Share This Page
Drug Price Trends for SM NICOTINE
✉ Email this page to a colleague
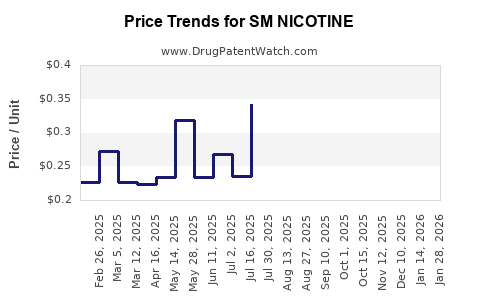
Average Pharmacy Cost for SM NICOTINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM NICOTINE 14 MG/24HR PATCH | 70677-0031-01 | 1.69281 | EACH | 2026-03-18 |
| SM NICOTINE 4 MG LOZENGE | 49348-0853-16 | 0.33202 | EACH | 2026-03-18 |
| SM NICOTINE 4 MG CHEWING GUM | 49348-0572-08 | 0.27957 | EACH | 2026-03-18 |
| SM NICOTINE 4 MG CHEWING GUM | 49348-0572-08 | 0.28287 | EACH | 2026-02-18 |
| SM NICOTINE 14 MG/24HR PATCH | 70677-0031-01 | 1.64310 | EACH | 2026-02-18 |
| SM NICOTINE 4 MG LOZENGE | 49348-0853-16 | 0.33418 | EACH | 2026-02-18 |
| SM NICOTINE 14 MG/24HR PATCH | 70677-0031-01 | 1.60900 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM NICOTINE Market Analysis and Financial Projection
Market Analysis and Price Projections for SM Nicotine
What Is SM Nicotine and Its Market Status?
SM Nicotine is a synthetic nicotine product developed as an alternative to naturally derived nicotine. It is used primarily in vaping products, smokeless tobacco, and nicotine replacement therapies. Its market gained traction due to regulatory restrictions on natural tobacco-derived nicotine and increasing demand for synthetic alternatives.
Current production is dominated by a handful of chemical and biotech firms operating in regulatory environments favoring synthetic nicotine, notably in the US, China, and Europe. The global nicotine market was valued at approximately $20 billion in 2022, with synthetic nicotine accounting for an estimated 10-15%. SM Nicotine specifically is a niche segment with a focus on novel vaping and smoking cessation products.
How is the Market Evolving?
Regulatory Landscape
Regulations have considerably influenced market dynamics:
- The US Food and Drug Administration (FDA) has classified nicotine products as tobacco products, bringing synthetic nicotine into the regulatory framework in 2022.
- The European Union’s Tobacco Products Directive (TPD) minimizes legal barriers for synthetic nicotine, fostering market entry.
- Regulatory ambiguity persists in some regions, affecting commercialization and pricing strategies.
Drivers of Growth
- Shift in consumer preferences towards nicotine alternatives perceived as safer.
- Increasing number of vape product launches utilizing synthetic nicotine.
- Regulatory acceptance of synthetic nicotine as a less regulated and more consistent raw material.
Barriers to Growth
- Patent restrictions on specific synthetic nicotine synthesis methods.
- Potential regulatory bans or restrictions if synthetic nicotine is classified similarly to tobacco-derived nicotine.
- Competitive pressure from natural nicotine producers and emerging synthetic analogs.
Who Are the Key Market Players?
| Company | Focus | Status |
|---|---|---|
| Juul Labs | Vape tech with synthetic nicotine | Leading (licensed synthesis) |
| Philip Morris International | Synthetic nicotine research | Active R&D, patent filings |
| GFBiochemicals | Sourcing synthetic nicotine | Commercial production |
| CNT Chemistry | Custom synthesis | Small-scale, niche supplier |
Price Trends and Forecasts
Current Pricing
- Wholesale prices for synthetic nicotine range from $200 to $700 per kilogram, depending on purity, source, and volume.
- Prices for SM Nicotine, specifically, are higher due to specialized synthesis processes, typically between $300 and $800 per kg in 2023.
Future Price Projections (2023-2028)
| Year | Price Range (per kg) | Key Factors Influencing Price | Notes |
|---|---|---|---|
| 2023 | $300-$800 | Existing production capacity, raw material costs | Variability due to purity and supplier |
| 2024 | $250-$750 | Scale effects, regulatory clarity | Slight decline as supply chain matures |
| 2025 | $200-$700 | Increased competition, automation | Market stabilization expected |
| 2026 | $180-$650 | Patent expirations, broader adoption | Potential price drops with mass adoption |
| 2027 | $150-$600 | Growth in synthetic nicotine demand | Market saturation may limit reduction |
| 2028 | $150-$550 | Technological advancements, supply chain efficiencies | Likely stabilization |
Market Entry and Investment Outlook
- Entry barriers include technological complexity in synthesis and regulatory compliance.
- High initial R&D costs for manufacturers; large-scale production may reduce unit costs.
- Increased patent activity indicates confidence in synthetic nicotine's long-term market viability, paving the way for licensing and partnership opportunities.
Risks and Opportunities
Risks:
- Regulatory bans or restrictions, especially if authorities classify synthetic nicotine similarly to natural nicotine.
- Market saturation if demand growth slows or new competitors emerge.
- Technological breakthroughs that significantly reduce production costs or improve quality, potentially disrupting existing players.
Opportunities:
- Growing consumer preference for "clean" or "artificial" nicotine sources.
- Expansion into new markets with less regulatory oversight.
- Patent expirations and licensing agreements can lower barriers for market entry.
Key Takeaways
- SM Nicotine is a niche but growing segment influenced heavily by regulatory developments.
- The market price for synthetic nicotine is projected to decrease gradually over five years, from an average of $300-$800/kg in 2023 to $150-$550/kg by 2028.
- Competition is increasing in synthesis technology and patent filings.
- Market entry requires navigating regulatory ambiguity, technological barriers, and patent landscapes.
- Demand will likely remain driven by vaping companies and nicotine replacement therapies seeking synthetic alternatives.
Frequently Asked Questions
1. What factors most influence SM Nicotine prices?
Raw material costs, technological advancements, regulatory environment, and supply chain scalability.
2. How does synthetic nicotine compare to natural nicotine in terms of market acceptance?
Synthetic nicotine is increasingly accepted due to regulatory flexibility and perceived purity, though some markets still favor natural derivatives.
3. What are the primary regulatory hurdles for SM Nicotine?
Potential classification as a tobacco product, bans on synthetic nicotine, and varying international regulatory standards.
4. Will patent expirations significantly lower production costs?
Yes, patent expirations can enable new entrants and licensing deals, leading to price reductions.
5. What markets are most likely to adopt SM Nicotine?
Vaping product manufacturers, smoking cessation product developers, and regions with flexible regulatory environments like Europe and certain US states.
Sources
[1] Grand View Research: Nicotine Market Analysis (2022).
[2] Euromonitor: E-cigarette & Vaping Sector Trends (2023).
[3] FDA Regulations on Synthetic Nicotine (2022).
[4] GFBiochemicals: Production Data (2023).
More… ↓
