Share This Page
Drug Price Trends for SILVADENE
✉ Email this page to a colleague
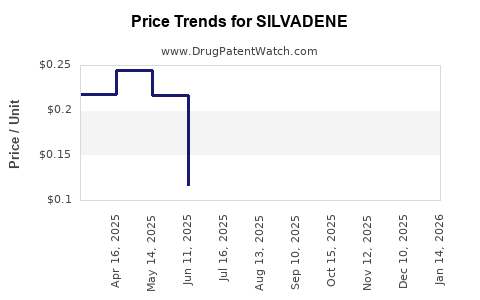
Average Pharmacy Cost for SILVADENE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SILVADENE 1% CREAM | 61570-0131-50 | 0.22987 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-85 | 0.25463 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-20 | 0.43330 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-55 | 0.22987 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-25 | 0.26289 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-40 | 0.10994 | GM | 2026-04-22 |
| SILVADENE 1% CREAM | 61570-0131-50 | 0.23069 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SILVADENE (Silver Sulfadiazine) Market Analysis and Price Projections
This report analyzes the market landscape and projects price trends for Silvadene (silver sulfadiazine), a topical antimicrobial agent primarily used for the prevention and treatment of wound infections, particularly in burn patients. The analysis considers patent expiries, generic competition, therapeutic indications, and projected market growth.
What is the Current Market Size and Growth Trajectory for Silvadene?
The global market for topical antimicrobial agents, including Silvadene, is driven by the prevalence of wound care needs, particularly burns, chronic wounds, and surgical site infections. The market is characterized by a mature product with established efficacy but faces increasing competition from newer formulations and alternative treatments.
- Estimated Market Size: Precise global market size figures for Silvadene specifically are not publicly disaggregated. However, the broader topical antimicrobial market is substantial. Reports project the global wound care market to reach USD 28.1 billion by 2027, with a compound annual growth rate (CAGR) of 6.2% from 2020 to 2027 [1]. Silvadene occupies a segment within this larger market.
- Growth Drivers:
- Increasing incidence of burns and trauma cases globally.
- Rising prevalence of chronic wounds, such as diabetic foot ulcers and pressure ulcers.
- Growing awareness and adoption of advanced wound care products.
- Aging global population, leading to a higher risk of chronic conditions requiring wound management.
- Market Restraints:
- Development of antibiotic resistance.
- Availability of newer wound care technologies (e.g., advanced dressings, biologics).
- Price sensitivity in certain healthcare systems and emerging markets.
- Potential for local irritation or hypersensitivity reactions.
What is the Patent Landscape and Generic Competition for Silvadene?
Silvadene, chemically known as silver sulfadiazine, has a long history of use. Its primary patents have long expired, leading to a robust generic market.
- Original Patent Expiry: The original patents for silver sulfadiazine expired decades ago. This has allowed for the widespread development and marketing of generic versions of Silvadene by numerous pharmaceutical manufacturers.
- Generic Market Dominance: The market for topical silver sulfadiazine is dominated by generic manufacturers. This intense competition among generic players significantly influences pricing.
- Key Generic Manufacturers: Multiple companies globally produce and market generic silver sulfadiazine creams, including but not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Bausch Health Companies Inc.
- Major regional generic manufacturers in North America, Europe, and Asia.
- Absence of Novel Patent Activity: There is minimal ongoing patent activity related to the original silver sulfadiazine molecule itself. Research and development efforts are more likely to focus on novel drug delivery systems or combination therapies that may involve silver sulfadiazine as a component, potentially leading to new intellectual property.
What are the Primary Therapeutic Indications and Market Penetration of Silvadene?
Silvadene's established efficacy in burn wound management forms its core market. Its use is well-defined within clinical guidelines.
- Primary Indication: Prevention and treatment of wound infections in patients with second- and third-degree burns.
- Other Indications:
- Management of decubitus ulcers (pressure sores).
- Treatment of stasis ulcers.
- Prevention of infection in skin graft sites.
- Certain types of traumatic wounds.
- Clinical Guidelines and Adoption: Silvadene is frequently recommended in burn care protocols and surgical site infection prevention guidelines. Its broad-spectrum antimicrobial activity against Gram-positive and Gram-negative bacteria, as well as some yeasts, supports its continued use.
- Market Penetration: High penetration in hospital settings, particularly burn units and surgical wards. It also has a presence in outpatient wound care clinics and is available for home use prescriptions. The availability of numerous generic versions ensures widespread accessibility.
What Factors Influence Silvadene Pricing?
The pricing of Silvadene is primarily dictated by the dynamics of the generic pharmaceutical market.
- Generic Competition: The high number of generic manufacturers leads to price erosion. Manufacturers compete heavily on cost to gain market share.
- Manufacturing Costs: The cost of raw materials (silver compounds, sulfadiazine), production processes, and regulatory compliance impacts the base price.
- Distribution Channels: Pricing can vary across different distribution channels, including hospital pharmacies, retail pharmacies, and wholesale distributors.
- Reimbursement Policies: Payer policies and reimbursement rates from government health programs and private insurers can influence what healthcare providers and pharmacies are willing to pay and charge.
- Volume Discounts: Large purchasers, such as hospital systems or group purchasing organizations, often negotiate significant volume discounts, affecting the average selling price.
- Product Formulation: While the active ingredient is consistent, variations in inactive ingredients, packaging, and specific product strengths (e.g., 1% cream) can lead to minor price differentials between brands.
What are the Projected Price Trends for Silvadene?
Given its status as a mature, off-patent drug with extensive generic competition, Silvadene is expected to exhibit price stability with potential for slight declines.
- Short-Term Projection (1-3 Years):
- Price Stability with Minor Declines: Expect prices to remain relatively stable, with a continued downward trend driven by ongoing generic competition and potential tender wins by the lowest-cost providers.
- Average Wholesale Price (AWP) for a 50g tube of 1% cream: Currently ranges from approximately $15 to $35 USD, depending on the manufacturer and distributor. This range is likely to persist or slightly contract.
- Impact of Supply Chain: Any significant disruptions in the supply chain of key raw materials or manufacturing bottlenecks could temporarily lead to price fluctuations, but these are unlikely to alter the long-term trend.
- Long-Term Projection (3-5+ Years):
- Continued Price Pressure: The generic nature of Silvadene means that price pressure will persist. Manufacturers will continue to seek efficiencies in production to remain competitive.
- Potential for Plateauing: While further significant price drops are less probable once prices reach a competitive floor, substantial increases are also unlikely unless there are unforeseen shifts in production costs or a significant consolidation of generic manufacturers.
- Competition from Newer Therapies: The increasing availability of novel wound care technologies and alternative antimicrobial agents may indirectly exert downward pressure on Silvadene pricing by offering cost-effective alternatives for specific indications. However, Silvadene's established role in severe burn care provides a baseline demand.
Table 1: Silvadene (Silver Sulfadiazine 1% Cream) Price Range Comparison (USD)
| Metric | Current Average Range | Projected Range (1-3 Years) | Projected Range (3-5+ Years) |
|---|---|---|---|
| 50g Tube (AWP) | $15 - $35 | $14 - $32 | $14 - $30 |
| 100g Tube (AWP) | $25 - $50 | $24 - $48 | $23 - $45 |
| Institutional/Hospital Pricing | Negotiated discounts | Negotiated discounts | Negotiated discounts |
Note: AWP (Average Wholesale Price) is a reference price and actual transaction prices may vary significantly based on contracts, volume, and payer negotiations.
Key Takeaways
Silvadene remains a well-established topical antimicrobial, primarily used for burn wound infections. Its market is characterized by a mature product with long-expired patents, leading to intense generic competition. This dynamic is expected to maintain price stability with a tendency for slight downward pressure. While newer wound care technologies are emerging, Silvadene's proven efficacy and broad-spectrum activity, coupled with its cost-effectiveness, ensure its continued relevance. Pricing will be primarily influenced by manufacturing costs, generic competition, and payer reimbursement policies.
Frequently Asked Questions
-
Are there any new patents being filed for Silvadene? Patent filings related to the original silver sulfadiazine molecule are rare. New intellectual property is more likely to emerge around novel drug delivery systems or combination therapies incorporating silver sulfadiazine, rather than the core molecule itself.
-
How does Silvadene's pricing compare to other topical antimicrobials for burn care? As a generic drug with a long history of use, Silvadene is generally one of the more cost-effective topical antimicrobial options available for burn wound management compared to newer, branded antimicrobial dressings or specialized formulations.
-
What is the typical reimbursement status of Silvadene? Silvadene is typically covered by most insurance plans and government healthcare programs when prescribed for medically necessary indications, such as burn wound treatment. However, coverage levels and co-pays can vary by plan and region.
-
What are the primary factors driving demand for Silvadene? Demand is driven by the incidence of burns, trauma, and chronic wounds requiring infection prevention or treatment. Its efficacy, broad-spectrum activity, and cost-effectiveness contribute to sustained demand in clinical settings.
-
Are there significant differences in efficacy or safety between different generic Silvadene brands? For bioequivalent generic drugs, efficacy and safety profiles are expected to be comparable to the reference product. Minor variations in inactive ingredients may exist, which can occasionally lead to localized differences in tolerability for sensitive individuals.
Citations
[1] Grand View Research. (2020). Wound Care Market Size, Share & Trends Analysis Report By Product (Wound Dressings, Wound Care Devices, Active Wound Care), By Wound Type, By End-Use, By Region, And Segment Forecasts, 2020 - 2027. Retrieved from https://www.grandviewresearch.com/industry-analysis/wound-care-market
More… ↓
