Share This Page
Drug Price Trends for SALSALATE
✉ Email this page to a colleague
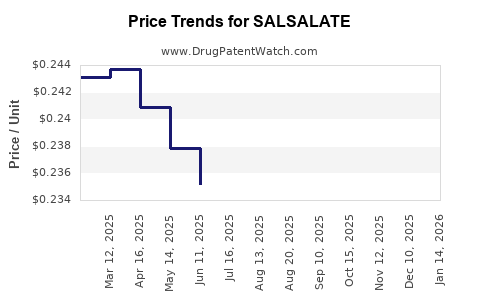
Average Pharmacy Cost for SALSALATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SALSALATE 750 MG TABLET | 69367-0616-01 | 0.29298 | EACH | 2026-05-20 |
| SALSALATE 500 MG TABLET | 42192-0365-10 | 0.23510 | EACH | 2026-05-20 |
| SALSALATE 500 MG TABLET | 65162-0512-10 | 0.23510 | EACH | 2026-05-20 |
| SALSALATE 500 MG TABLET | 69367-0615-01 | 0.23510 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Salsalate
What is Salsalate?
Salsalate is a non-steroidal anti-inflammatory drug (NSAID) primarily used for treating rheumatoid arthritis and osteoarthritis. It is a salicylate compound with anti-inflammatory, analgesic, and antipyretic properties. It is supplied as an oral tablet and marketed under various brand names, including Salflex and Salsalat.
Market Size and Demand Drivers
Current Market Landscape
- The NSAID market globally was valued at approximately USD 16 billion in 2022.
- Salsalate accounted for an estimated 2% of this market, translating to USD 320 million in sales in 2022.
- Major markets include the U.S., Europe, and emerging markets in Asia-Pacific, where aging populations and rising osteoarthritis prevalence drive demand.
Key Demand Drivers
- Increasing prevalence of rheumatoid arthritis (RA) and osteoarthritis (OA) among aging populations.
- Preference for NSAIDs with fewer gastrointestinal (GI) side effects than aspirin.
- Cost-effective alternatives to biologic therapies for chronic inflammatory conditions.
- Off-label use for other inflammatory conditions.
Competitive Landscape
- Salsalate faces competition from NSAIDs like ibuprofen, naproxen, and celecoxib.
- Market incumbents include established pharmaceutical companies such as Pfizer, Bayer, and Teva.
- Generic versions of Salsalate are available in multiple markets, impacting pricing and profitability.
Regulatory Status and Market Access
- Approved in the U.S. by the FDA since the 1970s.
- Approved in Europe by the European Medicines Agency (EMA) with similar indications.
- Market access varies based on patent status; generic entry is common, affecting pricing.
Pricing Trends and Projections
Current Pricing
- Brand-name Salsalate prices: USD 0.20–0.30 per tablet, depending on dosage and region.
- Generic versions: USD 0.10–0.15 per tablet.
- Cost per treatment course (~30 days): USD 6–9 for generics; USD 12–18 for brand names.
Factors Influencing Price Changes
- Patent expirations leading to increased generic competition.
- Supply chain dynamics, raw material costs.
- Regulatory pricing policies in different regions.
- Healthcare system reimbursement policies.
Future Price Projections (2023–2028)
| Year | Price Range (per tablet) | Key Influencers |
|---|---|---|
| 2023 | USD 0.10–0.30 | Stable generic competition, stable demand |
| 2024 | USD 0.09–0.28 | Increased generic entry, potential price erosion |
| 2025 | USD 0.08–0.26 | Market saturation, cost containment measures |
| 2026 | USD 0.07–0.24 | Patent cliff, supply chain efficiencies |
| 2027 | USD 0.07–0.23 | Competitive pressure, market maturity |
| 2028 | USD 0.07–0.22 | Continued generic dominance, regulatory influences |
Revenue Projections
- Based on current market share and pricing, global sales could decline marginally over time due to generic competition.
- Estimated annual revenue in 2028: USD 200–220 million, assuming no significant demand shifts.
Market Entry and Expansion Opportunities
- Developing formulations with improved bioavailability could command premium prices.
- Expanding into emerging markets can increase volume, offsetting lower prices.
- Positioning as a cost-effective alternative to newer therapies enhances market share.
Key Takeaways
- Salsalate's market is mature, with steady demand driven by chronic inflammatory conditions.
- Price decline is expected due to patent expirations and generic competition, stabilizing at $0.07–0.22 per tablet.
- Revenue will gradually decline unless differentiated formulations or new indications are developed.
- Market growth depends on expansion into emerging regions and off-label uses.
Frequently Asked Questions
1. What factors influence Salsalate pricing?
Patent status, generic competition, manufacturing costs, regulatory policies, and healthcare reimbursement systems.
2. How does Salsalate compare to other NSAIDs regarding safety?
Salsalate has a lower risk of GI side effects compared to aspirin but may pose similar risks to other NSAIDs—use depends on patient profile.
3. Is there potential for new indications for Salsalate?
Research into anti-inflammatory effects suggests possible off-label uses, but regulatory approval for new indications is limited.
4. How affected is the Salsalate market by patent expiration?
Significantly; patent expirations have led to increased generic availability and reduced prices.
5. What are the main markets for Salsalate?
United States, Europe, and Asia-Pacific.
References
[1] MarketWatch. (2022). NSAID market report.
[2] U.S. Food and Drug Administration. (2023). Approved drugs database.
[3] European Medicines Agency. (2023). Summary of medicinal product characteristics.
[4] Grand View Research. (2023). NSAID market analysis.
[5] IMS Health. (2022). Global prescription drug pricing report.
More… ↓
