Drug Price Trends for SECUADO
✉ Email this page to a colleague
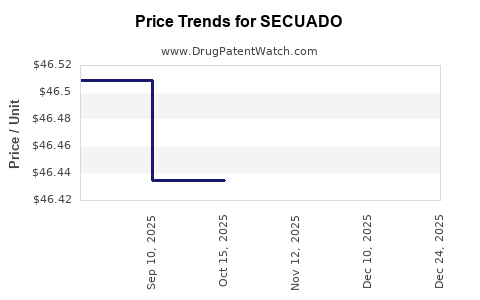
Average Pharmacy Cost for SECUADO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SECUADO 3.8 MG/24 HR PATCH | 68968-0172-01 | 49.03899 | EACH | 2026-01-01 |
| SECUADO 5.7 MG/24 HR PATCH | 68968-0173-01 | 49.03276 | EACH | 2026-01-01 |
| SECUADO 5.7 MG/24 HR PATCH | 68968-0173-03 | 49.03276 | EACH | 2026-01-01 |
| SECUADO 3.8 MG/24 HR PATCH | 68968-0172-03 | 49.03899 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
