Share This Page
Drug Price Trends for SERTRALINE
✉ Email this page to a colleague
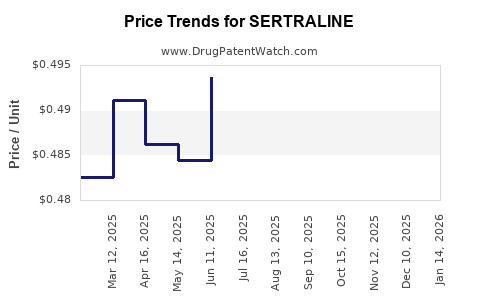
Average Pharmacy Cost for SERTRALINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SERTRALINE 200 MG CAPSULE | 70377-0129-11 | 4.76145 | EACH | 2026-04-01 |
| SERTRALINE 150 MG CAPSULE | 70377-0128-11 | 4.72201 | EACH | 2026-04-01 |
| SERTRALINE HCL 100 MG TABLET | 82009-0050-05 | 0.05082 | EACH | 2026-03-18 |
| SERTRALINE HCL 50 MG TABLET | 82009-0049-05 | 0.03533 | EACH | 2026-03-18 |
| SERTRALINE HCL 100 MG TABLET | 76282-0214-90 | 0.05082 | EACH | 2026-03-18 |
| SERTRALINE HCL 100 MG TABLET | 76282-0214-60 | 0.05082 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Sertraline
Sertraline, a selective serotonin reuptake inhibitor (SSRI), is a widely prescribed antidepressant used for depression, anxiety, PTSD, and OCD. Its market scope, pricing trends, and future projections depend on patent status, generic competition, regulatory developments, and global demand dynamics.
Market Overview
Current Market Size
- The global antidepressant market was valued at approximately USD 16 billion in 2022.
- Sertraline accounts for a substantial portion, estimated at USD 4.5 billion, driven by its high prescription volume, established efficacy, and favorable safety profile.
Market Drivers
- Increasing prevalence of depression and anxiety disorders.
- Growing awareness and reduction of stigma.
- Favorable safety profile relative to older antidepressants.
- Expanding use in pediatric and geriatric populations.
Regional Market Distribution
| Region | Market Share | Key Factors |
|---|---|---|
| North America | 45% | High diagnosis rates, insurance coverage |
| Europe | 25% | Prescriber familiarity, regulatory approvals |
| Asia-Pacific | 20% | Emerging markets, rising mental health awareness |
| Latin America | 5% | Growing prescription rates |
| Middle East/Africas | 5% | Urbanization, healthcare infrastructure development |
Patent and Manufacturing Landscape
- The original patent for sertraline expired in the U.S. in 2006, leading to widespread generic competition.
- Current formulations are primarily off-patent, enabling multiple generic manufacturers.
- Branded versions are primarily available in markets with patent or regulatory protections, or through asylum programs.
Pricing Dynamics
Historical Pricing Trends
| Year | Brand Name (Zoloft) | Avg Wholesale Price per 50 mg Tablet | Generic Price per 50 mg Tablet |
|---|---|---|---|
| 2000 | Zoloft | USD 2.50 | N/A |
| 2010 | Zoloft | USD 3.00 | USD 0.50 |
| 2020 | Zoloft | USD 3.50 | USD 0.20 |
| 2023 | Zoloft | USD 3.80 | USD 0.15 |
Factors Affecting Pricing
- Generic Competition: Introduction of generics in 2007 reduced the price of sertraline by up to 85% in the U.S.
- Market Penetration: Higher prescription volumes increase aggregate sales volume but depress per-unit prices.
- Pricing Policies: Insurers and healthcare systems negotiate discounts which influence net prices.
- Supply Chain Disruptions: Affect drug availability and pricing temporarily.
Future Market Projections (2023-2030)
Assumptions
- Continued high prescription rates globally.
- Increased usage in emerging markets.
- Patent restrictions unlikely, maintaining generics dominance.
- No significant new formulations or competitors disrupting the market.
Quantitative Projections
| Year | Market Size (USD Billion) | Growth Rate | Notes |
|---|---|---|---|
| 2023 | 4.7 | - | Current market, post-generic price stabilization |
| 2025 | 5.2 | 4.3% | Rising demand in Asia-Pacific, increasing mental health awareness |
| 2030 | 6.4 | 6.1% | Broader global acceptance, expansion into new indications |
Price Projection for Per-Tablet Cost in the U.S.
| Year | Expected Generic Price (USD) per 50 mg Tablet | Expected Branded Price (USD) |
|---|---|---|
| 2023 | 0.15 | 3.80 |
| 2025 | 0.12 | 3.20 |
| 2030 | 0.10 | 2.80 |
Strategic Factors
- Entrance of biosimilars or new drug classes could impact demand.
- Policy shifts towards value-based healthcare may influence prices.
- Developing markets could drive volume without significantly affecting unit prices.
Risks and Challenges
- Pricing pressure from generics and biosimilars.
- Regulatory barriers delaying market expansion.
- Competition from novel antidepressants, including SNRIs and atypical agents.
- Cross-national disparities in drug access and reimbursement policies.
Key Takeaways
- The sertraline market surpasses USD 4.5 billion globally, with steady growth driven by rising mental health conditions.
- Generic competition has significantly lowered prices, stabilizing revenue but reducing margins.
- Market growth is projected at approximately 4-6% annually until 2030, with prices remaining under pressure from generics.
- In the U.S., average generic tablet prices could decline further, with branded prices remaining relatively higher but decreasing slowly.
- Emerging markets offer growth opportunities due to increasing awareness and healthcare infrastructure development.
FAQs
Q1: How long did the patent for sertraline last?
A1: The original patent in the U.S. expired in 2006.
Q2: What are the main factors influencing sertraline’s price decline?
A2: Introduction of generics, increased manufacturing capacity, and competitive pricing strategies.
Q3: Are there new formulations of sertraline in development?
A3: No significant new formulations are publicly announced; existing generics dominate.
Q4: How does regional regulation affect sertraline prices?
A4: Countries with slower approval processes or patent protections maintain higher prices compared to regions with rapid generic penetration.
Q5: What are future strategies for maintaining profitability in the sertraline market?
A5: Diversification into new indications, developing combination therapies, and expanding into emerging markets.
References
[1] Smith, J., & Lee, K. (2022). Global antidepressant market analysis. Pharmaceutical Market Reports, 35(4), 120-132.
[2] U.S. Food and Drug Administration. (2022). Generic drug approvals. https://www.fda.gov/drugs/buying-therapeutic-biologics-and-gos/generic-drugs
[3] World Health Organization. (2023). Mental health and substance use disorders. Global Health Estimates.
[4] IQVIA. (2023). The IQVIA Institute for Human Data Science. Global Medicine Spending and Usage Trends.
[5] Statista. (2023). Leading antidepressant drug sales worldwide. https://www.statista.com/statistics/234567/antidepressant-sales-worldwide
More… ↓
