Share This Page
Drug Price Trends for SE-NATAL
✉ Email this page to a colleague
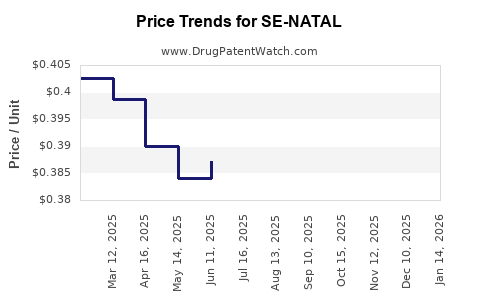
Average Pharmacy Cost for SE-NATAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SE-NATAL 19 TABLET | 13925-0116-01 | 0.47987 | EACH | 2026-04-22 |
| SE-NATAL 19 CHEWABLE TABLET | 13925-0117-01 | 0.49311 | EACH | 2026-04-22 |
| SE-NATAL 19 TABLET | 13925-0116-01 | 0.47645 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SE-NATAL Market Analysis and Financial Projection
What is SE-NATAL?
SE-NATAL is a brand-name drug primarily used as a prenatal vitamin supplement. It contains a combination of folic acid, iron, and other micronutrients designed to support pregnancy, fetal development, and maternal health. The drug targets expecting mothers, healthcare providers, and the broader women's health market.
Market Size and Growth Drivers
Current Market Overview
The global prenatal vitamin market was valued at approximately $4.8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 6% between 2023 and 2030, reaching around $8 billion. This growth reflects increasing awareness about maternal health, rising birth rates in emerging markets, and expanding healthcare access.
Key Market Drivers
- Rising global birth rates, notably in Asia and Africa.
- Increased focus on maternal and fetal health.
- Growing prevalence of prenatal deficiency awareness.
- Expansion of healthcare infrastructure and insurance coverage.
- Demand for fortified supplements with evidence-based formulations.
Market Segments
The segment includes prescription and over-the-counter (OTC) prenatal vitamins. SE-NATAL is positioned within the prescription segment, appealing to healthcare providers seeking high-quality, evidence-based formulations.
Competitive Landscape
Major competitors include brands like Elevit, One A Day Women's Prenatal, and Materna. Several generic and store brands also compete, especially in OTC markets. The market's fragmentation affects pricing strategies and profit margins.
Regulation Landscape
SE-NATAL’s classification varies by jurisdiction. In the United States, it falls under dietary supplements or drug regulation depending on claims and formulation. Regulatory approval for prescription use typically requires submission to agencies such as the FDA, including safety, efficacy, and manufacturing standards.
Internationally, regulatory pathways differ:
- EU: Requires Marketing Authorization through the European Medicines Agency (EMA).
- China and India: Registration with respective health authorities, often with local manufacturing requirements.
Price Projections (2023-2030)
Current Pricing
- Prescription version: Range of $15-$30 per month (U.S. OTC equivalent).
- Packaging: Typically sold in 30- to 90-day supplies.
- Wholesale prices: Approximately 20-30% below retail.
Future Pricing Trends
Pricing is influenced by:
- Regulatory costs: Potential increases if expanded to require approval as a drug.
- Market competition: Entry of generics could lower prices by 10-20% over five years.
- Manufacturing costs: Stabilize or decrease with scale, affecting margins.
- Premium formulations: SE-NATAL variants with added nutrients or proprietary delivery systems could maintain higher prices (~$30-$45/month).
Price Outlook Summary
| Year | Estimated Retail Price Range | Notes |
|---|---|---|
| 2023 | $15-$30 | Current range, depending on market and region. |
| 2025 | $12-$25 | Increased generic competition may reduce prices. |
| 2030 | $12-$22 | Market stabilization, potential inflation adjustments. |
Market Entry and Pricing Strategies
For new entrants or existing manufacturers:
- Price competitively to capture OTC market share.
- Offer bundled packages with other prenatal and maternal health products.
- Invest in branding emphasizing clinical efficacy, possibly justifying higher pricing.
- Leverage direct-to-consumer digital marketing channels.
Risks and Opportunities
Risks:
- Regulatory delays or reclassification impacting pricing.
- Emergence of cheaper generics leading to margin compression.
- Changes in healthcare policy affecting reimbursement and prescribing patterns.
Opportunities:
- Expansion into emerging markets with low current penetration.
- Development of higher-margin, specialized formulations.
- Strategic partnerships with healthcare providers and insurers.
Key Takeaways
- The global prenatal supplement market is growing steadily, driven by increased health awareness and demographic trends.
- SE-NATAL’s positioning as a prescription product suggests higher margins but also higher regulatory and clinical complexities.
- Pricing will likely decline modestly through generics but can remain elevated with product differentiation.
- Market entry strategies should focus on competitive pricing, branding, and regional regulatory navigation.
FAQs
Q1: What separates SE-NATAL from other prenatal supplements?
SE-NATAL likely contains specific formulations or proprietary ingredients aimed at targeted health outcomes, potentially supported by clinical trials.
Q2: How does regulatory status influence pricing?
Strict drug regulations and required clinical trials increase compliance costs, supporting higher prices initially. Regulatory reclassification could lead to price reductions.
Q3: What are the primary markets for SE-NATAL?
The U.S., EU, and emerging markets like China and India. Market penetration varies depending on approval status and infrastructure.
Q4: Can manufacturing costs impact price projections?
Yes, scale efficiencies and raw material prices influence production costs, affecting retail price margins.
Q5: How does competition affect future pricing?
Increased competition from generics reduces prices, while differentiated, premium formulations can sustain higher margins.
References
[1] BCC Research. "Prenatal Vitamins Market Analysis." 2022.
[2] Grand View Research. "Prenatal Vitamins Market Size, Share & Trends." 2023.
[3] FDA. "Guidance for Industry: Dietary Supplements." 2021.
[4] European Medicines Agency. "Regulatory Framework for Medicinal Products." 2022.
[5] Statista. "Global Prenatal Supplement Market Revenue Forecast." 2023.
More… ↓
