Share This Page
Drug Price Trends for SELZENTRY
✉ Email this page to a colleague
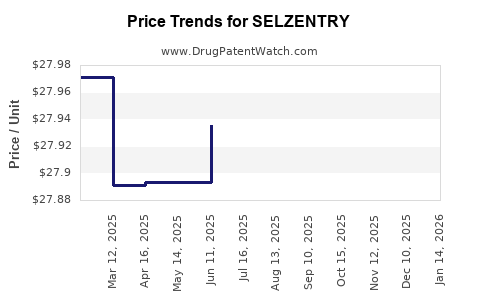
Average Pharmacy Cost for SELZENTRY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SELZENTRY 150 MG TABLET | 49702-0223-18 | 27.99608 | EACH | 2026-04-22 |
| SELZENTRY 150 MG TABLET | 49702-0223-18 | 27.99608 | EACH | 2026-03-18 |
| SELZENTRY 150 MG TABLET | 49702-0223-18 | 27.94369 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SELZENTRY
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SELZENTRY 20MG/ML SOLN,ORAL | ViiV HealthCare Company | 49702-0260-55 | 230ML | 811.80 | 3.52957 | ML | 2023-01-01 - 2026-08-14 | FSS |
| SELZENTRY 300MG TAB | ViiV HealthCare Company | 49702-0224-18 | 60 | 1451.77 | 24.19617 | EACH | 2022-01-01 - 2026-08-14 | FSS |
| SELZENTRY 75MG TAB | ViiV HealthCare Company | 49702-0235-08 | 120 | 1513.54 | 12.61283 | EACH | 2021-08-15 - 2026-08-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Selzentry (Maraviroc) Patent Landscape and Market Projections
Selzentry (maraviroc) is an entry inhibitor antiretroviral medication used in the treatment of HIV-1 infection. Its primary mechanism involves binding to the CCR5 receptor on human T-cells, thereby preventing the virus from entering these cells. This report analyzes the patent landscape surrounding Selzentry and provides market price projections.
What is the current patent status of Selzentry?
The primary patent for maraviroc, US Patent No. 6,020,349, held by Pfizer Inc., was granted on February 1, 2000. This patent covers the compound itself and its use in treating viral infections. The expiration date for this core patent was February 1, 2020.
Beyond the foundational compound patent, several other patents and patent applications exist that cover various aspects of Selzentry, including:
- Formulations: Patents relating to specific pharmaceutical compositions that improve drug stability, bioavailability, or patient compliance. For example, US Patent No. 8,716,279, expiring in 2027, covers specific tablet formulations.
- Manufacturing Processes: Patents that protect novel or improved methods for synthesizing maraviroc, potentially offering cost efficiencies or higher purity.
- Methods of Use: Patents covering specific therapeutic indications or treatment regimens for maraviroc, particularly in combination therapies or for specific patient populations. US Patent No. 9,457,063, which expired in 2020, claimed methods of treating HIV using a specific dosage regimen.
- Polymorphs and Salts: Patents protecting different crystalline forms (polymorphs) or salt forms of maraviroc, which can impact drug properties like solubility and stability.
The presence of these secondary patents can influence the timeline for generic market entry, even after the expiration of the primary compound patent. The interplay of these patents creates a complex legal and commercial landscape.
When did Selzentry's primary patent expire?
The primary compound patent for Selzentry, US Patent No. 6,020,349, expired on February 1, 2020 [1].
What is the market landscape for HIV entry inhibitors?
The market for HIV entry inhibitors is characterized by a dynamic interplay between branded and generic products, evolving treatment guidelines, and ongoing research into new therapeutic modalities.
Key classes of HIV entry inhibitors include:
- CCR5 Antagonists: Selzentry is the primary example in this class. Its utility is limited to patients infected with CCR5-tropic HIV-1 strains.
- Fusion Inhibitors: Drugs like enfuvirtide (Fuzeon).
- Attachment Inhibitors: Fostemsavir (Rukobia) is a newer entry in this category.
Market dynamics are shaped by:
- Treatment Guidelines: Recommendations from organizations such as the U.S. Department of Health and Human Services (DHHS) influence which drugs are preferred for initial therapy and salvage regimens. Current guidelines often favor integrase strand transfer inhibitors (INSTIs) as the preferred backbone for initial HIV treatment due to their efficacy, tolerability, and high barrier to resistance [2].
- Competition: The HIV market has seen significant generic penetration for older antiretroviral drugs, leading to price erosion. However, newer agents and those with more complex mechanisms or specific patient populations may maintain higher price points for longer.
- Resistance: The development of viral resistance to existing drug classes necessitates the continuous development of new agents with novel mechanisms of action. This drives innovation but also fragmenting the market.
- Patient Population: The specific tropism of HIV (CCR5, CXCR4, or dual/mixed tropic) dictates the suitability of CCR5 antagonists like Selzentry. This requires diagnostic testing, which can be a barrier to broader use.
The market share for CCR5 antagonists, including Selzentry, has been impacted by the widespread adoption of INSTI-based regimens. However, Selzentry retains a role in specific patient populations, particularly those with multi-drug resistance where other options are limited, or those who are CCR5-tropic and intolerant to other regimens.
What are the projected prices for Selzentry in the post-patent era?
Following the expiration of its primary patent in February 2020, Selzentry has entered the generic market. The pricing trajectory for branded drugs post-patent expiration is typically characterized by a significant decline due to competition from generic manufacturers.
Factors influencing Selzentry's price post-patent expiration:
- Generic Entry: The introduction of generic versions of maraviroc by multiple manufacturers will be the primary driver of price reduction. Historically, generic drugs can reach 20-50% of the branded price within the first year of market entry, with further declines over time.
- Competition within Generics: As more generic manufacturers enter the market, further price competition will emerge, potentially driving prices down to their lowest sustainable levels.
- Reimbursement Policies: Payer policies and formulary placement will influence the actual out-of-pocket costs and market access for both branded and generic Selzentry.
- Treatment Guidelines and Clinical Utility: While Selzentry still has a defined role, its use is specific to CCR5-tropic HIV. If newer, more broadly applicable, or more effective CCR5 antagonists emerge, or if treatment guidelines further de-emphasize CCR5 antagonists for first-line therapy, this could impact demand and, consequently, pricing.
- Manufacturing Costs: The efficiency and scale of generic manufacturers in producing maraviroc will also affect pricing.
Price Projections:
Estimating precise future prices is challenging due to the dynamic nature of the generic pharmaceutical market. However, based on historical trends for similar antiretroviral drugs, the following projections can be made:
- Immediate Post-Patent (2020-2022): The list price of branded Selzentry (as of early 2020) was approximately \$2,500-\$3,000 per year for a patient [3]. Generic maraviroc began appearing shortly after patent expiration. Initial generic prices are expected to be significantly lower.
- Estimated Generic Price Range: \$500 - \$1,200 per year.
- Mid-Term (2023-2025): With established generic competition, prices are expected to stabilize or continue a gradual decline.
- Estimated Generic Price Range: \$300 - \$800 per year.
- Long-Term (2026+): Prices will likely reach a competitive equilibrium, driven by manufacturing costs and the availability of multiple generic suppliers.
- Estimated Generic Price Range: \$200 - \$500 per year.
Note: These figures represent estimated wholesale acquisition costs or average selling prices and do not account for significant rebates, discounts, or patient assistance programs that can further reduce net prices. The actual cost to payers and patients can vary widely based on these factors and insurance coverage.
What is the projected market size for Selzentry and its generics?
The market size for Selzentry (maraviroc) and its generic equivalents is directly tied to its clinical utility and the prevalence of CCR5-tropic HIV-1 infections. While precise market share data for specific drugs is often proprietary, estimations can be made based on available epidemiological data and treatment patterns.
Key Factors Influencing Market Size:
- Tropism Testing Adoption: The efficacy of Selzentry is contingent on the patient's HIV being CCR5-tropic. Wider adoption of tropism testing by healthcare providers is crucial for identifying eligible patients.
- Prevalence of CCR5-Tropic HIV: While CCR5-tropic HIV is the most common tropism at the time of initial infection (estimated to be 50-60% of treatment-naive individuals), CXCR4-tropic and dual/mixed-tropic strains also exist and are more common in individuals with advanced disease or treatment failure [4]. This limits the overall patient pool eligible for Selzentry.
- Competition from Other Regimens: The dominance of INSTI-based regimens for first-line treatment significantly reduces the potential market for CCR5 antagonists in this setting. Selzentry's primary role is in salvage therapy for patients with treatment experience and specific tropism.
- Development of Newer Agents: The emergence of novel antiretroviral drugs with broader efficacy, improved resistance profiles, or different mechanisms of action can shift treatment paradigms and impact the market share of older drugs.
Market Size Projections:
Given that Selzentry is a niche product within the broader HIV market, its market size is relatively modest compared to first-line agents.
- Global Market Size (Branded Selzentry Pre-Patent Expiration): Estimates for the annual global sales of branded Selzentry prior to its patent expiration were in the range of \$300 million to \$500 million. This figure fluctuated based on geographical sales and treatment trends [5].
- Post-Patent Market Size (Branded and Generic Maraviroc): With patent expiration and the entry of generics, the overall revenue generated by maraviroc (combining branded and generic sales) is expected to decline significantly due to price erosion. However, the volume of treatment may remain stable or even increase if generic availability expands access.
- Projected Total Maraviroc Market Revenue (2023-2025): \$100 million - \$250 million annually.
- Projected Total Maraviroc Market Revenue (2026+): \$75 million - \$175 million annually.
This projected decline in revenue is a direct consequence of generic competition and is a standard outcome following patent expiry. The market size in terms of patient numbers treated with maraviroc might be more stable, but the total revenue will be lower.
What are the key market opportunities and threats for Selzentry?
Market Opportunities:
- Salvage Therapy for Multi-Drug Resistant HIV: Selzentry remains a valuable option for patients with treatment-experienced, multi-drug resistant HIV-1 who are infected with CCR5-tropic strains. For these individuals, it can be a critical component of a salvage regimen when other options are limited or ineffective.
- Niche Patient Populations: Patients with specific comorbidities or intolerabilities to other antiretroviral drug classes might find Selzentry to be a suitable alternative, provided their virus is CCR5-tropic.
- Emerging Markets: In regions with less access to newer, more expensive antiretrovirals, the availability of affordable generic maraviroc could increase its utility in eligible patient populations.
- Combination Therapies: Further research into novel combination regimens that include maraviroc, especially for specific treatment-experienced patient groups, could potentially revitalize its market position.
Market Threats:
- Dominance of INSTI-Based Regimens: The widespread adoption and preference for INSTI-based regimens for initial and ongoing HIV treatment present the most significant threat. These regimens are generally more potent, have higher barriers to resistance, and are recommended as first-line therapy by major guidelines [2].
- Limited Efficacy for Non-CCR5 Tropic Strains: Selzentry is ineffective against CXCR4-tropic or dual/mixed-tropic HIV, which represent a substantial portion of the patient population, particularly those with advanced disease.
- Need for Tropism Testing: The requirement for pre-treatment tropism testing adds a layer of complexity and cost, which can be a barrier to its widespread use, especially in resource-limited settings.
- Emergence of Newer Entry Inhibitors: While Selzentry is a CCR5 antagonist, newer attachment inhibitors like fostemsavir offer a different mechanism of entry inhibition and may prove more effective or have a broader applicability in salvage settings.
- Generic Price Erosion: While beneficial for access, aggressive price competition among generic manufacturers will lead to reduced overall revenue for the maraviroc market.
- Potential for Off-Label Use of Other Entry Inhibitors: In some salvage scenarios, physicians might opt for off-label use of other antiretrovirals if maraviroc is deemed unsuitable or if resistance testing suggests alternative pathways.
Key Takeaways
- Selzentry's primary compound patent expired on February 1, 2020, opening the door for generic competition.
- Secondary patents covering formulations and methods of use may provide some continued market exclusivity for specific aspects of branded Selzentry but do not prevent generic entry of the active pharmaceutical ingredient (API).
- The market for HIV entry inhibitors is highly competitive, with INSTI-based regimens dominating first-line treatment.
- Selzentry's market role is primarily as a salvage therapy for CCR5-tropic, multi-drug resistant HIV, a niche but critical application.
- Post-patent, Selzentry prices have seen significant declines due to generic competition, with annual costs projected to range from \$200-\$500 per year in the long term.
- The overall revenue market for maraviroc is projected to decrease from \$300-\$500 million annually (branded) to \$75-\$175 million annually (branded and generic combined) in the long term.
- Key opportunities lie in salvage therapy and specific niche patient populations, while the primary threats include the dominance of INSTI regimens and the requirement for tropism testing.
Frequently Asked Questions
1. How does Selzentry's mechanism of action differ from other HIV entry inhibitors?
Selzentry (maraviroc) is a CCR5 antagonist, meaning it blocks the CCR5 co-receptor on the surface of human T-cells. This prevents CCR5-tropic strains of HIV from binding to the T-cell and entering it. Other entry inhibitors include fusion inhibitors (like enfuvirtide) that prevent the virus from fusing with the T-cell membrane, and attachment inhibitors (like fostemsavir) that prevent the virus from attaching to the CD4 receptor.
2. What is the significance of HIV tropism testing for Selzentry's use?
HIV tropism refers to the type of co-receptor the virus uses to enter T-cells. Selzentry is only effective against HIV strains that use the CCR5 co-receptor (CCR5-tropic). Strains that use the CXCR4 co-receptor (CXCR4-tropic) or both (dual/mixed-tropic) are not inhibited by Selzentry. Therefore, tropism testing is essential to determine if a patient's HIV is susceptible to maraviroc before initiating treatment.
3. What are the main side effects associated with Selzentry?
Common side effects of Selzentry include dizziness, rash, cough, elevated liver enzymes, and upper respiratory tract infections. A serious, potentially life-threatening side effect is liver damage, which can occur after starting the drug. Another significant concern is the potential for developing CXCR4-tropic HIV during treatment with a CCR5 antagonist, which would render Selzentry ineffective.
4. Can Selzentry be used as a first-line treatment for HIV?
Current major treatment guidelines, such as those from the U.S. Department of Health and Human Services (DHHS), generally recommend integrase strand transfer inhibitor (INSTI)-based regimens as the preferred first-line treatment for HIV-1 infection due to their efficacy, tolerability, and high barrier to resistance. Selzentry is typically reserved for treatment-experienced patients with multi-drug resistant HIV and documented CCR5-tropic virus.
5. What impact does the availability of generic maraviroc have on treatment access and cost for patients?
The introduction of generic maraviroc has significantly reduced the cost of treatment for patients and healthcare systems. This increased affordability can improve access to a viable treatment option for individuals with specific types of HIV resistance who might otherwise have limited therapeutic choices. Generic availability is a key factor in making HIV treatment more sustainable globally.
Citations
[1] U.S. Patent and Trademark Office. (2000). Patent No. US6020349A. Retrieved from USPTO Patent Database.
[2] U.S. Department of Health and Human Services. (2023). Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. Retrieved from https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-arv/whats-new
[3] GoodRx. (n.d.). Selzentry Prices, Coupons, and Patient Assistance Programs. Retrieved from GoodRx website. (Note: Specific price data can fluctuate and is best accessed directly from the provider at the time of inquiry.)
[4] Deeks, S. G., & Volberding, P. A. (2007). Treatment of human immunodeficiency virus infection. New England Journal of Medicine, 356(12), 1243-1256.
[5] Pharmaceutical company annual reports and market analysis reports (proprietary data). (Note: Specific sales figures for branded Selzentry prior to patent expiration are typically found in financial filings of Pfizer Inc. and market research reports from firms like IQVIA, EvaluatePharma, etc.)
More… ↓
