Share This Page
Drug Price Trends for SERTRALINE HCL
✉ Email this page to a colleague
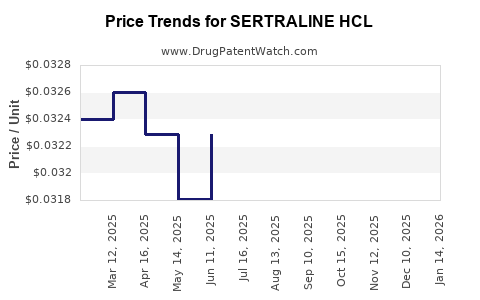
Average Pharmacy Cost for SERTRALINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SERTRALINE HCL 100 MG TABLET | 82009-0050-05 | 0.05082 | EACH | 2026-03-18 |
| SERTRALINE HCL 50 MG TABLET | 82009-0049-05 | 0.03533 | EACH | 2026-03-18 |
| SERTRALINE HCL 100 MG TABLET | 76282-0214-90 | 0.05082 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SERTRALINE HCL Market Analysis and Financial Projection
What Is SERTRALINE HCL, and Why Is It Significant in the Market?
Sertraline hydrochloride (HCL) is an SSRI (selective serotonin reuptake inhibitor) primarily prescribed for depression, anxiety, PTSD, and OCD. It is marketed under brands like Zoloft (Pfizer), and its generic versions are widely available.
The global antidepressant market, including SSRIs like sertraline, is valued at over $15 billion as of 2022 and is projected to grow at a CAGR of approximately 2.8% through 2027[1]. The demand is driven by increasing mental health awareness and aging populations.
What Are Current Market Dynamics and Key Players?
Market Share Distribution
- Pfizer (Zoloft): Historically held over 50% of the branded market but has gradually lost share owing to generic competition.
- Generics: Account for approximately 70-80% of sales globally, with multiple manufacturers offering bioequivalent products.
- Emerging Markets: Exhibit rapid growth, especially in Asia-Pacific, driven by increasing psychiatric treatment access.
Patent and Regulatory Status
- Patent Expiry: Pfizer's patent for Zoloft expired around 2006 in the U.S., leading to a proliferation of generics.
- Regulatory Approvals: Generics are approved through abbreviated pathways, reducing R&D barriers but intensifying price competition.
What Are Price Trends and Forecasts?
Current Pricing Landscape
| Product Type | Approximate Retail Price (per 30-count 50mg tablet) | Competitive Market Share |
|---|---|---|
| Brand (Zoloft) | $600–$900 (in the U.S.) | 20-30% |
| Generic (Bioequivalent) | $10–$20 | 70-80% |
Factors Affecting Pricing
- Patent status: Past patent expiry led to a price drop for generics.
- Manufacturing volume: Increased competition has driven costs down.
- Market penetration in emerging economies: Lower prices support broader access.
Price Projections (2023–2030)
Market analysts forecast the following for sertraline offerings:
- Generics: Prices will remain stable or decline marginally, given market saturation.
- Brand (Zoloft): Prices may decline by ~10% annually due to generics' dominance but could persist in niche markets or premium formulations.
Overall, in the U.S., retail prices are expected to stay within the current range but with potential for further reductions, especially as market access improves and competition intensifies.
How Will Market Trends Affect Pricing and Supply?
- Increased Generic Competition: Will sustain downward pressure on prices.
- Pricing Strategies: Manufacturers may offer discount programs or biosimilars to retain market share.
- Regulatory Changes: Policies encouraging biosimilar and generic use could further suppress prices.
- Market Expansion: Entry into developing markets could lead to volume increases, offsetting unit price declines.
What Are Future Market Opportunities and Risks?
Opportunities
- Development of new formulations (e.g., extended-release).
- Integration with digital health platforms.
- Expansion in regions with rising mental health awareness.
Risks
- Regulatory hurdles for new delivery systems.
- Price control policies in major markets.
- Competition from newer antidepressants with different efficacy profiles.
What Are Key Takeaways?
- The eroding patent protection for sertraline has resulted in a highly competitive generics market.
- Prices for generic sertraline are expected to remain stable or decline slightly through 2030.
- The branded market (Zoloft) persists as a niche, primarily in select regions or formulations.
- The combination of regulatory pressure, market saturation, and manufacturing efficiencies limits significant price increases.
- Emerging markets represent potential for volume growth but at lower price points.
FAQs
1. When did the patent for Zoloft expire, and how has that affected the market?
The patent expired around 2006 in the U.S., enabling numerous generic manufacturers to enter, leading to significant price decreases and increased market competition.
2. What are the primary factors driving the price of generic sertraline?
Manufacturing costs, patent status, market competition, regulatory policies, and regional economic factors influence generic prices.
3. How does the price of branded Zoloft compare to generics?
Branded Zoloft typically costs 30-50 times more than generics on a per-tablet basis in the U.S.
4. Are there opportunities for new formulations or delivery methods of sertraline?
Yes. Extended-release formulations or combination therapies are areas being explored, potentially commanding premium pricing.
5. What factors could disrupt current market price projections?
Regulatory changes, patent litigations, or the development of alternative therapies with superior efficacy could impact prices.
References
[1] MarketWatch, "Global Antidepressants Market," 2022.
More… ↓
