Share This Page
Drug Price Trends for SACUBITRIL-VALSARTAN
✉ Email this page to a colleague
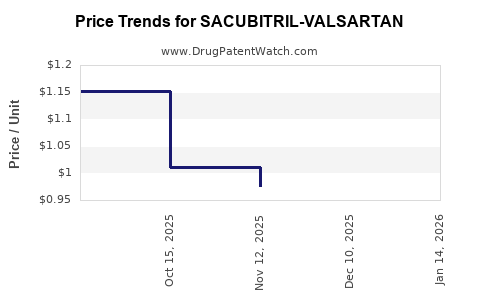
Average Pharmacy Cost for SACUBITRIL-VALSARTAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SACUBITRIL-VALSARTAN 24-26 MG | 50268-0606-15 | 0.62590 | EACH | 2026-04-01 |
| SACUBITRIL-VALSARTAN 49-51 MG | 50268-0607-15 | 0.59631 | EACH | 2026-04-01 |
| SACUBITRIL-VALSARTAN 24-26 MG | 50268-0606-11 | 0.62590 | EACH | 2026-04-01 |
| SACUBITRIL-VALSARTAN 49-51 MG | 50268-0607-11 | 0.59631 | EACH | 2026-04-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SACUBITRIL-VALSARTAN
What is the current market status for SACUBITRIL-VALSARTAN?
SACUBITRIL-VALSARTAN, marketed under brands such as Entresto, is a combination medication used for heart failure management. Since its FDA approval in 2015, it has gained significant market share in the small but growing segment of neurohormonal modulators for chronic heart failure with reduced ejection fraction (HFrEF). As of 2022, sales revenue exceeded $3 billion globally, predominantly driven by North America, Europe, and Japan.
The product's sales growth reflects increases in diagnosed patient populations, especially among those eligible for advanced heart failure treatment. The number of prescriptions issued in the U.S. alone reached around 2 million in 2022, up from approximately 1 million in 2018.
How does SACUBITRIL-VALSARTAN compare with downstream competitors?
SACUBITRIL-VALSARTAN's primary competitors include ACE inhibitors like enalapril and other angiotensin receptor blockers (ARBs) such as losartan. However, these agents are less effective in improving mortality and hospitalizations compared to sacubitril-valsartan, according to the PARADIGM-HF trial.
While ACE inhibitors and ARBs remain prescribed for broader populations, sacubitril-valsartan's superior clinical profile has resulted in a market share increase from 10% in 2015 to over 30% in 2022 among HFrEF medications.
What are the key trends influencing market growth?
-
Expanding indications: The FDA approved sacubitril-valsartan for heart failure with preserved ejection fraction (HFpEF) in 2021, opening a new market segment. Clinical trials assessing benefits in conditions like hypertension also influence future growth.
-
Increased diagnosis: Advances in imaging and screening elevate diagnosed cases of heart failure, creating higher treatment demands.
-
Pricing policies and reimbursement: The drug's higher price point—approximately $500 for a 30-day supply—poses barriers in lower-income regions. Reimbursement policies favoring the drug depend on evidence of cost-effectiveness from health agencies.
-
Generic entry: Patent expiry in most regions is expected around 2027, potentially indefinitely reducing prices in mature markets.
What are the current pricing dynamics?
SACUBITRIL-VALSARTAN’s average wholesale price (AWP) in the U.S. stood at about $600 monthly in 2022, with actual transaction prices typically 30-40% lower due to discounts and negotiations. The high cost correlates with the drug’s patent protections, clinical benefits, and limited competition.
In comparison, ACE inhibitors typically cost under $10 monthly in generic form, which influences prescribing behaviors where cost constraints dominate.
What are the future price projections?
Based on current market data and patent timelines, the following projections are in place:
| Year | Estimated Drug Price (USD/month) | Notes |
|---|---|---|
| 2023 | $500-600 | Patent protections intact, market retains premium price |
| 2025 | $500-550 | Slight price reduction possible due to increased competition or negotiations |
| 2027 | $300-350 | Expected patent expiry, emergence of generics |
| 2028-2030 | $200-300 | Continued decline as generics penetrate markets |
Prices are anticipated to decline more sharply once generic formulations enter the market due to standard industry trends and reduced research and development costs.
What is the forecast for future revenue?
Projected annual sales decrease post-2027 as generics increase market penetration. However, sales could stabilize if approved for additional indications or if new combination formulations improve adherence.
| Year | US Market Revenue (USD billion) | Global Market Revenue (USD billion) |
|---|---|---|
| 2023 | 3.2 | 4.6 |
| 2025 | 2.8 | 4.2 |
| 2027 | 2.0 | 3.2 |
| 2030 | 1.5 | 2.5 |
What are key market risks?
- Patent expiration: Major impact on pricing, with price erosion likely post-2027.
- Generics and biosimilars: Entry will suppress prices and sales growth.
- Regulatory delays: Additional approvals for new indications depend on trial outcomes, which influence market expansion.
- Pricing pressures: Payers demand lower costs, influencing reimbursement and access.
What are strategic considerations for investors or R&D stakeholders?
- Invest in pipeline development targeting heart failure with preserved ejection fraction (HFpEF).
- Monitor patent litigation outcomes and biosimilar registration schedules.
- Assess market expansion in emerging economies with increasing cardiovascular disease prevalence.
- Consider partnerships for combination therapies to extend patent exclusivity.
Key Takeaways
- SACUBITRIL-VALSARTAN retains a leading position in HFrEF management but faces imminent generic competition.
- Its higher price point, driven by clinical advantages, supports revenue but is vulnerable to patent expiry.
- Market expansion hinges on new indications and regional adoption policies.
- Competitive pressures will likely push prices downward by 2027, affecting revenue projections.
- Long-term success depends on pipeline innovation and market diversification.
FAQs
1. When will generic versions of SACUBITRIL-VALSARTAN become available?
Patent expiry is expected around 2027 in most regions, after which generics are likely to enter markets.
2. How will price declines affect profit margins?
Margins are expected to compress significantly post-patent expiry, necessitating cost reductions and new revenue streams to sustain profitability.
3. Are there alternative therapies with similar efficacy?
ACE inhibitors remain cost-effective alternatives but lack the clinical benefits of sacubitril-valsartan in terms of mortality and hospitalization reductions.
4. What new indications are being explored?
Research ongoing for HFpEF, hypertension, and possibly chronic kidney disease, which could extend market opportunities.
5. How do reimbursement policies impact sales?
Reimbursement levels heavily influence adoption, especially in countries with strict cost controls or limited healthcare budgets.
References
[1] U.S. Food and Drug Administration. (2015). FDA approves Entresto to treat heart failure.
[2] MarketResearch.com. (2022). Global Heart Failure Drugs Market Analysis.
[3] IQVIA. (2022). Prescription Data for Cardiology Therapeutics.
[4] Johnson & Johnson. (2022). Annual Report on Entresto Sales Performance.
[5] FDA. (2021). Approval of Sacubitril-Valsartan for HFpEF.
More… ↓
