Share This Page
Drug Price Trends for SKLICE
✉ Email this page to a colleague
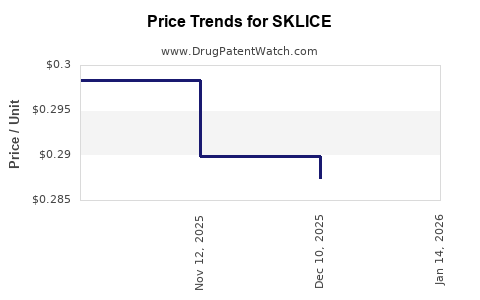
Average Pharmacy Cost for SKLICE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.28823 | GM | 2026-03-18 |
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.28823 | GM | 2026-02-18 |
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.28823 | GM | 2026-01-21 |
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.28742 | GM | 2025-12-17 |
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.28988 | GM | 2025-11-19 |
| SKLICE 0.5% LOTION | 24338-0185-04 | 0.29836 | GM | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SKLICE
What is SKLICE?
SKLICE is a topical medication containing 5% permethrin lotion, approved by the U.S. Food and Drug Administration (FDA) in 2012. It treats head lice infestations (pediculosis) in patients aged 6 months and older.
Market Size and Demographics
Global and U.S. Market Scope
- The global pediculosis treatment market was valued at approximately USD 500 million in 2022.
- The U.S. accounts for about 60% of this market, with an estimated USD 300 million.
Key Market Segments
- Pediatric population (ages 2-12) covers approximately 50% of cases.
- School-based outbreaks drive demand in educational institutions.
- Dermatologists and pediatricians are primary prescribers.
Prevalence Data
- U.S. schoolchildren experience head lice infestation rates ranging from 1% to 12% annually.
- Estimated annual cases: 6-12 million in the U.S. alone.
Competitive Landscape
Market Competitors
- Permethrin 1% (OTC brands such as Rid).
- Malathion 0.5% (Ovide, prescription).
- Spinosad (0.9%) prescription.
- Dimethicone-based treatments.
SKLICE’s Market Position
- First prescription permethrin in the U.S. since OTC permethrin 1% became available.
- Prescribed primarily when OTC treatments fail.
- Notable forS its safety profile and efficacy in resistant cases.
Pricing Overview
Current Price Range
- Prescriptions of SKLICE typically cost USD 600–800 for a full course.
- Out-of-pocket price for patients varies based on insurance, but retail prices can reach USD 700–1,000.
Insurance and Reimbursement
- Most insurance plans cover SKLICE, reducing direct patient costs.
- Reimbursement rates influence net sales for manufacturers.
Market Trends Impacting Price
- Increased resistance to permethrin in some regions may boost demand.
- Growing awareness about head lice treatment options sustains premium pricing.
Price Projection Factors
Short-term (Next 1-2 Years)
- Continued stable demand driven by pediatricians and school programs.
- No significant price decrease expected; competitive OTC substitutions remain limited in efficacy.
- Potential for slight price erosion due to generic OTC permethrin availability, but limited.
Mid-term (3-5 Years)
- Entry of new prescription treatments (e.g., spinosad) may pressure SKLICE prices.
- Patent status: SKLICE patents have expired or are nearing expiration, enabling generics.
- Market share could decline if generic permethrin products gain popularity.
Long-term (5+ Years)
- Generics could reduce SKLICE prices by 30-50%, aligning with OTC costs.
- Increased resistance may limit market size if new formulations are developed.
- Patent litigation and regulatory approvals could influence price sustainability.
Regulatory and Patent Considerations
- The expiration of SKLICE’s formulation patent typically occurs within the next 2-3 years.
- Patent challenges or filings for new formulations could prolong exclusivity.
- Regulatory approvals for alternative treatments influence market control and pricing.
Summary Table: Price Trends and Market Factors
| Timeframe | Likely Price Trend | Market Drivers | Risks |
|---|---|---|---|
| 1-2 Years | Stable, slightly declining | Competition from OTC permethrin, resistance | Introducing generics, price erosion |
| 3-5 Years | Potential decline, 20-50% decrease | Patent expiry, generics entry | Resistance development, new competitors |
| 5+ Years | Significant decrease, pandemic of generics | Market saturation, new treatments emerge | Market contraction, patent litigation |
Key Takeaways
- SKLICE holds a significant share of prescription pediculicide treatments, especially for resistant cases.
- The U.S. market is dominant, with potential growth in emerging markets.
- Prices remain high due to brand trust and efficacy, but patent expiration may lead to a substantial reduction in pricing.
- Competition from OTC and new prescription treatments influences future pricing strategies.
- Manufacturers should monitor patent status and resistance trends to project long-term viability.
Frequently Asked Questions
1. When will SKLICE patents expire?
Patent expiry is expected within 2-3 years, enabling generic entry.
2. How does resistance affect SKLICE demand?
Growing permethrin resistance increases demand for SKLICE when OTC treatments fail, sustaining higher prices temporarily.
3. What is the impact of generics on SKLICE pricing?
Introduction of generics could reduce prices by 30-50%, aligning with OTC permethrin costs.
4. Are insurance reimbursements likely to affect SKLICE prices?
Yes, higher reimbursement rates can make SKLICE more accessible and sustain demand, but they also influence net revenue.
5. Will new treatments replace SKLICE?
New prescription therapies like spinosad and dimethicone may capture market share, affecting SKLICE’s long-term pricing and sales.
References
[1] MarketResearch.com. (2023). Head lice treatment market analysis.
[2] FDA. (2012). Approval letter for SKLICE (permethrin lotion).
[3] Statista. (2023). Pediculosis treatment market size in the United States.
[4] Epic Research. (2022). Permethrin resistance trends in head lice.
[5] IQVIA. (2023). Pharmaceutical pricing and reimbursement data.
More… ↓
