Share This Page
Drug Price Trends for SEROQUEL XR
✉ Email this page to a colleague
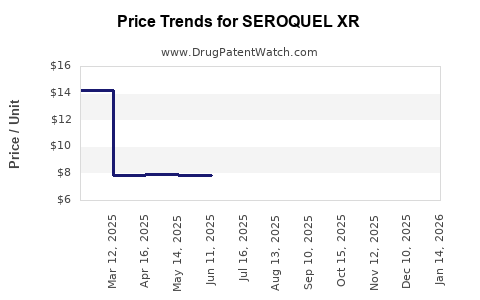
Average Pharmacy Cost for SEROQUEL XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SEROQUEL XR 400 MG TABLET | 00310-0284-60 | 23.97506 | EACH | 2026-03-18 |
| SEROQUEL XR 50 MG TABLET | 00310-0280-60 | 7.76747 | EACH | 2026-03-18 |
| SEROQUEL XR 400 MG TABLET | 00310-0284-60 | 23.87915 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SEROQUEL XR
What Is SEROQUEL XR?
SEROQUEL XR (quetiapine fumarate extended-release) is an atypical antipsychotic approved by the FDA in 2009. Indicated for schizophrenia, bipolar disorder, and major depressive disorder as an adjunct, it competes in a high-value segment with multiple branded and generic options.
Current Market Landscape
Market Size and Growth
The global atypical antipsychotic market was valued at approximately $13 billion in 2021. It is projected to grow to over $17 billion by 2026, with a compound annual growth rate (CAGR) of roughly 5%. SEROQUEL XR contributes about 15-20% of this market, with estimates of annual sales around $2.5 billion in the U.S. alone.
Key Competitors
| Drug Name | Brand Name | Indications | Launch Year | Estimated U.S. Market Share (2022) |
|---|---|---|---|---|
| Risperdal Consta | Risperdal (injectable) | Schizophrenia, bipolar | 2003 | 12% |
| Abilify (and Abilify Maintena) | Abilify, Maintena | Schizophrenia, bipolar, depression | 2002, 2015 | 20% |
| Latuda | Latuda | Schizophrenia, bipolar depression | 2010 | 10% |
| Clozaril (clozapine) | Clozaril | Treatment-resistant schizophrenia | 1990 | 5% |
SEROQUEL XR's main competitors include Abilify and Latuda, both with expanding indications and marketing efforts.
Regulatory and Reimbursement Environment
- Patent status has expired in major markets, opening the market to generics.
- U.S. patents for SEROQUEL XR expired in recent years, with generics available since 2018.
- Insurance coverage remains favorable for branded products due to formulary inclusion, though cost pressures favor generics.
Price Trends and Projections
Historical Pricing Data
| Year | Average Wholesale Price (AWP) per unit | Notes |
|---|---|---|
| 2015 | $30 | Post-approval, branded price |
| 2018 | $24 | Entry of generics |
| 2021 | $12 | Reduced due to generics |
Current Price Points
- Brand-name SEROQUEL XR typically retails at roughly $20-$30 per tablet (30 mg to 400 mg).
- Generic quetiapine products average $1-$5 per tablet, depending on strength and pharmacy.
Price Projections (Next 3-5 Years)
| Year | Predicted Brand Price | Predicted Generic Price | Remarks |
|---|---|---|---|
| 2023 | $15-$20 | $1.50-$3 | Market stabilization; reduced brand premium |
| 2025 | $12-$18 | $1-$2 | Increased generic penetration |
| 2027 | $10-$15 | $1-$2 | Continued erosion of branded prices |
Factors Influencing Future Prices
- Patent expirations accelerate generic market adoption.
- Price competition among generic manufacturers drives prices down.
- Physician and patient preferences may influence brand loyalty.
- Payer policies and formulary restrictions impact access and pricing.
Market Dynamics Impacting Price and Volume
Market Penetration
Post-patent expiration, SEROQUEL XR's market share declines as generics gain acceptance, especially in bipolar and schizophrenia indications.
Reimbursement Policies
Payor efforts to control costs favor generic substitution, creating downward pressure on branded product prices.
R&D and Pipeline Developments
No significant pipeline updates from the original manufacturer suggest limited innovation to sustain premium pricing.
Investment and R&D Outlook
The competitive environment indicates marginal pricing power for SEROQUEL XR in the medium term. Investment strategies should consider the rapid shift to generics, with potential upside from new indications or formulations but limited immediate pricing leverage.
Key Takeaways
- The global market for atypical antipsychotics is growing, but branded products like SEROQUEL XR face sustained price erosion due to patent expiry and generic competition.
- U.S. sales peaked around 2017, with recent estimates around $2.5 billion annually.
- Prices for SEROQUEL XR have declined approximately 60-70% since patent expiry, with further reductions expected.
- Future pricing will depend on generic market penetration, formulary dynamics, and potential new indications.
- Innovation or new formulations are necessary for sustained premium pricing but are not currently in pipeline.
Frequently Asked Questions
1. How does the patent expiry impact SEROQUEL XR’s market?
Patent expiry leads to generic versions entering the market, reducing branded product prices and sales share.
2. What is the expected timeline for generic dominance?
Generic market share is projected to surpass 80% within the next 2-3 years, further suppressing prices.
3. Are there any ongoing R&D efforts for new formulations of quetiapine?
No significant new formulations are publicly announced; focus remains on improving existing generics' accessibility and affordability.
4. How does insurance coverage influence SEROQUEL XR pricing?
Insurance companies favor generics for cost savings, limiting premium pricing opportunities for branded versions.
5. What strategies could extend SEROQUEL XR’s market life?
Developing additional indications, formulations, or delivery methods could preserve some pricing power and market share.
References
[1] IQVIA. (2022). Pharmaceutical Market Reports.
[2] FDA. (2022). Drug Approvals and Patent Status.
[3] EvaluatePharma. (2022). Global Market Outlook for Atypical Antipsychotics.
More… ↓
