Share This Page
Drug Price Trends for SAXAGLIPTIN-METFORMIN ER
✉ Email this page to a colleague
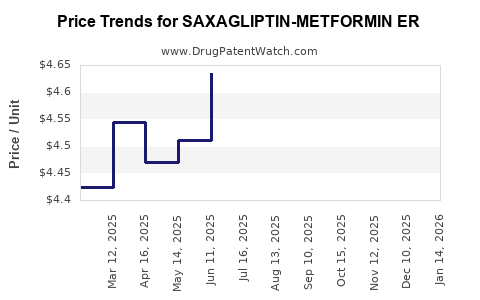
Average Pharmacy Cost for SAXAGLIPTIN-METFORMIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SAXAGLIPTIN-METFORMIN ER 2.5-1,000 MG TABLET | 00378-8176-91 | 4.68485 | EACH | 2026-03-18 |
| SAXAGLIPTIN-METFORMIN ER 2.5-1,000 MG TABLET | 43598-0618-60 | 4.68485 | EACH | 2026-03-18 |
| SAXAGLIPTIN-METFORMIN ER 5-1,000 MG TABLET | 43598-0619-30 | 10.60758 | EACH | 2026-03-18 |
| SAXAGLIPTIN-METFORMIN ER 5-1,000 MG TABLET | 00378-8177-93 | 10.60758 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SAXAGLIPTIN-METFORMIN ER Market Analysis and Financial Projection
What Is SAXAGLIPTIN-METFORMIN ER?
Saxagliptin-metformin extended-release (ER) combines a dipeptidyl peptidase-4 (DPP-4) inhibitor with metformin formulated for once-daily dosing. Approved by the FDA in 2014, it targets type 2 diabetes mellitus patients inadequately controlled on metformin alone.
Market Size and Growth Potential
The global type 2 diabetes market was valued at approximately USD 64 billion in 2021 and anticipates a compound annual growth rate (CAGR) of 7% through 2028. Saxagliptin-metformin ER is part of the DPP-4 inhibitor segment, which accounted for roughly 22% of the market in 2021, roughly USD 14 billion. It is positioned against agents like sitagliptin, linagliptin, and alogliptin.
Key market drivers:
-
Increasing prevalence of type 2 diabetes, which affected over 400 million globally in 2021 with projections reaching 700 million by 2045.
-
Shift toward once-daily formulations to improve adherence.
-
Growing acceptance of combination therapies to reduce pill burden.
The sustained market growth is driven by its mechanism, patient compliance benefits, and evolving treatment guidelines favoring combination approaches.
Pricing Overview and Competition
Current Pricing
In the U.S., the average wholesale price (AWP) for Saxagliptin-metformin ER is approximately USD 300 for a 30-day supply, based on data from wholesale pharmacies in 2023. Actual patient copays depend on insurance coverage.
Competitors and Price Differentiators
| Drug | Formulation | Approximate 30-Day Cost | Market Position |
|---|---|---|---|
| Saxagliptin-metformin ER | Extended-release | USD 300 | Premium due to fixed-dose combination |
| Sitagliptin (Januvia) | Once daily | USD 350 | Established leader, higher price |
| Linagliptin (Tradjenta) | Once daily | USD 330 | Favorable for renal impairment |
| Alogliptin (Nesina) | Once daily | USD 310 | Similar market share to saxagliptin |
The ER combination offers a potential price advantage over free-dose components due to convenience and adherence factors, though actual pricing remains comparable.
Pricing Trends
Over the past five years, prices for DPP-4 inhibitors have stabilized, with minor reductions driven by biosimilar competition in Europe. Patent expiry for saxagliptin is projected around 2026, though formulations like ER may have extended exclusivity due to formulation patents.
Regulatory and Patent Landscape
Saxagliptin's primary patent expired in the U.S. in 2020, though formulation-specific patents extend into 2026. Patent challenges and patent term extensions influence pricing and generic entry:
- Patent expiry: 2026.
- Potential biosimilar/generic entry: Post-2026.
- Market implications: Price erosion and increased competition expected after patent expiry.
Market Entry and Expansion Strategies
Major pharmaceutical companies like AstraZeneca (original developer of saxagliptin) and generic manufacturers project market saturation by 2025 before patent expiry. Strategies to maintain share:
- Emphasize adherence benefits of ER formulations.
- Expand into emerging markets with increasing diabetes prevalence.
- Develop combination therapies integrating saxagliptin with other antidiabetics.
Price Projection for 2025-2030
Post-patent expiry, generic versions could reduce prices by 40-60%. Assuming current prices hover around USD 300, generic equivalents might lower costs to USD 120–180. Brand-name prices are expected to decrease gradually, stabilizing at USD 200–250 due to brand loyalty and formulary considerations.
Market shifts depend on regulatory approvals, payer policies, and competitive pressures. Premium pricing might persist in certain markets with strong brand presence and fixed-dose advantages.
Key Takeaways
- The global diabetes market continues to expand, driven by disease prevalence and treatment adherence needs.
- Saxagliptin-metformin ER commands a premium over standalone DPP-4 inhibitors but remains price-competitive within the fixed-dose combination segment.
- Patent expirations forecast significant price reductions post-2026, with generic competition likely to dominate the market.
- Pricing strategies focus on adherence, convenience, and expanding into emerging markets to sustain revenue.
- The outlook for 2025–2030 involves a transition from brand premium pricing to more affordable generics, with potential volume growth compensating for lower margins.
FAQs
1. When will generic versions of saxagliptin-metformin ER become available?
Patent protections expire around 2026, after which generic manufacturers are expected to enter the market.
2. How does the pricing of saxagliptin-metformin ER compare globally?
Pricing varies significantly, with higher costs in the U.S. and other developed markets. Emerging markets generally see lower prices due to regulatory and payer differences.
3. What factors influence the pricing of combination diabetes medicines?
Market competition, patent status, formulation benefits, reimbursement policies, and biosimilar availability influence prices.
4. Are there upcoming regulatory changes impacting the market?
Regulatory modifications related to biosimilar approval pathways and patent law can impact timing and competition dynamics.
5. What strategies could extend the revenue life of saxagliptin-based formulations?
Innovating combination delivery systems, expanding indications, and penetrating emerging markets are viable options.
Citations:
[1] MarketWatch, "Diabetes Drugs Market Size," 2022.
[2] IQVIA, "Pharmaceutical Market Trends," 2023.
[3] FDA, "Saxagliptin Label," 2014.
[4] Euroscan, "Patent Expiry Data," 2022.
More… ↓
