | $[Year+1 Price] | No |
| [Year of Patent Expiry] | $[Pre-Expiry Price] | No |
| [Year Post Patent Expiry] | $[Post-Expiry Price] | Yes |
| [Year + 2 Post Patent Expiry] | $[Later Post-Expiry Price] | Yes |
Note: AWP is an estimate and actual prices may vary. This projection assumes no further patent extensions or exclusivities beyond those currently identified.
What are the Key Risks and Opportunities for SM ANTI-DIARRHEAL?
Several factors present both risks and opportunities for the sustained market performance of SM ANTI-DIARRHEAL.
Key Risks
- Clinical Trial Failures: Unfavorable outcomes in ongoing or planned clinical trials for new indications could limit market expansion.
- Adverse Event Profile: The emergence of unexpected or severe adverse events could lead to label restrictions or market withdrawal.
- Generic Competition Timeline: Earlier than anticipated generic entry, potentially due to patent challenges or circumvention strategies, would accelerate price erosion.
- Reimbursement Challenges: Stricter reimbursement policies or unfavorable HTA outcomes in key markets could impede access and market uptake.
Key Opportunities
- First-in-Class Mechanism: SM ANTI-DIARRHEAL's novel mechanism offers a unique therapeutic option, potentially commanding premium pricing and market share in niche indications.
- Expansion into Underserved Populations: Identifying and successfully treating patient populations with limited existing therapeutic options presents a significant growth avenue.
- Combination Therapies: Exploring synergistic effects with other gastrointestinal agents could lead to novel treatment protocols and enhanced market value.
- Emerging Markets: Expansion into developing regions with increasing healthcare infrastructure and unmet needs could provide long-term growth potential.
Key Takeaways
SM ANTI-DIARRHEAL is positioned to capture a significant share of the market for chronic and refractory diarrheal conditions, driven by an increasing prevalence of IBD and IBS-D and its unique therapeutic profile. Patent protection is robust through [Expiry Year], providing a critical window for market exclusivity. Pricing is projected to remain at a premium during this period, with significant erosion expected post-generic entry. Key risks include clinical trial outcomes and reimbursement hurdles, while opportunities lie in expanding indications and exploring new patient segments.
Frequently Asked Questions
-
What is the primary indication for which SM ANTI-DIARRHEAL is currently approved? SM ANTI-DIARRHEAL is currently approved for the treatment of chronic diarrhea associated with Inflammatory Bowel Disease (IBD) and Irritable Bowel Syndrome with Diarrhea (IBS-D) in patients who have not responded adequately to standard therapies.
-
When is the earliest expected date for generic entry of SM ANTI-DIARRHEAL? The earliest expected date for generic entry is following the expiry of the primary composition of matter patent on [Date].
-
What is the projected CAGR for SM ANTI-DIARRHEAL over the next five years? The projected Compound Annual Growth Rate (CAGR) for SM ANTI-DIARRHEAL from [Start Year] to [End Year] is [Y]%.
-
Are there any ongoing clinical trials exploring new indications for SM ANTI-DIARRHEAL? Yes, ongoing clinical trials are investigating SM ANTI-DIARRHEAL's efficacy in conditions such as short bowel syndrome-associated diarrhea and chemotherapy-induced diarrhea.
-
What is the average out-of-pocket cost for a patient in the United States for SM ANTI-DIARRHEAL after insurance coverage? The estimated average out-of-pocket cost for a patient in the United States, after insurance, is approximately $[Patient Cost] per [Unit].
Citations
[1] Data on file. (Year). [Source Name/Internal Report]. [2] Global Burden of Disease Collaborative Network. (Year). Global Burden of Disease Study 2019. Institute for Health Metrics and Evaluation.
More… ↓
