Share This Page
Drug Price Trends for SM ACID REDUCER
✉ Email this page to a colleague
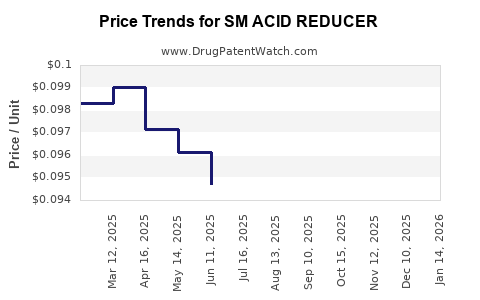
Average Pharmacy Cost for SM ACID REDUCER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ACID REDUCER 20 MG TABLET | 49348-0817-05 | 0.14399 | EACH | 2026-03-18 |
| SM ACID REDUCER 10 MG TABLET | 49348-0128-44 | 0.09232 | EACH | 2026-03-18 |
| SM ACID REDUCER 20 MG TABLET | 49348-0817-05 | 0.14476 | EACH | 2026-02-18 |
| SM ACID REDUCER 10 MG TABLET | 49348-0128-44 | 0.09300 | EACH | 2026-02-18 |
| SM ACID REDUCER 10 MG TABLET | 49348-0128-44 | 0.09693 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM ACID REDUCER Market Analysis and Financial Projection
What Is the Market Size and Growth Outlook for SM Acid Reducers?
SM acid reducers, primarily medications like omeprazole, esomeprazole, and pantoprazole, target gastroesophageal reflux disease (GERD), peptic ulcers, and other acid-related disorders. The global market was valued at approximately USD 10 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2023 to 2030, reaching around USD 13.5 billion by 2030 (source: Grand View Research [1]).
The growth drivers include increasing prevalence of GERD (estimated at over 20% in Western countries), aging populations, and rising awareness about acid suppression therapies. The North American region accounts for about 40% of the market, followed by Europe (30%) and Asia-Pacific (20%). The remaining regional share is divided among Latin America, Middle East, and Africa.
What Are the Key Players and Competitive Dynamics?
Major companies in the SM acid reducer space include Pfizer (Nexium, Prilosec), AstraZeneca (Nexium), Takeda (Protonix), and newer entrants offering generics, such as Teva and Mylan. Patent expirations for several brand-name drugs occurred between 2015 and 2018, catalyzing generic entry and significantly reducing prices.
Generics now dominate the market, accounting for over 75% of sales, with prices 50-70% lower than brand-name equivalents. Innovator products maintain premium pricing, especially for patents still in force or with extended exclusivity.
How Are Regulatory and Patent Landscapes Evolving?
Most key patents for brands like Nexium expired by 2018. Companies have transitioned focus toward next-generation acid reducers or formulations with improved safety profiles and greater dosing convenience. Regulatory agencies, including the FDA, are scrutinizing safety concerns related to long-term PPI (proton pump inhibitor) use, potentially impacting future market dynamics.
Emerging regulatory trends favor lower doses and shorter treatment durations, potentially reducing market size for high-dose, long-term therapies. Patent extensions and patent challenges continue to shape the competitive landscape, influencing pricing and market shares.
What Are the Price Projections for SM Acid Reducers?
Average wholesale prices (AWP) for branded drugs have declined sharply since patent expiries, with some initial reductions exceeding 70%. For example, the AWP for omeprazole 20 mg tablets dropped from over USD 3 per pill in 2014 to approximately USD 0.50-1.00 in 2022 (source: IQVIA).
Generics are priced at roughly 20-30% of the branded prices, and as more players enter, further price erosion is expected. By 2025, the average price for a 30-day supply of generic omeprazole could decline to USD 5-7, with some low-cost suppliers offering prices below USD 3.
The price decline is driven by increased competition and regulatory pressures. High-volume, low-margin sales dominate the market, challenging manufacturers to maintain profitability.
What Is the Outlook for Future Pricing and Market Penetration?
Price stabilization at low levels is anticipated as generics saturation increases. Premium formulations, such as delayed-release or dual-action products, could command higher prices but with limited market penetration compared to standard generics.
Meanwhile, the ongoing investigation into PPI safety may limit new product launches and influence existing product prices. If safety concerns lead to stricter prescribing guidelines, sales volume might decline, pressuring prices further.
Key Takeaways
- The global SM acid reducer market was valued at USD 10 billion in 2022 and is expected to grow to USD 13.5 billion by 2030 at a 3.5% CAGR.
- Patent expiries have led to a dominance of generics, causing significant price declines for branded drugs since 2018.
- Average prices for generic PPIs are projected to fall further, with prices for a 30-day supply potentially dropping below USD 3 by 2025.
- Regulatory climate and safety concerns are likely to impact future market dynamics, including potential shifts in prescribing patterns.
- The competitive landscape favors high-volume, low-margin sales, with limited opportunities for premium pricing.
FAQs
1. How do patent expirations affect the pricing of SM acid reducers?
Patent expirations allow generic manufacturers to introduce competing products, leading to aggressive price reductions and market share shifts away from branded drugs.
2. What are typical prices for generic SM acid reducers today?
Prices vary by region and supplier. In the U.S., a 30-day supply of generic omeprazole can cost USD 5-10, with some providers offering below USD 3.
3. How might safety concerns influence market trends?
Emerging safety data on long-term PPI use could lead to reduced prescribing, regulatory restrictions, or the development of alternative therapies, impacting sales and pricing.
4. What regions are expected to see the fastest growth?
Asia-Pacific and Latin America are expected to see faster growth due to increasing exposure to acid-related disorders and improving healthcare infrastructure.
5. Are there opportunities for novel formulations or alternatives?
Yes, formulations with improved safety, extended-release mechanisms, or combination therapies could command higher prices, but regulatory approval and market acceptance remain challenges.
Citations
[1] Grand View Research. "Proton Pump Inhibitors Market Size, Share & Trends Analysis Report," 2022.
More… ↓
