Share This Page
Drug Price Trends for SEMGLEE (YFGN)
✉ Email this page to a colleague
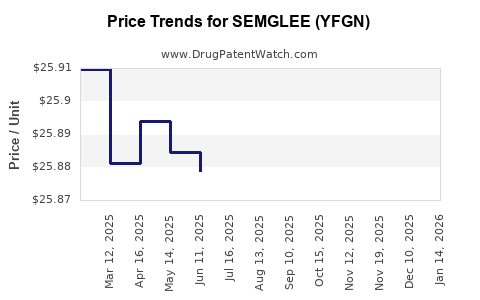
Average Pharmacy Cost for SEMGLEE (YFGN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SEMGLEE (YFGN) 100 UNIT/ML PEN | 83257-0012-33 | 25.81312 | ML | 2026-05-20 |
| SEMGLEE (YFGN) 100 UNIT/ML VL | 83257-0011-11 | 25.93320 | ML | 2026-05-20 |
| SEMGLEE (YFGN) 100 UNIT/ML PEN | 83257-0012-33 | 25.82406 | ML | 2026-04-22 |
| SEMGLEE (YFGN) 100 UNIT/ML VL | 83257-0011-11 | 25.89393 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SEMGLEE (YFGN): Market Analysis and Price Projections
What is SEMGLEE (YFGN)?
SEMGLEE (insulin glargine-yfgn) is a biosimilar insulin developed by Mylan (now Viatris), approved by the FDA in May 2020. It is used to control blood sugar levels in adults with diabetes mellitus and intended as a comparable alternative to Sanofi's Lantus (insulin glargine).
Market Context and Competition
Market Size and Growth
The global diabetes therapeutics market was valued at USD 60 billion in 2022 and projected to grow at a compound annual growth rate (CAGR) of 8% through 2030 [1]. Insulin products account for roughly 30% of this market, driven by rising prevalence of type 1 and type 2 diabetes.
Key Competitors
- Lantus (Sanofi): Dominant insulin glargine product, with approximately USD 7.4 billion in sales in 2022 [2].
- Basaglar (Eli Lilly/Biocon): Biosimilar insulin glargine, launched in 2016, holds around USD 1.5 billion annual revenue.
- Toujeo (Sanofi): U300 formulation, approximately USD 1 billion annually.
- Semglee (Viatris): Marketed since 2020, gains share as an affordable biosimilar.
Regulatory and Market Trends
- Increased adoption of biosimilars due to cost pressures.
- Payers and health systems favor biosimilars to reduce healthcare costs.
- Biocon's Lusduna and other biosimilars are entering markets, intensifying competition.
Market Penetration of SEMGLEE
- Sold in the U.S., Australia, Europe, and select emerging markets.
- Pricing strategies position it 10-20% below Lantus.
- Distribution channels include hospital pharmacies and outpatient clinics.
Pricing Overview
Current Pricing
- In the U.S., wholesale acquisition costs (WAC) for SEMGLEE are approximately USD 100-110 per 10 mL pen, about 10-15% lower than Lantus (~USD 125-135).
- The average retail price for Lantus exceeds USD 300 per 10 mL pen without insurance discounts.
Reimbursement and Payer Coverage
- Payers favor biosimilars for their cost savings.
- Contractual discounts and formularies influence actual patient prices.
Price Projections (2023-2030)
| Year | Estimated Wholesale Price (USD per 10 mL pen) | Notes |
|---|---|---|
| 2023 | 100-110 | Stabilized market share, price competition base |
| 2024 | 95-105 | Price pressures increase, volume growth continues |
| 2025 | 90-100 | Further price reductions, volume gains sustain revenue |
| 2026 | 85-95 | Entry of new biosimilars dampens pricing |
| 2027 | 80-90 | Market saturation, margins flatten |
| 2028-2030 | 75-85 | Competitive dynamics intensify, pricing stabilizes |
Factors Influencing Pricing
- Market Share: Predicted to increase 15-25% annually through 2025.
- Payer Negotiations: Will likely lead to further discounts.
- Regulatory Changes: Pathways for biosimilar substitution could pressure prices.
- Patent Expiry and Biosimilar Entries: Accelerate price competition, especially post-2024.
Revenue and Sales Forecasts
Assuming volume growth of 10% annually and incremental market share gains, revenue from SEMGLEE could reach USD 2-3 billion globally by 2030. The U.S. could generate over USD 1.5 billion annually, sustained by formulary preferences and generic price advantages.
Risks and Opportunities
Risks
- Entry of new biosimilars or branded competition.
- Regulatory barriers delaying biosimilar substitution.
- Payer resistance to price reductions.
Opportunities
- Expanding into emerging markets.
- Potential for formulary wins driven by cost savings.
- Development of next-generation biosimilars with cost advantages.
Key Takeaways
- SEMGLEE is positioned as a value-based biosimilar to Lantus with a current price differential of 10-15%.
- Market growth driven by diabetes prevalence and biosimilar acceptance.
- Long-term price decline projected due to intensifying competition and market saturation.
- Revenue forecasts indicate significant growth potential, primarily in the U.S. and Europe.
- Competitive pressures and regulatory changes pose ongoing risks.
FAQs
1. What is the primary competitive advantage of SEMGLEE?
It is priced 10-15% below Lantus, offering an affordable biosimilar alternative with comparable efficacy and safety profiles.
2. How does biosimilar market penetration affect prices?
Increased biosimilar adoption leads to lower prices for insulin products, especially as market share shifts from originators to biosimilars.
3. When will biosimilar insulin glargine prices stabilize?
Pricing is expected to stabilize around USD 75-85 per 10 mL pen by 2028, depending on market dynamics.
4. What factors could accelerate the decline in SEMGLEE's prices?
Introduction of new biosimilars, payer negotiations, regulatory approvals for substitution, and patent expirations.
5. How does regional regulation influence SEMGLEE's pricing?
Regions with supportive biosimilar policies, such as the U.S. and Europe, tend to see faster adoption and greater price competition.
References
[1] Grand View Research. (2023). Diabetes Therapeutics Market Size, Share & Trends.
[2] Evaluate Pharma. (2022). Global insulin market sales.
[3] IQVIA. (2022). U.S. Prescription Data.
[4] FDA. (2020). Approval for Semglee (insulin glargine-yfgn).
More… ↓
