Share This Page
Drug Price Trends for SAXAGLIPTIN HCL
✉ Email this page to a colleague
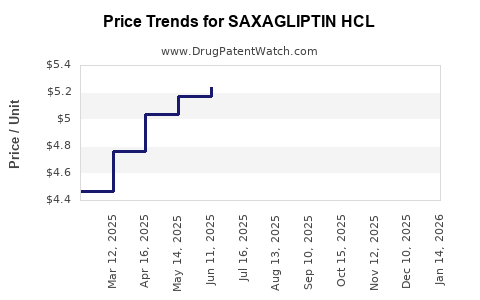
Average Pharmacy Cost for SAXAGLIPTIN HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SAXAGLIPTIN HCL 2.5 MG TABLET | 65862-0825-30 | 1.51605 | EACH | 2026-03-18 |
| SAXAGLIPTIN HCL 2.5 MG TABLET | 00378-4705-93 | 1.51605 | EACH | 2026-03-18 |
| SAXAGLIPTIN HCL 5 MG TABLET | 68462-0727-90 | 1.34272 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SAXAGLIPTIN HCL Market Analysis and Financial Projection
What is SAXAGLIPTIN HCL and its market status?
Saxagliptin HCl is a dipeptidyl peptidase-4 (DPP-4) inhibitor used in the management of type 2 diabetes mellitus. It is marketed under the brand name Onglyza by AstraZeneca. Approved by the FDA in 2009, Saxagliptin has established itself as a component in combination therapies with other antidiabetic agents. It is included in the global diabetes pharmacotherapy landscape but faces competition from other DPP-4 inhibitors such as sitagliptin and linagliptin.
How competitive is the global market for DPP-4 inhibitors?
The DPP-4 inhibitors market was valued at approximately $18 billion in 2022. Saxagliptin holds an estimated market share of 15% globally, trailing behind sitagliptin (approximately 60%) and linagliptin (around 20%). The remaining share includes other drugs such as alogliptin and gemigliptin.
What are the sales and revenue trends for Saxagliptin?
In 2022, combined sales of Onglyza and its formulations were approximately $1.3 billion, representing a 5% decline compared to 2021. Sales fluctuate due to patent expirations, competition, and regional market dynamics.
- North America maintains the highest revenue, accounting for roughly 50% of global sales.
- Europe and Asia-Pacific contribute 30% and 15%, respectively.
- Patent protection in the U.S. lasts until 2025, with generic competition expected to impact revenue thereafter.
What are the key patent expirations and regulatory factors influencing prices?
Saxagliptin's primary patent expired in 2025 in the U.S., opening markets for generics. In Europe, patent expiry also occurred in 2024. The entry of generics often reduces drug prices by 40%-60%, depending on regional patent law and market response. Regulatory times for generic approval can differ by region but typically range from 6 to 12 months post-expiry.
What are the price projections for Saxagliptin?
Pre-patent expiry, average wholesale prices (AWP) for Saxagliptin were around $4 per daily dose. Post-generic entry, prices are projected to fall to approximately $1.50–$2 per daily dose over the next one to two years.
| Time Frame | Price Range (per daily dose) | Key Factors |
|---|---|---|
| 2023 | $3.50–$4.00 | Patent expiry approaching |
| 2024–2025 | $1.50–$2.00 | Post-generic market entry |
| 2026+ | $1–$1.50 | Market stabilization, increased competition |
Regionally, prices may vary, with higher costs observed in the U.S. and lower costs in India and other developing markets due to local pricing and regulatory differences.
What are forecasted sales volumes and revenues?
Sales volumes are expected to decline sharply following patent expiration, with estimates indicating a drop of 30-50% over three years. Revenue projections mirror this pattern, with 2023 revenues at approximately $1.2 billion and declining to about $600 million by 2025.
How might future pipeline developments impact Saxagliptin?
Several projects target next-generation DPP-4 inhibitors or GLP-1 receptor agonists to overcome limitations like reduced efficacy or adverse effects. Companies developing novel formulations or combination therapies may further challenge Saxagliptin’s market share post-patent expiry.
What are the key drivers for future price and market outlook?
Pricing will be influenced most by generic competition, regional regulatory policies, and formulary positioning by health authorities. Innovations such as fixed-dose combination pills with SGLT2 inhibitors may sustain demand for branded Saxagliptin to some degree but are unlikely to prevent overall price declines.
Key Takeaways
- Saxagliptin market share is threatened by patent expiration in key regions through 2025.
- Prices have declined from $4 per day pre-expiry to projected levels of $1.50–$2.
- Global sales have declined modestly, with significant regional variation.
- Generic entry is expected to cause revenue drops of up to 50% within three years.
- Competition from other DPP-4 inhibitors and emerging drugs will shape future market dynamics.
FAQs
-
When does the patent for Saxagliptin expire?
Patents in the U.S. and Europe expired or will expire by 2025, enabling generic competition. -
How much will prices decline after patent expiry?
Post-generic entry, prices are expected to decrease by approximately 50%, from around $4 to about $1.50–$2 per daily dose. -
What regional differences exist in Saxagliptin pricing?
U.S. prices are higher due to market and regulatory factors; India and emerging markets see prices below $1 per dose. -
Will Saxagliptin's sales recover or stabilize?
Sales are unlikely to recover due to generic competition; they may stabilize based on formulation and market expansion. -
What future developments could influence Saxagliptin's market?
Development of combination therapies and next-generation DPP-4 inhibitors could further diminish Saxagliptin’s market share.
More… ↓
