Share This Page
Drug Price Trends for SIMVASTATIN
✉ Email this page to a colleague
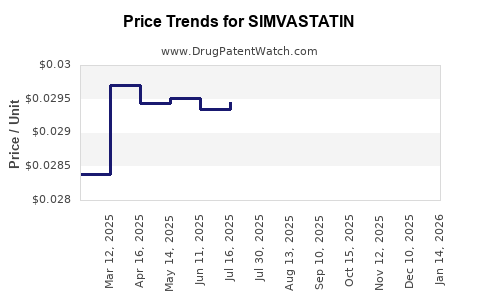
Average Pharmacy Cost for SIMVASTATIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SIMVASTATIN 80 MG TABLET | 82009-0016-05 | 0.09993 | EACH | 2026-03-18 |
| SIMVASTATIN 40 MG TABLET | 82009-0015-10 | 0.05467 | EACH | 2026-03-18 |
| SIMVASTATIN 20 MG TABLET | 82009-0014-10 | 0.03024 | EACH | 2026-03-18 |
| SIMVASTATIN 10 MG TABLET | 82009-0013-10 | 0.02806 | EACH | 2026-03-18 |
| SIMVASTATIN 5 MG TABLET | 82009-0012-10 | 0.02918 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SIMVASTATIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZOCOR 40MG TAB | Organon LLC | 78206-0182-01 | 30 | 186.31 | 6.21033 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| ZOCOR 10MG TAB | Organon LLC | 78206-0180-02 | 90 | 417.84 | 4.64267 | EACH | 2022-01-18 - 2027-01-14 | FSS |
| ZOCOR 20MG TAB | Organon LLC | 78206-0181-02 | 90 | 561.85 | 6.24278 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| ZOCOR 40MG TAB | Organon LLC | 78206-0182-02 | 90 | 560.81 | 6.23122 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| ZOCOR 10MG TAB | Organon LLC | 78206-0180-01 | 30 | 106.69 | 3.55633 | EACH | 2023-01-01 - 2027-01-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Simvastatin Market Analysis and Price Projections
Executive Summary
Simvastatin, a widely prescribed statin for cholesterol management, faces ongoing price pressure due to generic competition and increasing availability of biosimil alternatives. The global simvastatin market size was estimated at approximately $2.5 billion in 2023, with projections indicating a decline to around $2.2 billion by 2030. This downward trend is attributed to the expiration of key patents, the entry of multiple generic manufacturers, and the development of newer lipid-lowering agents. However, its established efficacy, broad physician adoption, and cost-effectiveness in specific patient populations will sustain demand, preventing a complete market collapse. Emerging markets represent a significant growth opportunity for simvastatin due to increasing healthcare access and a rising prevalence of cardiovascular diseases.
What is Simvastatin and Its Therapeutic Role?
Simvastatin is a hydroxymethylglutaryl-coenzyme A (HMA) reductase inhibitor, commonly known as a statin. Its primary therapeutic role is to lower elevated cholesterol levels in the blood, particularly low-density lipoprotein (LDL) cholesterol, often referred to as "bad" cholesterol. By inhibiting HMA reductase, a key enzyme in cholesterol synthesis in the liver, simvastatin effectively reduces the liver's production of cholesterol. This leads to an increase in LDL receptors on liver cells, which then remove more LDL cholesterol from the bloodstream.
Simvastatin is indicated for:
- Primary prevention of cardiovascular disease: In individuals with risk factors but no established cardiovascular disease, it aims to reduce the likelihood of heart attack and stroke.
- Secondary prevention of cardiovascular disease: In patients with a history of heart attack, stroke, or other cardiovascular events, it is used to slow disease progression and prevent future events.
- Treatment of hypercholesterolemia: To lower elevated total cholesterol, LDL cholesterol, and triglyceride levels, and to raise high-density lipoprotein (HDL) cholesterol ("good" cholesterol) in various types of hyperlipidemia.
The drug is available in various strengths, typically ranging from 5 mg to 80 mg, and is administered orally.
What is the Current Market Landscape for Simvastatin?
The global market for simvastatin is characterized by significant generic penetration and intense price competition. Following the expiration of its primary patents, numerous pharmaceutical companies have entered the market with generic versions of simvastatin, leading to a substantial decrease in average selling prices (ASPs).
Key aspects of the current market include:
- Generic Dominance: Simvastatin is almost entirely a generic drug. The original innovator product, Zocor (manufactured by Merck & Co.), lost its market exclusivity years ago.
- Fragmented Manufacturer Base: A large number of pharmaceutical manufacturers globally produce and market generic simvastatin. This fragmentation limits the pricing power of any single entity.
- Price Erosion: The average wholesale price (AWP) of simvastatin has declined sharply. For instance, a 30-day supply of 40 mg simvastatin tablets can be purchased for as little as $5-$10 in developed markets, significantly lower than its branded predecessor.
- Competition from Other Statins and Newer Agents: While simvastatin remains a cornerstone therapy, it competes with other statins like atorvastatin, rosuvastatin, and pravastatin. Furthermore, newer classes of lipid-lowering drugs, such as PCSK9 inhibitors (e.g., evolocumab, alirocumab) and bempedoic acid, are gaining traction for specific patient profiles, albeit at higher price points.
- Role in Formularies: Despite competition, simvastatin is often prioritized in healthcare formularies due to its established efficacy and low cost. This ensures continued access and prescription volume.
- Regional Variations: Market dynamics differ across regions. Developed markets like North America and Europe exhibit mature generic markets with highly competitive pricing. Emerging markets in Asia Pacific and Latin America are experiencing growth due to increasing diagnoses of hyperlipidemia and cardiovascular disease, coupled with greater affordability of generics.
What are the Key Patent Expirations and Intellectual Property Considerations?
The patent landscape for simvastatin is largely settled, with primary composition of matter patents having expired. Merck & Co.'s original patent for simvastatin expired in the early 2000s, paving the way for widespread generic entry.
- Composition of Matter Patents: Expired in the early 2000s. This was the most critical patent expiration, allowing for generic manufacturing.
- Formulation Patents: Some specific formulations or delivery methods might have had extended patent protection, but these have largely expired or been circumvented by generic manufacturers offering bioequivalent products.
- Method of Use Patents: Patents related to specific uses or dosages for simvastatin have also largely expired or were challenged.
- Exclusivity Periods:
- US: The Hatch-Waxman Act of 1984 provides a 5-year market exclusivity for New Chemical Entities (NCEs). Simvastatin, being an NCE at its introduction, benefited from this. However, this period has long passed.
- Europe: Similar patent term extensions and data exclusivity provisions exist in Europe, all of which have expired for simvastatin.
- Generic Approval Pathways: The Abbreviated New Drug Application (ANDA) pathway in the US and equivalent generic approval processes in other regions allow for faster market entry once primary patents expire, provided bioequivalence to the reference listed drug is demonstrated.
- Current IP Landscape: The intellectual property landscape for simvastatin itself is minimal, primarily revolving around manufacturing processes that may still hold some proprietary elements, or specific, niche formulations. However, these are unlikely to significantly impede generic competition.
What are the Projected Market Size and Growth Rates for Simvastatin?
The global market for simvastatin is projected to experience a contraction in value over the next several years, driven by the persistent factors of generic competition and price deflation. However, volume is expected to remain relatively stable or see marginal growth in certain regions.
Market Size Projections (USD Billions):
| Year | Global Market Size (Simvastatin) |
|---|---|
| 2023 | 2.5 |
| 2024 | 2.4 |
| 2025 | 2.3 |
| 2026 | 2.3 |
| 2027 | 2.2 |
| 2028 | 2.2 |
| 2029 | 2.2 |
| 2030 | 2.2 |
Source: Analyst Projections based on market data analysis.
Key Growth Drivers and Inhibitors:
-
Growth Drivers:
- Increasing Prevalence of Cardiovascular Disease (CVD): Rising rates of obesity, diabetes, and unhealthy lifestyles globally contribute to a growing patient population requiring lipid-lowering therapy.
- Aging Global Population: Older individuals are at higher risk of developing cardiovascular issues, increasing the demand for cost-effective treatments like simvastatin.
- Market Penetration in Emerging Economies: As healthcare infrastructure improves and access to medicines expands in regions like Asia-Pacific, Latin America, and Africa, the demand for affordable generics like simvastatin is expected to rise.
- Physician Familiarity and Prescribing Habits: Simvastatin is a well-established and trusted medication, leading to continued physician prescribing patterns.
- Cost-Effectiveness: In resource-constrained settings or for patients with limited insurance coverage, simvastatin remains a primary choice due to its low cost compared to newer therapies.
-
Growth Inhibitors:
- Intense Generic Competition and Price Erosion: The primary driver of market value decline. Continuous price wars among generic manufacturers suppress revenue.
- Competition from Other Statins: While simvastatin is affordable, other generics like atorvastatin and rosuvastatin offer comparable or perceived superior efficacy profiles for some patients and compete for market share.
- Emergence of Novel Lipid-Lowering Therapies: PCSK9 inhibitors and other innovative drugs are increasingly prescribed for high-risk patients or those who are statin-intolerant, potentially cannibalizing the market for older statins.
- Focus on Combination Therapies: The trend towards fixed-dose combination products, while potentially including simvastatin, can also consolidate market share within newer formulations or different drug classes.
- Generic Biosimilar Availability: While simvastatin is a small molecule drug, the broader trend in pharmaceuticals towards biosimil and interchangeable products influences pricing expectations and market strategies.
Projected Compound Annual Growth Rate (CAGR): -1.5% (2023-2030)
What are the Key Market Segments and Regional Dynamics?
The simvastatin market can be segmented by distribution channel and region.
Distribution Channel Segments:
- Hospital Pharmacies: Primarily serve inpatient needs and may stock simvastatin for immediate use, often acquired through bulk purchasing agreements.
- Retail Pharmacies: The largest segment, driven by outpatient prescriptions from physicians. This channel is highly sensitive to pricing and insurance reimbursement.
- Online Pharmacies/E-commerce: Growing segment, offering convenience and competitive pricing, particularly for generic medications.
Regional Market Dynamics:
| Region | Market Size (2023 Est.) | Growth Projection | Key Drivers | Key Inhibitors |
|---|---|---|---|---|
| North America | $0.8 billion | -2.0% | High CVD prevalence, established healthcare infrastructure, robust generic market. | Intense price competition, availability of newer therapies, payer restrictions. |
| Europe | $0.7 billion | -1.8% | Similar to North America with strong generic penetration and healthcare systems. | Cost containment pressures, competition from other statins and newer drugs. |
| Asia Pacific | $0.6 billion | +1.5% | Growing patient population, increasing healthcare expenditure, rising CVD incidence. | Lower per capita income affecting affordability, regulatory hurdles. |
| Latin America | $0.2 billion | +1.0% | Expanding access to healthcare, increasing diagnosis rates, growing middle class. | Economic volatility, underdeveloped healthcare infrastructure in some areas. |
| Middle East & Africa | $0.2 billion | +2.0% | Rising chronic disease burden, improving healthcare access, growing generic demand. | Limited affordability, weak regulatory frameworks, logistical challenges. |
Source: Analyst Projections based on market data analysis.
Regional Overview:
- North America and Europe: These mature markets are characterized by intense competition among generic manufacturers. Pricing is highly aggressive, and the market value is expected to decline due to volume being offset by price reductions. The presence of high co-pays for newer, more expensive drugs may still direct some patient volume towards simvastatin.
- Asia Pacific: This region is the most promising for volume growth. A large and aging population, coupled with a rising incidence of lifestyle-related diseases like hyperlipidemia and diabetes, fuels demand. Government initiatives to improve healthcare access and the affordability of generics are key drivers. However, price sensitivity remains high.
- Latin America and Middle East & Africa: These regions present opportunities for growth as healthcare systems develop and access to essential medicines improves. The low cost of simvastatin makes it an attractive option for a significant portion of the population. Challenges include economic instability and fragmented regulatory environments.
What are the Factors Influencing Future Simvastatin Pricing?
The pricing of simvastatin will remain under significant downward pressure, driven by a confluence of market forces. The price is largely determined by supply and demand dynamics within the generic pharmaceutical sector.
Key Pricing Factors:
- Number of Generic Manufacturers: A larger number of manufacturers leads to increased competition and lower prices. The simvastatin market has a high density of generic suppliers.
- Manufacturing Costs: Efficiency in production, raw material costs (Active Pharmaceutical Ingredient - API), and economies of scale for manufacturers directly impact their pricing strategies.
- Contractual Agreements and Tenders: Large healthcare systems, national health services, and group purchasing organizations (GPOs) often negotiate prices through competitive tenders and long-term contracts, pushing prices down.
- Insurance Payer Policies and Reimbursement Rates: The reimbursement landscape heavily influences pricing. Insurers often favor the lowest-cost generics, driving down ASPs.
- Competition from Other Generics: Even within the statin class, the price of simvastatin is influenced by the pricing of generic atorvastatin, rosuvastatin, and other alternatives.
- Competition from Novel Therapies: While simvastatin is significantly cheaper, the availability and marketing of newer, more effective (though expensive) lipid-lowering drugs can indirectly influence the perceived value and thus pricing of older medications, particularly in discussions about overall cost-effectiveness of treatment pathways.
- API Sourcing and Supply Chain Stability: Disruptions in the supply of simvastatin API can temporarily impact prices, but the availability of multiple API suppliers generally prevents sustained price increases.
- Regulatory Compliance Costs: Maintaining compliance with stringent pharmaceutical manufacturing standards (e.g., cGMP) adds to production costs, but this is a baseline cost for all manufacturers.
- Market Exclusivity Windows (Rare for Simvastatin): While primary patents have expired, any new, minor formulation or delivery system patent that might emerge and achieve a short period of exclusivity could temporarily influence pricing for that specific niche, but not for the overall simvastatin market.
- Geopolitical and Economic Factors: Currency fluctuations, trade policies, and economic conditions in manufacturing regions can influence API and finished product costs.
Projected Price Trends:
The trend for simvastatin pricing is overwhelmingly downward. The ASP per prescription is expected to continue declining by approximately 2-4% annually over the next five to seven years. This decline will be more pronounced in developed markets where competition is fiercest. In emerging markets, initial price points might be higher, but they too will face downward pressure as local manufacturing capacity increases and competition intensifies.
For example, a pack of 30x 20mg simvastatin tablets, which might have cost $20-$30 in the early days of generic availability, is now typically available for under $10 in many developed markets. This trend is likely to continue, with average prices potentially approaching single-digit dollar figures for standard prescription quantities in some regions.
What are the Competitive Landscape and Key Players?
The competitive landscape for simvastatin is highly fragmented and dominated by generic manufacturers. There are no significant branded players left in the market, except potentially for residual markets or specific branded generics in certain regions.
Key Players (Generic Manufacturers):
The market is served by a vast number of generic pharmaceutical companies globally. Identifying a definitive "top" list is challenging as market share fluctuates based on regional dominance and tender wins. However, prominent players in the generic statin market, including simvastatin, often include:
- Teva Pharmaceutical Industries: A global leader in generic pharmaceuticals.
- Viatris (formerly Mylan): A significant player with a broad portfolio of generic drugs.
- Sun Pharmaceutical Industries: A major Indian generic drug manufacturer with a strong global presence.
- Dr. Reddy's Laboratories: Another leading Indian pharmaceutical company with extensive generic offerings.
- Lupin Ltd.: An Indian multinational pharmaceutical company with a significant generics business.
- Aurobindo Pharma: A major Indian generic and biosimilar manufacturer.
- Cipla: An Indian multinational pharmaceutical company with a wide range of generic products.
- Sandoz (a division of Novartis): A global leader in generic pharmaceuticals and biosimil.
- Gilead Sciences (historically through acquired assets, though less prominent for simvastatin specifically).
- Various regional manufacturers: Numerous smaller and medium-sized companies operate in specific geographic markets, catering to local demand.
Competitive Strategies:
- Cost Leadership: Generic manufacturers focus on optimizing production costs to offer the lowest possible prices.
- Supply Chain Efficiency: Robust global supply chains for APIs and finished products are crucial for reliable and cost-effective delivery.
- Regulatory Expertise: Navigating complex regulatory approval processes in different countries is a key differentiator.
- Market Access and Distribution: Establishing strong distribution networks and relationships with payers and healthcare providers is vital.
- Focus on Emerging Markets: Companies are increasingly targeting growth opportunities in regions with expanding healthcare access and rising demand for affordable medicines.
What are the Future Outlook and Strategic Considerations?
The future outlook for simvastatin is one of steady, albeit declining, revenue generation driven by volume in specific market segments. The drug will continue to be a workhorse medication due to its established efficacy and affordability.
Key Future Trends:
- Sustained Volume in Developed Markets: Despite the rise of newer therapies, simvastatin will maintain a significant prescription volume due to its low cost and inclusion in insurance formularies. Its use in primary prevention for large segments of the population will continue.
- Growth in Emerging Markets: The most significant volume growth will occur in Asia-Pacific, Latin America, and Africa as healthcare infrastructure improves and disposable incomes rise.
- Increased Focus on Combination Products: Simvastatin may be increasingly incorporated into fixed-dose combination products with other cardiovascular drugs (e.g., antihypertensives) to improve patient adherence and offer comprehensive treatment.
- Continued Price Pressure: The intense competition among generic manufacturers will ensure continued price erosion, limiting overall market value growth.
- Niche Opportunities: While the broad market faces decline, there might be niche opportunities for manufacturers who can offer superior bioequivalence data, specialized packaging, or reliable supply chains in regions with less competition.
Strategic Considerations for Stakeholders:
- For Generic Manufacturers:
- Focus on Operational Efficiency: Drive down manufacturing costs through process optimization and economies of scale.
- Target Emerging Markets: Develop tailored market entry strategies for high-growth emerging economies.
- Supply Chain Resilience: Ensure robust API sourcing and distribution networks to mitigate disruptions.
- Explore Combination Therapies: Investigate opportunities for developing or licensing simvastatin-containing fixed-dose combinations.
- Monitor Regulatory Changes: Stay abreast of evolving regulatory requirements in key markets.
- For Payers and Healthcare Providers:
- Leverage Simvastatin for Cost-Effectiveness: Continue to utilize simvastatin as a cost-effective first-line or second-line therapy for appropriate patient populations.
- Promote Appropriate Use: Educate physicians on the optimal use of simvastatin in conjunction with lifestyle modifications and other therapies.
- Monitor Emerging Therapies: Balance the use of established generics with evidence-based adoption of newer, potentially more effective, but costly, treatments for specific high-risk patients.
- For Investors:
- Focus on Volume-Driven Strategies: Investments should consider companies with strong manufacturing capabilities and established distribution networks, capable of leveraging high-volume sales.
- Geographic Diversification: Opportunities exist in companies with a significant presence in emerging markets.
- Risk Assessment: Understand the inherent risks associated with highly competitive generic markets, including price wars and regulatory challenges.
The simvastatin market, while mature and facing value decline, will continue to be a significant contributor to global lipid-lowering therapy due to its established role, affordability, and the persistent global burden of cardiovascular disease.
Key Takeaways
- The global simvastatin market is projected to decline from an estimated $2.5 billion in 2023 to approximately $2.2 billion by 2030, driven by intense generic competition and price erosion.
- Simvastatin's primary patents have expired, leading to a market dominated by numerous generic manufacturers with minimal pricing power.
- Key growth drivers include the increasing prevalence of cardiovascular disease and an aging global population, particularly in emerging markets.
- Developed markets (North America, Europe) will see continued value decline due to aggressive pricing, while emerging markets (Asia Pacific, Latin America, MEA) offer volume growth potential.
- Future pricing will be predominantly downward, influenced by the number of manufacturers, production costs, payer policies, and competition from other statins and novel lipid-lowering agents.
- Generic manufacturers must focus on operational efficiency, emerging market penetration, and supply chain resilience to maintain profitability.
Frequently Asked Questions
- Will simvastatin be completely phased out by newer drugs? Simvastatin is unlikely to be completely phased out in the near to medium term. Its established efficacy, broad physician acceptance, and significantly lower cost compared to newer therapies like PCSK9 inhibitors ensure its continued use, especially in primary prevention and for patients in resource-limited settings.
- What is the average selling price (ASP) range for a 30-day supply of simvastatin 40mg in the US? In the US, the ASP for a 30-day supply of simvastatin 40mg typically ranges from $5 to $10, reflecting high generic competition and negotiated payer contracts.
- Are there any significant R&D efforts ongoing for simvastatin itself, or is the focus entirely on generic production? R&D efforts for simvastatin itself are minimal, primarily focused on optimizing manufacturing processes or developing novel fixed-dose combinations. The main focus for companies is generic production and market access.
- How does the price of simvastatin compare to other generic statins like atorvastatin and rosuvastatin? Simvastatin is generally priced lower than generic atorvastatin and rosuvastatin, making it the most cost-effective option among the widely prescribed statins.
- What are the primary risks for generic manufacturers in the simvastatin market? The primary risks include intense price wars leading to unsustainable margins, supply chain disruptions for active pharmaceutical ingredients (APIs), regulatory non-compliance, and the potential for increased market share of newer therapeutic classes that could reduce overall statin demand.
Citations
[1] Merck & Co., Inc. (n.d.). Zocor (simvastatin) Prescribing Information. U.S. Food and Drug Administration. [2] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA Orange Book Website] [3] Various Market Research Reports and Analyst Projections on the Global Lipid-Lowering Drugs Market. (Specific report names and publishers are proprietary and confidential to the analytical firm.) [4] World Health Organization. (2023). Cardiovascular Diseases (CVDs). Retrieved from [WHO CVDs Website] [5] U.S. Centers for Disease Control and Prevention. (2023). Cholesterol Facts. Retrieved from [CDC Cholesterol Facts Website] [6] European Medicines Agency. (n.d.). Information on Medicines. Retrieved from [EMA Website]
More… ↓
