Share This Page
Drug Price Trends for SEVELAMER
✉ Email this page to a colleague
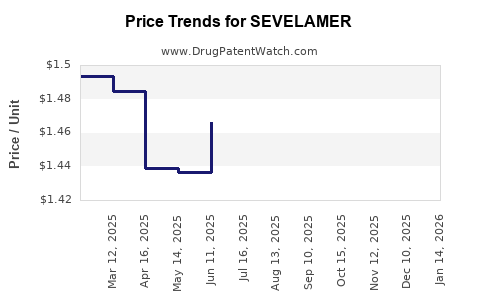
Average Pharmacy Cost for SEVELAMER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SEVELAMER 0.8 GM POWDER PACKET | 33342-0288-68 | 1.25530 | EACH | 2026-04-22 |
| SEVELAMER 0.8 GM POWDER PACKET | 00115-1365-30 | 1.25530 | EACH | 2026-04-22 |
| SEVELAMER 0.8 GM POWDER PACKET | 00115-1365-29 | 1.25530 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SEVELAMER
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SEVELAMER CARBONATE 2.4GM/PKT PWDR | AvKare, LLC | 69452-0127-19 | 90 | 132.04 | 1.46711 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SEVELAMER HCL 800MG TAB | AvKare, LLC | 68462-0447-18 | 180 | 307.27 | 1.70706 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SEVELAMER CARBONATE 0.8GM/PKT PWDR | AvKare, LLC | 69452-0126-19 | 90 | 137.67 | 1.52967 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
SEVELAMER: PATENT LANDSCAPE AND MARKET FORECAST
This analysis examines the patent landscape and market projections for sevelamer, a phosphate binder used to treat hyperphosphatemia in patients with chronic kidney disease (CKD). Key patents covering sevelamer carbonate and hydrochloride formulations are nearing expiration or have expired, opening opportunities for generic competition. Market growth is projected to be moderate, driven by increasing CKD prevalence and the accessibility of lower-cost generic options.
What is the Current Patent Status of Sevelamer?
The primary active pharmaceutical ingredient in sevelamer is a non-absorbed polymer. Patents have historically focused on specific salt forms, formulations, and manufacturing processes.
- Sevelamer Hydrochloride (Renagel®): Patents for the original sevelamer hydrochloride formulations have expired in major markets. For example, U.S. Patent No. 5,496,847, which claimed sevelamer hydrochloride, expired in 2016.
- Sevelamer Carbonate (Renvela®): Sevelamer carbonate is a distinct salt form. Key patents protecting sevelamer carbonate formulations and methods of use are also approaching or have passed their expiration dates. U.S. Patent No. 7,205,303, which claims sevelamer carbonate, expired in 2021. Supplementary Protection Certificates (SPCs) or Patent Term Extensions (PTEs) in various jurisdictions may have extended market exclusivity for specific formulations beyond the original patent expiry dates. For instance, SPCs in Europe for Renvela® have expired in key countries.
- Manufacturing Processes and Polymorphs: While composition of matter patents have expired or are expiring, secondary patents related to specific crystalline forms (polymorphs), manufacturing processes, and novel delivery systems may still be in effect for some manufacturers. These can create barriers to entry for generic competitors attempting to replicate a specific product. However, the core polymer and its therapeutic utility are no longer protected by primary patents.
Who are the Major Market Players for Sevelamer?
The market for sevelamer is transitioning from brand-name dominance to a more competitive generic landscape.
- Brand Name Manufacturers: Genzyme (a Sanofi company) is the originator of both Renagel® (sevelamer hydrochloride) and Renvela® (sevelamer carbonate).
- Generic Manufacturers: Several generic companies have entered or are preparing to enter the market. These include, but are not limited to:
- Teva Pharmaceuticals
- Viatris (formerly Mylan and Upjohn)
- Sun Pharmaceutical Industries
- Torrent Pharmaceuticals
- Dr. Reddy's Laboratories
- Apotex
- Market Share Shift: With patent expiries, market share is increasingly shifting towards generics due to price competition. The availability of multiple generic suppliers intensifies this dynamic.
What are the Projected Market Sizes and Growth Rates for Sevelamer?
The global sevelamer market is influenced by the prevalence of CKD and the economics of phosphate binder treatment.
- Market Size: The global sevelamer market was estimated to be between $1.5 billion and $1.8 billion in 2023. This figure encompasses both branded and generic sales.
- Growth Drivers:
- Increasing CKD Prevalence: The global incidence and prevalence of CKD are rising due to factors such as diabetes, hypertension, and an aging population. This expands the patient pool requiring phosphate binders.
- Generic Availability: The widespread availability of lower-cost generic sevelamer products is increasing demand, particularly in price-sensitive markets.
- Therapeutic Indication: Sevelamer remains a first-line therapy for hyperphosphatemia in many CKD patients, particularly those on dialysis, due to its non-systemic absorption and favorable safety profile compared to some calcium-based binders.
- Projected Growth Rate: The market is projected to experience a compound annual growth rate (CAGR) of 3% to 5% from 2024 to 2029. This moderate growth reflects the maturity of the product, the competitive pricing environment, and the ongoing expansion of the CKD patient population.
- Regional Variations: Growth rates will vary by region, with higher growth anticipated in emerging markets due to increasing healthcare access and diagnosis rates, while developed markets will see more stable, albeit slower, growth driven by patient volume.
What is the Pricing Landscape for Sevelamer?
Pricing for sevelamer is characterized by a significant disparity between branded and generic versions.
- Branded Pricing: Original branded sevelamer products (Renagel® and Renvela®) command premium pricing, reflecting R&D investment and market exclusivity. Prices can range from $15 to $40 per daily dosage, depending on formulation, dosage strength, and market.
- Generic Pricing: Generic sevelamer products are priced substantially lower. The introduction of multiple generic competitors has led to price erosion. Generic prices typically range from $3 to $10 per daily dosage, representing a discount of 60% to 85% compared to branded equivalents.
- Factors Influencing Generic Pricing:
- Number of Suppliers: A higher number of generic manufacturers generally leads to more aggressive pricing.
- Manufacturing Costs: Efficiency in API synthesis and formulation significantly impacts cost of goods and subsequent pricing.
- Reimbursement Policies: Payer formularies and reimbursement rates influence the effective price paid by healthcare systems and patients.
- Competition with Other Phosphate Binders: Sevelamer competes with other classes of phosphate binders, such as calcium-based binders, lanthanum carbonate, and iron-based binders. The pricing of these alternatives also influences sevelamer's price positioning.
What are the Key Therapeutic Uses and Market Drivers for Sevelamer?
Sevelamer's primary indication and market position are well-established.
- Primary Therapeutic Use:
- Hyperphosphatemia in Chronic Kidney Disease (CKD): Sevelamer is indicated for the control of elevated serum phosphorus levels in adult and pediatric patients (aged 6 years and older) with CKD on hemodialysis or peritoneal dialysis.
- Mechanism of Action: It is a non-absorbed polymer that binds to dietary phosphate in the gastrointestinal tract, preventing its absorption and subsequent elevation in serum phosphate levels.
- Market Drivers:
- High Prevalence of CKD: The global burden of CKD, particularly among diabetic and hypertensive patients, directly drives demand.
- Dialysis Patient Population: Patients undergoing hemodialysis and peritoneal dialysis are at high risk for hyperphosphatemia and are primary candidates for phosphate binders. This segment represents a substantial portion of the sevelamer market.
- Physician Preference and Clinical Guidelines: Sevelamer, especially the carbonate formulation, is often favored due to its efficacy and the avoidance of calcium loading, which can contribute to vascular and soft tissue calcification. Clinical guidelines continue to recommend it as a first-line option.
- Insurance and Payer Coverage: Broad insurance coverage and inclusion on formularies for both branded and generic sevelamer ensure patient access.
- Long-Term Management of CKD: As CKD is a chronic condition, continuous phosphate management is required, creating a sustained demand for effective binders like sevelamer.
What are the Competitive Threats and Future Outlook for Sevelamer?
The competitive landscape is evolving, with potential threats and opportunities.
- Generic Erosion: The primary competitive threat is the continued price erosion driven by an increasing number of generic manufacturers. This will likely suppress overall market value growth.
- Competition from Other Phosphate Binders:
- Calcium-based Binders (e.g., Calcium Acetate, Calcium Carbonate): These are generally less expensive but carry the risk of calcium accumulation, which can lead to hypercalcemia and vascular calcification.
- Lanthanum Carbonate (Fosrenol®): A metal-based binder that competes with sevelamer.
- Iron-based Binders (e.g., Ferric Citrate, Sucroferric Oxyhydroxide): These newer agents offer potential benefits such as iron supplementation and good phosphate binding, posing a competitive challenge. Ferric citrate, in particular, has gained market share in certain regions.
- Pipeline Drugs: While the sevelamer market is mature, the development of novel phosphate binders or alternative CKD management strategies could eventually impact its long-term market share. However, no immediate disruptors are anticipated to displace sevelamer in the near to medium term.
- Future Outlook: The sevelamer market will likely remain a significant, albeit mature, segment within the CKD therapeutics space. Volume growth will be driven by CKD prevalence, while value growth will be constrained by generic competition and pricing pressures. Manufacturers focusing on cost-efficient production and market access will be best positioned.
Key Takeaways
- Primary patents for sevelamer hydrochloride and carbonate have expired or are expiring, leading to increased generic competition.
- The market size for sevelamer is substantial, estimated at $1.5 billion to $1.8 billion in 2023.
- Projected market growth is moderate, at 3% to 5% CAGR from 2024 to 2029, driven by rising CKD prevalence and generic accessibility.
- Significant price differences exist between branded ($15-$40/day) and generic ($3-$10/day) sevelamer, with generics dominating volume.
- The increasing prevalence of CKD and its management, particularly in dialysis patients, are key market drivers.
- Competitive threats include a growing number of generic suppliers and the availability of alternative phosphate binders, such as iron-based agents.
Frequently Asked Questions
What is the difference between sevelamer carbonate and sevelamer hydrochloride?
Sevelamer carbonate and sevelamer hydrochloride are different salt forms of the same active polymer. Sevelamer carbonate has shown a lower incidence of metabolic acidosis compared to sevelamer hydrochloride in clinical studies, leading to its preferential use in many treatment guidelines.
How does generic competition impact sevelamer pricing?
The entry of multiple generic manufacturers significantly lowers the price of sevelamer due to increased supply and competitive pressures. This typically leads to a reduction in overall market value despite stable or increasing unit volumes.
Are there any new patents being issued for sevelamer formulations?
While primary composition of matter patents are expired, companies may still pursue patents for novel formulations, delivery systems, specific polymorphs, or manufacturing processes. These secondary patents could offer limited periods of exclusivity for specific branded or generic products.
What is the impact of kidney transplant on sevelamer demand?
Successful kidney transplantation can reduce or eliminate the need for phosphate binders, as kidney function improves. Therefore, factors influencing transplant rates, such as organ availability and transplant success, can indirectly affect the long-term demand for sevelamer.
How do iron-based phosphate binders compare to sevelamer in terms of market competition?
Iron-based phosphate binders represent a significant competitive threat to sevelamer. They offer dual benefits of phosphate binding and iron supplementation, which is beneficial for anemic CKD patients. This dual action, coupled with efficacy, has led to their increasing adoption and market share gains at the expense of older binders like sevelamer in some patient populations.
Citations
[1] U.S. Patent No. 5,496,847. (1996). Polymer phosphate binders. United States Patent Office. [2] U.S. Patent No. 7,205,303. (2007). Polymer phosphate binders. United States Patent Office. [3] Pharmaceutical market research reports and industry databases (2023-2024). [4] Clinical guidelines and comparative drug studies on hyperphosphatemia management in CKD.
More… ↓
