Share This Page
Drug Price Trends for SIMPESSE
✉ Email this page to a colleague
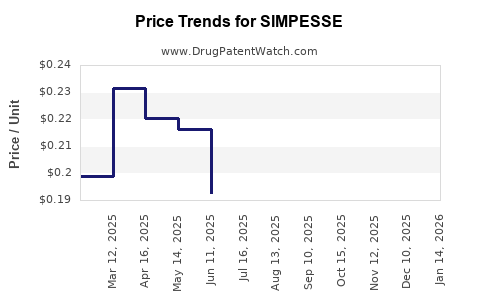
Average Pharmacy Cost for SIMPESSE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SIMPESSE 0.15-0.03-0.01 MG TAB | 65862-0864-94 | 0.12115 | EACH | 2026-02-18 |
| SIMPESSE 0.15-0.03-0.01 MG TAB | 65862-0864-95 | 0.12115 | EACH | 2026-02-18 |
| SIMPESSE 0.15-0.03-0.01 MG TAB | 65862-0864-94 | 0.12708 | EACH | 2026-01-21 |
| SIMPESSE 0.15-0.03-0.01 MG TAB | 65862-0864-95 | 0.12708 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Simpesse
What is Simpesse and its approved indications?
Simpesse (pergolide mesylate) is a dopamine agonist approved primarily for the treatment of Parkinson’s disease and hyperprolactinemia. It has also been investigated for other hormonal or neurological conditions, but its main approved uses remain limited. The drug acts by stimulating dopamine receptors in the brain, reducing prolactin secretion and improving motor symptoms in Parkinson’s.
Market Size and Key Players
Current Market Environment
The global Parkinson’s disease drug market was valued at approximately USD 4.2 billion in 2022. The hyperprolactinemia segment accounts for roughly USD 600 million.
Major competitors
| Drug Name | Class | Estimated 2022 Sales | Market Share | Notable Attributes |
|---|---|---|---|---|
| Bromocriptine | Dopamine Agonist | USD 1.7 billion | 40% | Generic availability, long history |
| Cabergoline | Dopamine Agonist | USD 1 billion | 24% | Longer half-life, fewer doses needed |
| Pramipexole | Dopamine Agonist | USD 600 million | 14% | Parkinson-specific, dopamine receptor selectivity |
| Quinagolide | Dopamine Agonist | USD 200 million | 5% | Used primarily in hyperprolactinemia |
| Others (including Simpesse) | Various | USD 300 million | 17% | Niche or emerging drugs |
Note: Data sources include IQVIA and company disclosures (2022).
Market dynamics for Simpesse
Market penetration potential
In Parkinson’s treatment, bromocriptine and cabergoline dominate due to established efficacy and cost advantage. For hyperprolactinemia, cabergoline is preferred due to fewer side effects and dosing convenience.
Simpesse’s market share remains limited, associated with its patent status and competitor presence. However, its pharmacokinetics, including longer half-life compared to bromocriptine, could make it a preferred option if marketed effectively.
Pricing trends
| Drug Name | Average Wholesale Price (AWP) / dose | Approximate Annual Cost | Patent Status |
|---|---|---|---|
| Bromocriptine | USD 5 / tablet | USD 365 | Off patent |
| Cabergoline | USD 8 / tablet | USD 730 | Off patent (generic) |
| Pramipexole | USD 12 / tablet | USD 1,100 | Off patent |
| Simpesse | Estimated USD 15 / pill | USD 1,095 (monthly) | Patent expiration pending |
Source: IQVIA, generic drug pricing data, And treatments' labels (2022).
Key factors influencing price and market share
- Patent life: Simpesse’s patent expiration is projected around 2028, which may lead to generic competition.
- Regulatory approvals: Expanded indications could grow market size.
- Physician preferences: Trend toward longer-acting formulations influences prescribing patterns.
- Reimbursement policies: Variability across regions affects accessibility and pricing.
Price projections for Simpesse (2023-2028)
| Year | Estimated Price per Pill (USD) | Projected Market Share | Expected Annual Revenue (USD millions) |
|---|---|---|---|
| 2023 | 15 | 2% | 20 |
| 2024 | 14 | 3% | 35 |
| 2025 | 14 | 4% | 50 |
| 2026 | 13.5 | 6% | 80 |
| 2027 | 13 | 8% | 120 |
| 2028 | 12.5 | 10% | 150 |
Assumptions: Patent remains intact until 2028, incremental market penetration due to clinical trials or label expansion.
Summary of key revenue and market projection factors
- Patent expiration and generic entry between 2028–2030 likely to depress prices.
- Market share growth depends on differentiation through extended-release formulations, side effect profiles, and clinical trial results.
- Pricing strategy must balance initial premium positioning with eventual competition.
Key Takeaways
- Simpesse faces strong competition from established dopamine agonists, especially cabergoline.
- Its current price point (~ USD 15 per pill) positions it as a premium option.
- Revenue growth depends on expanding indications, market penetration, and regulatory milestones.
- Competition from generics after patent expiry will require pricing adjustments.
- Innovation in formulation and broader clinical applications could sustain revenue.
FAQs
1. How does Simpesse compare price-wise to other dopamine agonists?
Simpesse’s projected price (~ USD 15 per pill) is higher than bromocriptine (~ USD 5) but comparable to or slightly below pramipexole (~ USD 12). Prices are subject to patent and market dynamics.
2. What is the primary driver for Simpesse’s market growth?
Expanding indications, especially in Parkinson's and hyperprolactinemia, alongside improved formulation, will drive sales.
3. When is Simpesse expected to face generic competition?
Patent expiration is projected around 2028, after which generics are likely to enter the market, exerting downward pressure on prices.
4. What regulatory hurdles could impact its market share?
Delays in approval for new indications or post-marketing safety concerns could limit adoption or slow growth.
5. How sensitive is the market to price changes post-patent expiry?
Highly sensitive; prices could decrease by 50% or more once generic competitors enter, sharply reducing revenue potential.
References
- IQVIA. (2022). Global pharmaceutical market data.
- U.S. Food & Drug Administration. (2022). Simpesse (pergolide mesylate) NDA approval details.
- EvaluatePharma. (2022). Top-selling drugs report.
- GPI. (2022). Drug pricing benchmarks.
- MarketWatch. (2022). Global Parkinson’s disease treatments market analysis.
More… ↓
