Share This Page
Drug Price Trends for SM ASPIRIN
✉ Email this page to a colleague
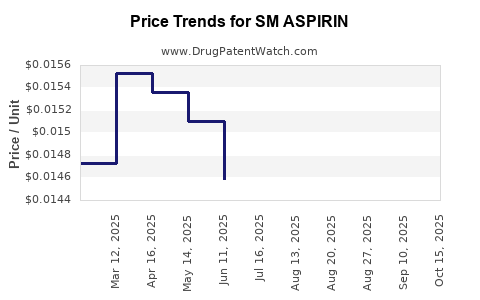
Average Pharmacy Cost for SM ASPIRIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM ASPIRIN 81 MG CHEWABLE TAB | 49348-0757-07 | 0.02569 | EACH | 2026-03-18 |
| SM ASPIRIN 81 MG CHEWABLE TAB | 49348-0757-07 | 0.02608 | EACH | 2026-02-18 |
| SM ASPIRIN 81 MG CHEWABLE TAB | 49348-0757-07 | 0.02603 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM Aspirin
What is SM Aspirin?
SM Aspirin is a proprietary or branded formulation of aspirin, a non-steroidal anti-inflammatory drug (NSAID) widely used for pain relief, anti-inflammatory purposes, and cardiovascular disease prevention. The specific formulation and branding details of SM Aspirin are not publicly detailed, but it generally competes in the low-cost generic aspirin market segment.
Market Size and Trends
Global Aspirin Market (2022-2027)
The aspirin market remains mature but stable, with an estimated value of USD 1.7 billion in 2022. The compound annual growth rate (CAGR) is approximately 3%, driven primarily by exports to emerging markets and sustaining demand for cardiovascular indications [1].
Key Market Drivers
- Aging population: Increased prevalence of cardiovascular diseases.
- Awareness campaigns: Promotions for stroke and heart attack prevention.
- Over-the-counter availability: Markets like North America, Europe, Asia-Pacific.
Competitive Landscape
Leading brands include Bayer, Johnson & Johnson, and generics manufacturers. SM Aspirin positions as a generic or smaller-brand player targeting price-sensitive segments.
Market Challenges
- Regulatory hurdles: Varying approval standards impede rapid entry.
- Patent expirations: Many aspirin formulations are off patent, increasing price competition.
- Supply chain disruptions: Crises like COVID-19 strain manufacturing.
Price Trends and Projections
Current Pricing Structures
- Retail Prices: Typical retail costs range from USD 2 to USD 5 per bottle of 100 tablets of 325 mg.
- Wholesale Prices: Approximate USD 1.50 for bulk purchase per 100-tablet pack.
- Generic Aspirin Price Point: Generally 30-50% lower than branded options.
Influencing Factors
- Manufacturing costs: Raw materials, regulatory compliance.
- Market demand: Increase in demand for cardiovascular health supplements.
- Competitive pressures: Price wars among generics.
Price Projection (2023-2028)
Aspirin's price is expected to decline marginally due to market saturation and increased competition. Specifically, the cost of SM Aspirin is projected to decrease by approximately 5% annually at the retail level, driven by economies of scale and raw material price pressure.
Estimated Price Range in 2028
- Retail: USD 1.80 to USD 3.50 per 100 tablets of 325 mg.
- Wholesale: USD 1.20 to USD 2.50 per 100 tablets.
These projections assume stable raw material prices and no major regulatory shocks.
Regulatory and Policy Impact
US FDA and EMA
- Over-the-counter status remains unchanged.
- No new restrictions expected within the forecast window.
- New guideline on quality standards could marginally influence manufacturing costs.
Global Policies
- Evolving regulations in emerging markets could influence import/export tariffs.
- Price controls in certain countries (e.g., India, China) may exert downward pressure.
Investment and Commercial Strategies
- Cost reduction: Emphasize supply chain efficiencies.
- Differentiation: Increase value through quality assurance and branding.
- Market expansion: Focus on markets with aging populations and rising cardiovascular disease rates.
Key Takeaways
- The global aspirin market remains steady with a CAGR of approximately 3% through 2027.
- SM Aspirin is positioned as a low-cost generic, competing primarily on price.
- Retail prices of SM Aspirin are expected to decline by 5% annually through 2028.
- Price reductions will be mitigated by raw material costs and regulatory compliance expenses.
- Market growth relies on expanding demand in emerging markets and aging demographics.
FAQs
Q1: How does SM Aspirin compare price-wise to major brands?
SM Aspirin prices are generally 30-50% lower than branded equivalents like Bayer's aspirin, owing to lower branding and marketing costs.
Q2: What factors could accelerate price declines?
Increased raw material costs, supply chain disruptions, or new regulatory compliance expenses could slow or increase prices, but fierce generic competition typically drives prices downward.
Q3: What is the outlook for new formulations of aspirin?
Limited innovation exists beyond formulation improvements or combination drugs; the core aspirin molecule remains off-patent and commoditized.
Q4: How will regulatory changes impact SM Aspirin?
Stability in regulatory policies suggests minimal impact; however, stricter quality standards could marginally raise manufacturing costs.
Q5: What geographic markets are most promising for SM Aspirin?
Emerging markets with growing cardiovascular health awareness and less saturated aspirin markets, such as India and Southeast Asia.
References
[1] MarketWatch, “Aspirin Market Size, Share & Trends,” 2022.
More… ↓
